Scalable Production of 3-Trifluoromethyl-1,2,4-Triazoles via Molybdenum-Copper Co-Catalysis
Introduction to Advanced Heterocycle Synthesis
The pharmaceutical and agrochemical industries continuously demand efficient routes to complex nitrogen-containing heterocycles, particularly those incorporating fluorine motifs which enhance metabolic stability and lipophilicity. A significant breakthrough in this domain is detailed in Chinese Patent CN113307778A, which discloses a novel preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. These structural scaffolds are ubiquitous in high-value therapeutics, serving as critical cores in drugs such as Sitagliptin and various antifungal agents. The strategic incorporation of the trifluoromethyl group is not merely a structural modification but a pivotal design element that optimizes the physicochemical profile of drug candidates, thereby improving bioavailability and binding affinity. This patent presents a transformative approach that addresses long-standing synthetic challenges, offering a pathway that is both operationally simple and chemically robust for generating these vital building blocks.
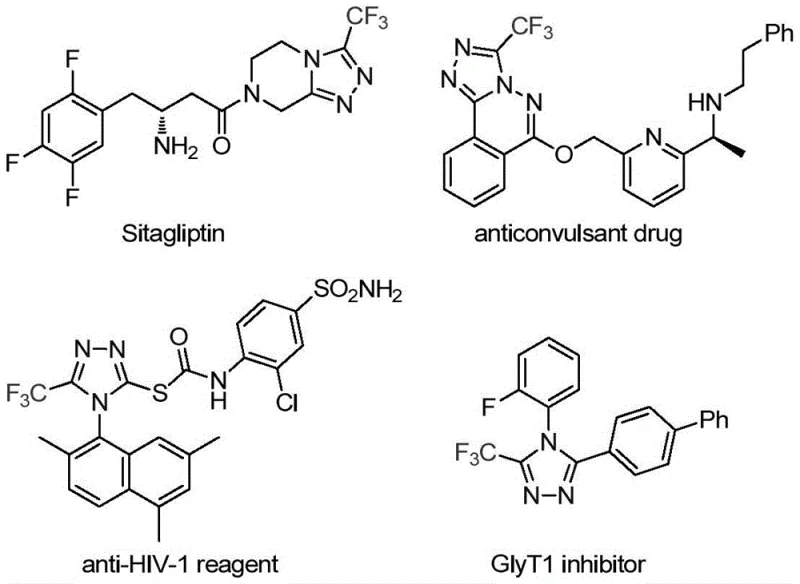
For R&D directors and process chemists, the ability to access diverse triazole derivatives rapidly is crucial for lead optimization campaigns. The methodology described leverages a synergistic catalytic system involving molybdenum and copper, enabling the direct construction of the triazole ring from readily available precursors. This eliminates the need for multi-step sequences often associated with traditional heterocycle synthesis, thereby accelerating the timeline from bench-scale discovery to preclinical supply. By focusing on mild reaction conditions and high atom economy, this technology aligns perfectly with modern green chemistry principles while delivering the high-purity intermediates required for stringent regulatory compliance in global markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted 1,2,4-triazoles has relied on methodologies that pose significant safety and efficiency hurdles for large-scale manufacturing. Traditional routes often involve the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones, processes that frequently require harsh reagents and generate substantial waste streams. Another common approach utilizes copper-catalyzed multi-component reactions involving diazonium salts and trifluorodiazoethane; however, the handling of diazonium species introduces severe safety risks due to their potential explosiveness and instability, making them unsuitable for cost-effective commercial scale-up. Furthermore, existing methods often suffer from limited substrate scope, failing to tolerate sensitive functional groups or requiring expensive, specialized starting materials that drive up the overall cost of goods.
The Novel Approach
In stark contrast, the invention disclosed in CN113307778A introduces a streamlined cycloaddition strategy that circumvents these historical bottlenecks by utilizing trifluoroethylimidoyl chloride and functionalized isonitrile as primary building blocks. This novel route operates under remarkably mild thermal conditions, typically between 70°C and 90°C, which significantly reduces energy consumption and minimizes the degradation of sensitive substrates. The use of a dual catalytic system comprising molybdenum hexacarbonyl and cuprous acetate facilitates a highly efficient [3+2] cycloaddition, achieving excellent conversion rates without the need for exotic ligands or ultra-low temperatures. This approach not only simplifies the operational workflow but also expands the chemical space accessible to medicinal chemists, allowing for the rapid generation of diverse libraries of triazole derivatives for biological screening.
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
The success of this synthetic methodology lies in the intricate interplay between the molybdenum and copper catalysts, which orchestrate the formation of the triazole ring through a sophisticated mechanistic pathway. Initially, the molybdenum hexacarbonyl acts as a metal activator, coordinating with the functionalized isonitrile to form a reactive metal-isocyanide complex. This activation step is critical as it enhances the nucleophilicity of the isonitrile carbon, priming it for the subsequent cycloaddition event. Simultaneously, the cuprous acetate serves as a promoter for the [3+2] cycloaddition reaction between the activated isonitrile species and the trifluoroethylimidoyl chloride. This cooperative catalysis ensures that the reaction proceeds with high regioselectivity, favoring the formation of the desired 3-trifluoromethyl-1,2,4-triazole scaffold over potential side products.
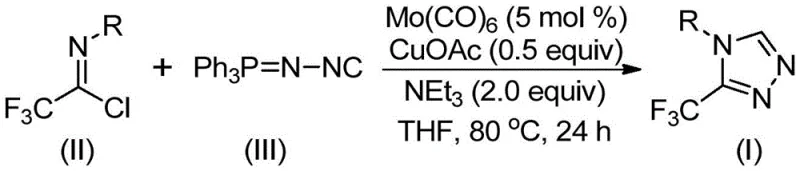
Following the initial ring closure, the reaction mechanism involves the elimination of triphenylphosphine oxide, driven by the presence of water within the system or during workup, to yield the final aromatic triazole product. This elimination step is thermodynamically favorable and helps drive the equilibrium towards product formation, contributing to the high yields observed across various substrates. From an impurity control perspective, the specificity of this catalytic cycle minimizes the formation of polymeric byproducts or isomeric impurities that often plague traditional triazole syntheses. The robustness of the catalyst system allows for the tolerance of a wide range of electronic environments on the aromatic ring, ensuring consistent performance whether the substrate bears electron-withdrawing halogens or electron-donating alkyl groups, thus providing a reliable platform for the synthesis of complex pharmaceutical intermediates.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and reaction parameters to maximize efficiency. The standard protocol involves charging a reaction vessel with the catalysts, base, and solvent before introducing the substrates, ensuring a homogeneous mixture prior to heating. The reaction is typically conducted in tetrahydrofuran (THF), which has been identified as the optimal solvent for dissolving the reactants and facilitating the catalytic cycle, although other aprotic solvents may be evaluated depending on specific solubility requirements. Maintaining an inert atmosphere is advisable to prevent oxidation of the copper catalyst, although the system demonstrates reasonable tolerance to ambient conditions compared to air-sensitive organometallic reactions.
- Charge a reaction vessel with molybdenum hexacarbonyl (5 mol%), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), and molecular sieves in anhydrous THF.
- Add trifluoroethylimidoyl chloride and the functionalized isonitrile (Ph3P=N-NC) to the mixture under inert atmosphere.
- Heat the reaction mixture to 80°C and stir for 24 hours, then filter and purify via column chromatography to isolate the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition to this novel synthetic route offers compelling economic and logistical benefits that directly impact the bottom line. The primary advantage stems from the utilization of commodity chemicals as starting materials; trifluoroethylimidoyl chlorides and functionalized isonitriles are widely available from global chemical suppliers, reducing the risk of supply chain disruptions associated with proprietary or custom-synthesized reagents. Furthermore, the replacement of hazardous diazonium salts with stable imidoyl chlorides significantly lowers the safety infrastructure costs required for manufacturing, as there is no need for specialized explosion-proof equipment or rigorous temperature control systems typically mandated for dangerous intermediates.
- Cost Reduction in Manufacturing: The economic viability of this process is enhanced by the use of inexpensive catalysts such as cuprous acetate and molybdenum hexacarbonyl, which are used in low loading amounts yet deliver high turnover numbers. By eliminating the need for precious metal catalysts like palladium or platinum, which are subject to volatile market pricing and require expensive recovery processes, manufacturers can achieve substantial cost savings in raw material expenditures. Additionally, the simplified workup procedure, which often involves basic filtration and standard column chromatography, reduces the consumption of solvents and silica gel, further driving down the operational expenses associated with purification and waste disposal.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more resilient supply chain by minimizing batch-to-batch variability and reducing the likelihood of failed runs due to sensitive parameters. Since the reaction tolerates a broad range of functional groups and proceeds efficiently at moderate temperatures, it is less susceptible to fluctuations in utility availability or minor deviations in process control. This reliability ensures a consistent output of high-quality intermediates, allowing downstream formulation teams to plan production schedules with greater confidence and reducing the need for safety stock buffers that tie up working capital.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this method aligns with increasingly stringent global standards for sustainable manufacturing. The avoidance of toxic heavy metals and explosive reagents simplifies the permitting process for new manufacturing lines and reduces the burden of hazardous waste management. The high atom economy of the cycloaddition reaction means that a larger proportion of the input mass is converted into the desired product, minimizing the generation of waste byproducts. This efficiency not only lowers disposal costs but also enhances the company's sustainability profile, which is becoming a critical factor in vendor selection criteria for major multinational pharmaceutical corporations.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is essential for stakeholders evaluating its adoption for commercial production. The following questions address common inquiries regarding the practical application and scope of this technology, drawing directly from the experimental data and findings presented in the patent literature. These insights are intended to clarify the operational capabilities of the method and its suitability for various project requirements.
Q: What are the key advantages of this Mo/Cu co-catalytic system over traditional methods?
A: Unlike traditional methods requiring hazardous diazonium salts or harsh cyclization conditions, this patent describes a mild protocol (70-90°C) using commercially available imidoyl chlorides and isonitriles, significantly improving safety and operational simplicity.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the patent explicitly demonstrates the method's scalability from milligram to gram levels with high efficiency. The use of inexpensive catalysts like cuprous acetate and robust reaction conditions supports potential scale-up to kilogram or tonnage production for API intermediates.
Q: What is the substrate scope for the R-group in this triazole synthesis?
A: The method exhibits broad substrate tolerance, successfully accommodating various aryl groups including those with electron-donating (methyl, methoxy) and electron-withdrawing (fluoro, chloro, nitro) substituents, as well as phenethyl groups, yielding products with up to 99% efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of next-generation therapeutics. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our facility is equipped to handle the specific requirements of fluorine chemistry, providing a safe and controlled environment for the synthesis of complex trifluoromethyl-containing compounds.
We invite you to collaborate with us to leverage this advanced synthetic technology for your upcoming projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume needs and timeline constraints. We encourage potential partners to contact us directly to obtain specific COA data for our catalog compounds or to discuss route feasibility assessments for custom targets. Let us help you optimize your supply chain and accelerate your drug development programs with our reliable and cost-effective manufacturing solutions.
