Advanced Cobalt-Catalyzed Synthesis of Tetrahydro-beta-carbolinones for Commercial API Production
Introduction: Revolutionizing Beta-Carboline Scaffold Access
The tetrahydro-beta-carbolinone skeleton represents a privileged structure in medicinal chemistry, serving as the core framework for numerous bioactive natural products and pharmaceutical candidates. As illustrated in the structural diversity of compounds like secofascaplysin A and the anxiolytic candidate SL651498, this heterocyclic motif is indispensable for developing next-generation therapeutics targeting viral infections and central nervous system disorders. 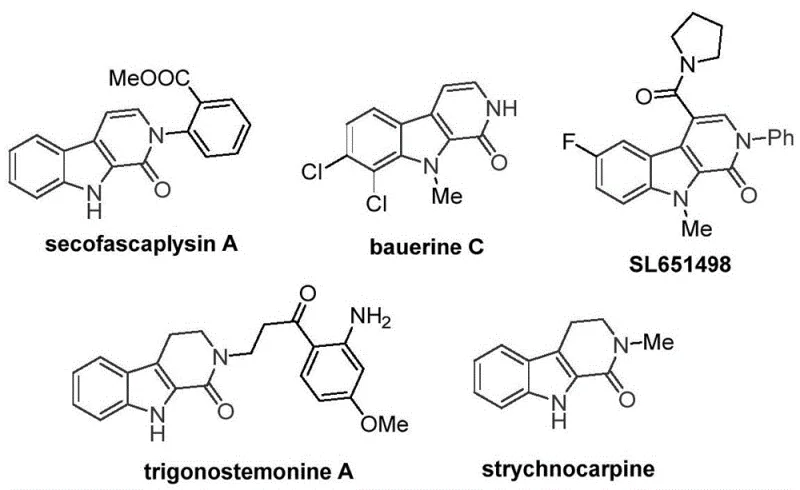 . However, traditional synthetic routes to access these valuable intermediates have often been plagued by harsh conditions, limited substrate scope, or reliance on scarce precious metals. A groundbreaking development detailed in Chinese Patent CN115260188A introduces a highly efficient preparation method utilizing transition metal cobalt catalysis. This innovation not only streamlines the synthetic pathway but also aligns perfectly with the industry's shift towards sustainable and cost-effective manufacturing processes for reliable pharmaceutical intermediate supplier networks.
. However, traditional synthetic routes to access these valuable intermediates have often been plagued by harsh conditions, limited substrate scope, or reliance on scarce precious metals. A groundbreaking development detailed in Chinese Patent CN115260188A introduces a highly efficient preparation method utilizing transition metal cobalt catalysis. This innovation not only streamlines the synthetic pathway but also aligns perfectly with the industry's shift towards sustainable and cost-effective manufacturing processes for reliable pharmaceutical intermediate supplier networks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the tetrahydro-beta-carbolinone core via carbonylation reactions has been predominantly dependent on palladium catalysis. While effective, palladium-based methodologies suffer from significant drawbacks that hinder their widespread adoption in large-scale commercial settings. The primary concern is the exorbitant cost of palladium precursors, which directly impacts the cost reduction in API manufacturing strategies. Furthermore, palladium residues are notoriously difficult to remove from final drug substances, often requiring complex and expensive scavenging steps to meet stringent regulatory purity specifications. Additionally, many conventional protocols require high-pressure carbon monoxide gas, posing severe safety risks and necessitating specialized reactor infrastructure that limits flexibility in multi-purpose production facilities.
The Novel Approach
The methodology disclosed in CN115260188A offers a transformative solution by replacing precious palladium with earth-abundant cobalt. This novel approach employs a cobalt-catalyzed C-H activation carbonylation reaction using tryptamine derivatives as starting materials. By utilizing a solid carbon monoxide substitute, specifically 1,3,5-tricarboxylic acid phenol ester, the process eliminates the hazards associated with gaseous CO. The reaction proceeds smoothly in dioxane at temperatures between 120-140°C, demonstrating exceptional compatibility with various functional groups including halogens, alkyls, and alkoxy substituents. This robustness ensures that complex molecular architectures can be assembled without extensive protection-deprotection sequences, thereby significantly shortening the overall synthetic timeline and enhancing the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Cobalt-Catalyzed C-H Activation Carbonylation
The success of this transformation lies in the elegant catalytic cycle driven by the cobalt species. The reaction initiates with the oxidation of the cobalt(II) catalyst, typically cobalt acetate tetrahydrate, by silver carbonate to generate an active cobalt(III) species. This high-valent metal center then coordinates with the tryptamine derivative, facilitating the critical C-H bond activation at the 2-position of the indole ring. This step is pivotal as it forms a stable cobalt(III) metallacycle, setting the stage for carbon-carbon bond formation. Subsequently, carbon monoxide, released in situ from the phenol ester surrogate, inserts into the cobalt-carbon bond to form an acyl-cobalt(III) intermediate.  . The cycle concludes with a reductive elimination step followed by hydrolysis, which releases the desired tetrahydro-beta-carbolinone product and regenerates the catalyst. This mechanism highlights the efficiency of base metal catalysis in achieving transformations previously reserved for noble metals.
. The cycle concludes with a reductive elimination step followed by hydrolysis, which releases the desired tetrahydro-beta-carbolinone product and regenerates the catalyst. This mechanism highlights the efficiency of base metal catalysis in achieving transformations previously reserved for noble metals.
From an impurity control perspective, this mechanism offers distinct advantages. The use of silver carbonate as a stoichiometric oxidant ensures clean conversion without generating toxic byproducts that could complicate the impurity profile. The specificity of the C-H activation at the 2-position minimizes the formation of regioisomers, a common issue in electrophilic substitution reactions on indole rings. Moreover, the mild reaction conditions prevent the degradation of sensitive functional groups, ensuring that the final high-purity pharmaceutical intermediate maintains its structural integrity. The ability to tolerate diverse substituents (R1 and R2) allows for the rapid generation of analog libraries, accelerating the drug discovery process while maintaining a clean and manageable impurity spectrum.
How to Synthesize Tetrahydro-beta-carbolinone Efficiently
The practical implementation of this cobalt-catalyzed protocol is designed for ease of execution in both laboratory and pilot plant environments. The procedure involves a straightforward one-pot reaction where all reagents are combined in a Schlenk tube or standard reactor. The simplicity of the workup, involving filtration and standard column chromatography, makes it highly attractive for process chemists looking to optimize throughput. For detailed operational parameters and specific stoichiometric ratios validated across multiple substrates, please refer to the standardized synthesis guide below.
- Combine cobalt catalyst (Co(OAc)2·4H2O), base (Et3N), CO substitute (TFBen), additive (PivOH), tryptamine derivative, and oxidant (Ag2CO3) in dioxane solvent.
- Heat the reaction mixture to 120-140°C and maintain stirring for 16-24 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the final tetrahydro-beta-carbolinone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from palladium to cobalt catalysis represents a strategic opportunity to optimize raw material costs and secure supply continuity. The reliance on base metals drastically reduces the exposure to volatile precious metal markets, stabilizing the cost structure of the final intermediate. Furthermore, the use of commercially available, off-the-shelf reagents such as triethylamine and pivalic acid ensures that the supply chain remains resilient against disruptions. The elimination of high-pressure gas equipment requirements also lowers the barrier for contract manufacturing organizations (CMOs) to adopt this technology, expanding the pool of potential manufacturing partners and reducing lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The substitution of expensive palladium catalysts with inexpensive cobalt salts results in substantial cost savings on a per-kilogram basis. Since cobalt is orders of magnitude cheaper than palladium, the direct material cost is significantly lowered. Additionally, the simplified purification process required to remove base metal residues compared to noble metals reduces downstream processing costs, contributing to overall economic efficiency without compromising quality standards.
- Enhanced Supply Chain Reliability: The starting materials, including tryptamine derivatives and the CO surrogate, are readily synthesized from bulk chemicals or sourced from established suppliers. This accessibility mitigates the risk of single-source dependency often associated with specialized ligands or rare metal catalysts. By utilizing a robust and widely applicable synthetic route, manufacturers can ensure consistent delivery schedules and maintain inventory levels sufficient to meet fluctuating market demands for critical API precursors.
- Scalability and Environmental Compliance: The reaction conditions are amenable to scale-up, having been demonstrated effectively on gram scales with high yields. The use of a solid CO surrogate enhances workplace safety by removing the need for pressurized gas cylinders, aligning with strict environmental, health, and safety (EHS) regulations. This safety profile facilitates smoother regulatory approvals for manufacturing sites and reduces the administrative burden associated with handling hazardous gases, making it an ideal candidate for green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this cobalt-catalyzed synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for technical teams evaluating this route for potential adoption.
Q: What are the advantages of using cobalt over palladium for this synthesis?
A: Cobalt catalysts are significantly more earth-abundant and cost-effective than precious palladium metals. Furthermore, the cobalt-catalyzed method described in CN115260188A operates under relatively mild conditions with high functional group tolerance, reducing the need for expensive protecting groups and simplifying downstream purification.
Q: What is the role of 1,3,5-tricarboxylic acid phenol ester in this reaction?
A: This compound acts as a solid carbon monoxide (CO) surrogate. It safely releases CO in situ under the reaction conditions, eliminating the need for handling hazardous high-pressure CO gas cylinders, which greatly enhances operational safety and scalability in a manufacturing setting.
Q: Can this method be scaled for industrial production?
A: Yes, the patent explicitly states that the method is simple to operate and has been successfully demonstrated on a gram scale. The use of commercially available reagents and standard organic solvents like dioxane supports straightforward scale-up to multi-kilogram or ton-level production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydro-beta-carbolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the modern pharmaceutical landscape. Our team of expert process chemists has extensively evaluated the cobalt-catalyzed carbonylation technology described in CN115260188A and is fully equipped to translate this academic innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from benchtop discovery to full-scale manufacturing. Our state-of-the-art rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of tetrahydro-beta-carbolinone intermediate meets the highest global regulatory standards.
We invite you to collaborate with us to leverage this advanced technology for your drug development programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain is optimized for speed, cost, and reliability in the competitive global market.
