Mastering Parecoxib Sodium Genotoxic Impurity Synthesis for Global Pharmaceutical Compliance
The pharmaceutical industry faces escalating regulatory pressure regarding the control of genotoxic impurities in active pharmaceutical ingredients (APIs), particularly for potent drugs like Parecoxib Sodium. As a water-soluble prodrug of Valdecoxib used for postoperative pain management, Parecoxib Sodium requires rigorous quality control to ensure patient safety. The patent CN110938043A introduces a groundbreaking preparation method for specific genotoxic impurities associated with this molecule, addressing a critical gap in analytical standard availability. These impurities, specifically aryl sulfonate esters, are formed as byproducts during the sulfonation and esterification stages of the main synthesis route. Without high-purity reference standards, quality control laboratories struggle to accurately quantify these trace contaminants against strict toxicological thresholds. This technical insight explores the sophisticated chemical engineering behind this new synthesis route, offering a robust solution for producing reference materials with exceptional purity and yield.
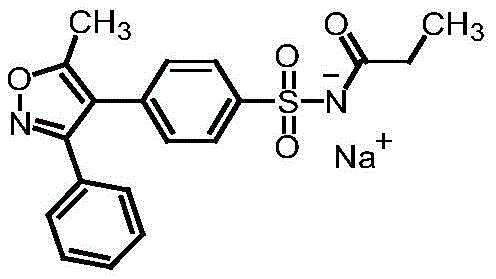
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl sulfonate impurities for reference purposes has relied on methods that introduce significant purification challenges and environmental hazards. A notable prior art example, disclosed in patent 201510843521.8, utilizes a chlorosulfonation step followed by esterification in the presence of pyridine as an acid-binding agent. While functional, this approach suffers from inherent drawbacks that compromise both product quality and process efficiency. The use of pyridine introduces a new organic impurity profile that is notoriously difficult to separate from the target sulfonate esters, often requiring complex chromatographic purification. Furthermore, the chlorosulfonation step in conventional methods often lacks precise kinetic control, leading to the formation of ortho- and meta-isomers that contaminate the final product. These isomers not only reduce the overall yield but also complicate the analytical validation of the reference standard. Additionally, the esterification conditions in older methods can promote hydrolysis of the sensitive sulfonate ester bond, further degrading the quality of the material intended for high-precision analysis.
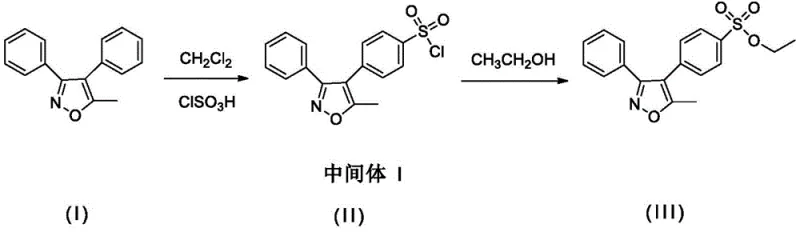
The Novel Approach
In stark contrast, the methodology outlined in CN110938043A represents a paradigm shift in how these critical reference standards are manufactured. The core innovation lies in the elimination of the acid-binding agent during the esterification phase, replacing it with a direct reaction using sodium alkoxide solutions. This strategic modification removes the risk of introducing nitrogen-containing organic impurities, thereby streamlining the purification workflow. The process begins with a highly controlled chlorosulfonation of 5-methyl-3,4-diphenyl isoxazole, where the addition rate of chlorosulfonic acid is strictly maintained below 3 mL/min at low temperatures. This kinetic control is pivotal in suppressing the formation of unwanted regioisomers, ensuring that the para-chlorosulfonated intermediate is generated with high selectivity. By coupling this precise upstream control with a mild, room-temperature esterification using sodium methoxide, ethoxide, or isopropoxide, the process achieves a level of chemical integrity previously difficult to attain. The result is a synthesis pathway that is not only cleaner but also more robust against the thermal decomposition that plagues traditional high-temperature esterification methods.
Mechanistic Insights into Controlled Chlorosulfonation and Alkoxide Esterification
The success of this synthesis hinges on a deep understanding of electrophilic aromatic substitution and nucleophilic displacement mechanisms under constrained conditions. During the initial chlorosulfonation, the isoxazole ring system presents multiple sites for potential substitution, but the process parameters are tuned to favor the para-position exclusively. By maintaining the reaction temperature between -5°C and 0°C during the addition of chlorosulfonic acid, the energy available for forming higher-energy transition states leading to ortho-substitution is minimized. The subsequent heating to 35 ± 5°C allows for the completion of the reaction without triggering excessive degradation or rearrangement. Crucially, the process employs High-Performance Liquid Chromatography (HPLC) for real-time monitoring, ensuring that the starting material and meta-isomer levels remain below 1.0% before proceeding. This analytical feedback loop is essential for maintaining the structural fidelity of the intermediate, which directly correlates to the purity of the final genotoxic impurity standard.
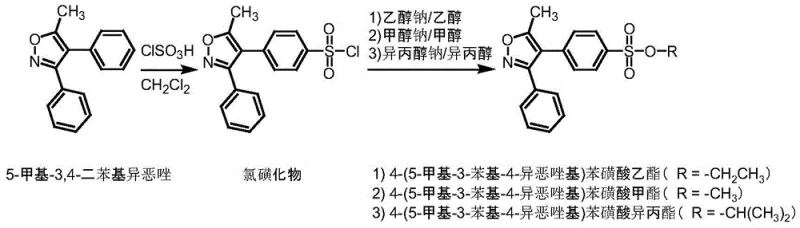
Following the formation of the sulfonyl chloride intermediate, the esterification mechanism proceeds via a nucleophilic attack by the alkoxide ion on the sulfur atom. In traditional methods, the generated HCl must be scavenged by a base like pyridine, which creates a salt byproduct that can trap solvent or react further. In this novel approach, the use of pre-formed sodium alkoxide solutions means that the stoichiometry is inherently balanced, and the byproduct is simply sodium chloride, which is easily removed by filtration or washing. This absence of organic bases prevents the formation of quaternary ammonium salts or other complex adducts that could co-elute with the target analyte during HPLC analysis. Furthermore, the reaction is conducted at room temperature, which preserves the integrity of the sulfonate ester linkage, preventing the hydrolysis back to the sulfonic acid that occurs under acidic or high-thermal stress conditions. The final crystallization at 0 ± 5°C ensures that the product precipitates in a highly ordered lattice, excluding remaining soluble impurities and yielding a solid with purity exceeding 99.5%.
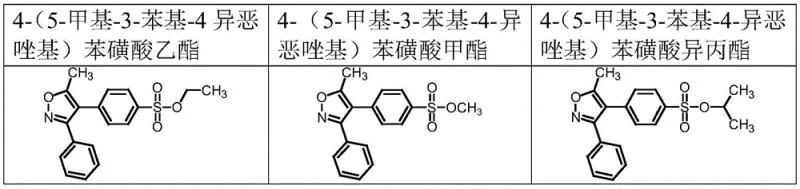
How to Synthesize Parecoxib Sodium Impurities Efficiently
Implementing this synthesis route requires adherence to strict operational protocols to maximize yield and safety. The process is designed to be scalable while maintaining the precision necessary for reference standard production. Operators must ensure that the addition rate of chlorosulfonic acid is meticulously controlled using metering pumps to prevent exothermic runaways that could compromise regioselectivity. The quenching step into ice water must also be managed carefully to control the evolution of HCl gas and prevent local overheating. For the esterification phase, the preparation of the sodium alkoxide solution should be done under anhydrous conditions to prevent premature hydrolysis of the alkoxide reagent. Detailed standardized synthesis steps see the guide below.
- Perform controlled chlorosulfonation of 5-methyl-3,4-diphenyl isoxazole at -5 to 0°C with strict addition rate monitoring to minimize isomers.
- Quench the reaction with ice water and extract the para-chlorosulfonated intermediate into dichloromethane for purification.
- React the intermediate with sodium alkoxide solutions (methoxide, ethoxide, or isopropoxide) at room temperature to form the target sulfonate esters.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this optimized synthesis route offers substantial benefits for procurement managers and supply chain directors looking to secure reliable sources of high-purity pharmaceutical intermediates. The elimination of pyridine and the simplification of the workup procedure translate directly into reduced processing time and lower solvent consumption. This efficiency gain allows for a more streamlined manufacturing schedule, enhancing the overall reliability of supply for critical quality control materials. Furthermore, the high selectivity of the reaction minimizes the loss of valuable starting materials to byproduct formation, optimizing the raw material utilization rate. For organizations managing tight budgets, the reduction in purification complexity means lower operational expenditures associated with chromatography resins and extensive solvent recycling.
- Cost Reduction in Manufacturing: The removal of the acid-binding agent pyridine eliminates a significant cost center associated with both the purchase of the reagent and the subsequent disposal or recovery of pyridine-contaminated waste streams. Additionally, the ability to perform esterification at room temperature reduces energy consumption compared to processes requiring reflux or heating. The high yield achieved (>67%) ensures that the cost per gram of the final high-purity impurity standard is significantly optimized, providing better value for quality control laboratories that require frequent replenishment of reference materials.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route, characterized by its tolerance to minor variations and its reliance on common, commercially available reagents like sodium alkoxides, ensures consistent production capabilities. Unlike methods that rely on specialized catalysts or hard-to-source reagents, this process utilizes bulk chemicals that are readily available in the global market. This accessibility reduces the risk of supply disruptions caused by raw material shortages. Moreover, the simplified purification process reduces the batch cycle time, allowing manufacturers to respond more quickly to urgent requests for certificates of analysis and reference samples from regulatory bodies.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the absence of hazardous amine bases and the controlled nature of the exothermic sulfonation step. The waste profile is significantly cleaner, consisting primarily of aqueous salt solutions and recoverable organic solvents, which simplifies wastewater treatment and aligns with modern green chemistry principles. This environmental compatibility is increasingly important for pharmaceutical suppliers who must adhere to strict corporate sustainability goals and regulatory environmental standards. The process design inherently supports large-scale manufacturing without the need for specialized equipment beyond standard glass-lined or stainless steel reactors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these genotoxic impurity standards. Understanding these details helps stakeholders make informed decisions about their quality control strategies and supplier partnerships. The answers are derived directly from the technical specifications and beneficial effects reported in the underlying patent data.
Q: Why is the control of genotoxic impurities critical in Parecoxib Sodium manufacturing?
A: Parecoxib Sodium synthesis often involves sulfonation steps that can generate aryl sulfonate esters, which are recognized warning structures for genotoxicity. Regulatory guidelines like ICH M7 require these impurities to be controlled below strict thresholds (e.g., 18 ppm), necessitating high-purity reference standards for accurate analytical detection.
Q: How does the novel synthesis method improve upon traditional pyridine-based esterification?
A: Traditional methods often utilize pyridine as an acid-binding agent, which introduces new organic impurities that are difficult to remove and increases environmental hazards. The novel method employs sodium alkoxide solutions directly, eliminating the need for pyridine, reducing side reactions, and significantly simplifying the downstream purification process.
Q: What purity levels can be achieved with this optimized chlorosulfonation process?
A: By strictly controlling the addition rate of chlorosulfonic acid and monitoring the reaction via HPLC to limit meta-isomers, this process consistently achieves chemical purities exceeding 99.5%, with some batches reaching 99.73%, making it ideal for producing certified reference materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Parecoxib Sodium Impurities Supplier
At NINGBO INNO PHARMCHEM, we understand that the integrity of your analytical data depends on the quality of your reference standards. Our team of expert chemists has extensively evaluated the synthesis route described in CN110938043A and is fully equipped to replicate and scale this technology for commercial needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you need grams for method validation or kilograms for routine QC, we can deliver. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of Parecoxib Sodium genotoxic impurities meets the >99.5% purity benchmark required by international pharmacopoeias.
We invite you to collaborate with us to optimize your supply chain for these critical materials. By leveraging our technical expertise, you can secure a stable source of high-quality impurities that support your regulatory filings and product releases. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your annual volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities align with your quality objectives.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
