Scalable Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazines for Advanced Pharmaceutical Intermediates
Scalable Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazines for Advanced Pharmaceutical Intermediates
The rapid evolution of medicinal chemistry has placed a premium on heterocyclic scaffolds that offer enhanced metabolic stability and bioavailability, with the trifluoromethyl group being a cornerstone modification in modern drug design. Patent CN116253692A introduces a groundbreaking preparation method for trifluoromethyl substituted 1,2,4-triazine compounds, addressing critical bottlenecks in the synthesis of these valuable pharmacophores. This novel approach leverages a synergistic [3+3] cycloaddition strategy between chlorohydrazones and trifluoroacetyl sulfur ylides, facilitated by inexpensive potassium carbonate. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this technology represents a significant leap forward, offering a pathway to high-purity intermediates without the burden of heavy metal contamination or complex reaction engineering.
The strategic incorporation of the trifluoromethyl moiety into the 1,2,4-triazine core is not merely a structural exercise but a functional imperative for optimizing lipophilicity and electronegativity in potential therapeutic agents. Traditional synthetic routes often struggle to introduce this group efficiently while maintaining the integrity of the heterocyclic ring. The disclosed method overcomes these hurdles by utilizing readily accessible starting materials that react under exceptionally mild conditions. By operating at temperatures between 20°C and 40°C in an ambient air atmosphere, the process drastically reduces energy consumption and infrastructure requirements compared to cryogenic or high-pressure alternatives. This accessibility makes it an attractive candidate for cost reduction in API manufacturing, particularly for companies aiming to streamline their supply chains for oncology and antifungal drug candidates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 1,2,4-triazine skeletons has relied heavily on the condensation of amidrazones with 1,2-diketones or alkynes, as well as multicomponent reactions involving hydrazides and dicarbonyl compounds. While these classical methods established the foundational chemistry of the ring system, they are plagued by significant inefficiencies when applied to complex, fluorinated targets. Conventional protocols frequently necessitate harsh reaction conditions, including elevated temperatures and strict anhydrous environments, which can degrade sensitive functional groups and lead to poor yields. Furthermore, the structural diversity achievable through these older routes is often limited, restricting the ability of medicinal chemists to explore broad chemical space around the triazine core. The reliance on specific, hard-to-synthesize precursors also inflates the cost of goods, creating a barrier for the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology described in the patent utilizes a robust [3+3] cycloaddition between chlorohydrazones and trifluoroacetyl sulfur ylides to construct the triazine ring with high precision. This innovative route bypasses the need for pre-functionalized diketones, instead generating the necessary reactive intermediates in situ under the promotion of potassium carbonate. The reaction proceeds smoothly in common organic solvents like tetrahydrofuran (THF), eliminating the need for exotic or hazardous reagents. Crucially, the process is metal-free, avoiding the use of transition metal catalysts that often require rigorous and costly removal steps to meet regulatory purity standards for pharmaceutical ingredients. This simplicity not only accelerates the development timeline but also enhances the overall safety profile of the manufacturing process, aligning perfectly with modern green chemistry principles.
Mechanistic Insights into Metal-Free [3+3] Cycloaddition
The mechanistic elegance of this transformation lies in the generation of a nitrile imine intermediate from the chlorohydrazone precursor. Under the basic conditions provided by potassium carbonate, the chlorohydrazone undergoes dehydrohalogenation to release hydrogen chloride, forming the highly reactive nitrile imine species. This intermediate then engages in a concerted cycloaddition with the trifluoroacetyl sulfur ylide. The sulfur ylide acts as a three-atom synthon, donating its electron density to close the six-membered ring. Following the cyclization event, the elimination of dimethyl sulfoxide (DMSO) drives the reaction forward, aromatizing the system to yield the stable 1,2,4-triazine product. This cascade of events occurs with remarkable atom economy, minimizing waste generation and maximizing the conversion of starting materials into the desired high-purity pharmaceutical intermediate.
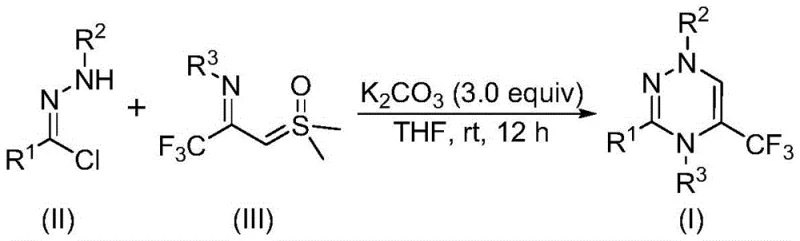
Beyond the primary cycloaddition pathway, the reaction conditions allow for significant structural modulation through the variation of substituents on the starting materials. The R1, R2, and R3 groups can be independently tuned to include alkyl chains, aryl rings, or heterocycles, with substituents such as halogens, alkoxy groups, and trifluoromethyl moieties being well-tolerated. This flexibility is critical for structure-activity relationship (SAR) studies, allowing researchers to rapidly generate libraries of analogues. The mechanism ensures that even sterically hindered substrates, such as those with ortho-substituted phenyl rings or bulky naphthyl groups, can participate effectively in the reaction. This broad substrate scope underscores the versatility of the method, making it a powerful tool for the commercial scale-up of complex polymer additives or specialized agrochemical intermediates where specific substitution patterns are required for biological activity.
How to Synthesize Trifluoromethyl 1,2,4-Triazines Efficiently
The operational simplicity of this synthesis protocol makes it highly accessible for laboratory execution and subsequent industrial translation. The procedure involves mixing the chlorohydrazone, trifluoroacetyl sulfur ylide, and potassium carbonate in a solvent like THF, followed by stirring at room temperature. The absence of stringent moisture or oxygen exclusion measures simplifies the setup, requiring only standard glassware and ambient conditions. Post-reaction processing is equally straightforward, typically involving filtration to remove inorganic salts followed by standard purification techniques. For detailed operational parameters and specific stoichiometric ratios optimized for different substrates, please refer to the standardized synthesis steps outlined below.
- Prepare the reaction mixture by adding potassium carbonate, chlorohydrazone, and trifluoroacetyl sulfur ylide into an organic solvent such as tetrahydrofuran (THF).
- Stir the reaction mixture at room temperature (20-40°C) under an air atmosphere for 10 to 14 hours to allow the [3+3] cycloaddition to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target trifluoromethyl substituted 1,2,4-triazine compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible benefits that extend beyond mere chemical yield. The elimination of heavy metal catalysts removes a major cost center associated with scavenging resins and extensive purification workflows, directly contributing to cost reduction in API manufacturing. Furthermore, the use of commodity chemicals like potassium carbonate and common solvents ensures a stable and resilient supply chain, mitigating the risks associated with sourcing specialized reagents. The robustness of the reaction under air atmosphere means that production can proceed without the capital expenditure required for extensive inert gas systems, lowering the barrier to entry for contract manufacturing organizations.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the replacement of expensive transition metal catalysts with inexpensive inorganic bases. By removing the necessity for precious metals, manufacturers avoid the volatility of metal markets and the high costs of metal removal validation. Additionally, the mild reaction conditions reduce energy consumption for heating or cooling, leading to substantial cost savings in utility expenses. The high atom efficiency of the cycloaddition minimizes raw material waste, further optimizing the cost per kilogram of the final product.
- Enhanced Supply Chain Reliability: The starting materials, including chlorohydrazones and sulfur ylides, are derived from widely available bulk chemicals such as acyl chlorides, hydrazines, and sulfoxides. This reliance on commodity feedstocks ensures that production schedules are not disrupted by the scarcity of niche reagents. The ability to source these precursors from multiple global suppliers enhances supply security, allowing for flexible procurement strategies that can adapt to market fluctuations. This reliability is crucial for maintaining continuous production lines for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: The protocol's compatibility with ambient air and room temperature conditions makes it inherently safer and easier to scale than exothermic or pressure-dependent reactions. The absence of toxic heavy metals simplifies waste treatment and disposal, ensuring compliance with increasingly stringent environmental regulations. This green chemistry profile not only reduces the environmental footprint but also streamlines the regulatory approval process for new drug applications, accelerating time-to-market for downstream products.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is essential for stakeholders evaluating its integration into existing production pipelines. The following questions address common inquiries regarding substrate compatibility, reaction conditions, and scalability. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a clear picture of the method's capabilities and limitations for potential adopters.
Q: What are the key advantages of this synthesis method over traditional routes?
A: This method eliminates the need for expensive and toxic heavy metal catalysts, operates under mild conditions (room temperature and air atmosphere), and utilizes cheap, readily available inorganic bases like potassium carbonate, significantly simplifying post-treatment and reducing environmental impact.
Q: What is the substrate scope for the R1, R2, and R3 groups in the triazine structure?
A: The process demonstrates excellent functional group tolerance. R1 can be alkyl, phenyl, naphthyl, or furyl; R2 and R3 can be substituted or unsubstituted phenyl or naphthyl groups. Substituents such as methyl, methoxy, halogens, nitro, and trifluoromethyl groups are well-tolerated at ortho, meta, or para positions.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the protocol is highly scalable. It avoids sensitive reagents requiring inert gas protection, uses common solvents like THF, and involves simple filtration and chromatography workups, making it ideal for scaling from gram-level laboratory synthesis to multi-kilogram commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl 1,2,4-Triazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis technology for the next generation of therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of trifluoromethyl 1,2,4-triazine intermediate meets the highest industry standards. We are committed to delivering high-purity pharmaceutical intermediates that empower your R&D teams to push the boundaries of drug discovery.
We invite you to collaborate with us to leverage this advanced synthetic route for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain and accelerate the development of your life-saving medicines with our reliable trifluoromethyl 1,2,4-triazine supply solutions.
