Scalable Synthesis of 4-Position Alkylated 2-Methylquinoline Derivatives for Commercial API Production
Scalable Synthesis of 4-Position Alkylated 2-Methylquinoline Derivatives for Commercial API Production
The strategic development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern pharmaceutical process research. Specifically, the functionalization of the quinoline scaffold at the 4-position presents unique challenges regarding regioselectivity and reaction efficiency. Patent CN114380741B introduces a transformative methodology for the preparation of 4-position alkylated derivatives of 2-methylquinoline compounds, addressing critical bottlenecks in existing synthetic technologies. This innovation leverages a silver-catalyzed radical decarboxylative alkylation strategy, utilizing Selectfluor as a potent oxidant within an aqueous medium. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, this technology represents a significant leap forward in process robustness. The ability to achieve high conversion rates under mild conditions while maintaining exceptional purity profiles makes this approach particularly attractive for the production of high-value active pharmaceutical ingredients (APIs) and advanced agrochemical intermediates.
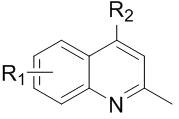
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alkylated quinoline derivatives has relied heavily on classical Minisci-type reactions, which, while effective, suffer from substantial operational drawbacks in a commercial setting. Traditional protocols often necessitate high-temperature conditions that can degrade sensitive functional groups and lead to the formation of complex impurity profiles requiring extensive purification. Furthermore, established methods frequently depend on stoichiometric amounts of hazardous oxidants or expensive photocatalysts that complicate the supply chain and increase the overall cost of goods. For instance, prior art involving dual-catalytic systems often requires precise light irradiation and thiol catalysts, creating scalability issues for large-batch manufacturing. The residue of heavy metals or sulfur-containing by-products poses significant regulatory hurdles for API manufacturers, necessitating additional downstream processing steps that erode profit margins and extend lead times.
The Novel Approach
In stark contrast, the methodology disclosed in the patent data utilizes a streamlined silver-catalyzed system that operates efficiently in water, a green solvent that drastically reduces environmental impact and disposal costs. By employing silver nitrate as a catalyst and Selectfluor as the terminal oxidant, the reaction achieves remarkable regioselectivity for the 4-position of the 2-methylquinoline ring without the need for external light sources or exotic ligands. This novel approach not only simplifies the reaction setup but also enhances the safety profile of the manufacturing process by avoiding volatile organic solvents. The general reaction scheme illustrates the direct coupling of 2-methylquinolines with various carboxylic acids, demonstrating the versatility of this chemistry for generating diverse molecular libraries.
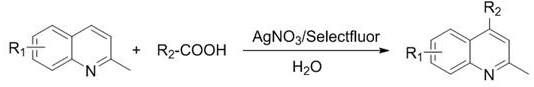
Mechanistic Insights into AgNO3-Catalyzed Radical Decarboxylation
The core of this technological advancement lies in the mechanistic pathway facilitated by the silver catalyst, which promotes the generation of alkyl radicals from carboxylic acids under mild oxidative conditions. The silver ion acts as a single-electron transfer mediator, activating the carboxylic acid substrate to undergo decarboxylation, thereby releasing carbon dioxide and generating a reactive alkyl radical species. This radical subsequently attacks the electron-deficient quinoline ring at the C4 position, driven by the electronic properties of the heterocycle and the specific coordination environment provided by the reaction medium. The use of Selectfluor ensures a continuous regeneration of the active silver species, maintaining the catalytic cycle without the accumulation of inactive metal precipitates. This mechanism is critical for R&D teams as it explains the high tolerance for various functional groups, allowing for the synthesis of complex molecules without protecting group strategies.
From an impurity control perspective, the aqueous nature of the reaction medium plays a pivotal role in suppressing side reactions that typically plague organic-phase radical chemistries. The high polarity of water helps to stabilize charged intermediates and facilitates the rapid quenching of non-productive radical pathways that could lead to dimerization or over-alkylation. Consequently, the crude reaction mixtures obtained from this process exhibit significantly cleaner HPLC profiles compared to those from traditional methods, reducing the burden on purification units. This inherent selectivity translates directly into higher isolated yields, with experimental data showing consistent results ranging from 80% to 96% across a broad spectrum of substrates. Such predictability is essential for ensuring batch-to-batch consistency in the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 4-Position Alkylated 2-Methylquinoline Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and minimize impurity formation, yet the operational procedure remains remarkably straightforward for industrial adoption. The process begins with the careful mixing of the 2-methylquinoline substrate and the chosen carboxylic acid in an aqueous solution containing the silver nitrate catalyst and Selectfluor oxidant. Maintaining the reaction temperature at the optimal range of 80°C for a duration of 8 hours ensures complete conversion while preventing thermal degradation of the product. Following the reaction, a standard workup procedure involving extraction with ethyl acetate and neutralization with sodium bicarbonate effectively isolates the organic product. The detailed standardized synthesis steps below outline the specific molar ratios and conditions required to replicate the high success rates reported in the patent documentation.
- Mix 2-methylquinoline compound with carboxylic acid, silver nitrate catalyst, and Selectfluor oxidant in an aqueous solution.
- Heat the reaction mixture to 50-100°C (preferably 80°C) and stir for approximately 8 hours to ensure complete conversion.
- Perform workup by extraction with ethyl acetate, dry over sodium sulfate, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this aqueous silver-catalyzed protocol offers tangible benefits that extend beyond mere chemical efficiency, directly impacting the bottom line through cost reduction in API manufacturing. The elimination of expensive photocatalysts and the replacement of volatile organic solvents with water significantly lowers the raw material expenditure and waste treatment costs associated with production. Moreover, the high atom economy of the decarboxylative alkylation ensures that a greater proportion of input materials are converted into the final product, reducing the overall material intensity of the process. This efficiency is compounded by the robustness of the reaction, which tolerates a wide range of substrate qualities, thereby reducing the risk of batch failures due to minor variations in starting material specifications.
- Cost Reduction in Manufacturing: The substitution of traditional organic solvents with water represents a massive decrease in solvent purchase and recovery costs, while the use of commercially available silver nitrate avoids the premium pricing of specialized ligands. By achieving high yields consistently, the process minimizes the loss of valuable intermediates, leading to substantial cost savings per kilogram of produced API. Additionally, the simplified workup procedure reduces labor hours and energy consumption associated with distillation and solvent exchange, further enhancing the economic viability of the route.
- Enhanced Supply Chain Reliability: The reagents required for this synthesis, including Selectfluor and silver salts, are commodity chemicals with stable global supply chains, mitigating the risk of shortages that often plague proprietary catalysts. The robustness of the reaction conditions means that manufacturing can proceed without the need for highly specialized equipment such as photoreactors, allowing for production in standard stainless steel reactors found in most multipurpose facilities. This flexibility ensures reducing lead time for high-purity pharmaceutical intermediates by enabling faster campaign turnover and easier scheduling.
- Scalability and Environmental Compliance: Operating in an aqueous medium inherently aligns with green chemistry principles, facilitating easier compliance with increasingly stringent environmental regulations regarding VOC emissions. The process generates minimal hazardous waste, primarily consisting of aqueous salt solutions that are easier to treat than organic solvent mixtures. This environmental compatibility simplifies the permitting process for new manufacturing lines and supports corporate sustainability goals, making it an attractive option for long-term supply partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method, providing clarity for stakeholders evaluating its adoption. These insights are derived directly from the experimental data and comparative examples provided in the patent specification, ensuring accuracy and relevance for decision-makers. Understanding these nuances is crucial for assessing the feasibility of integrating this technology into existing production workflows and for optimizing the supply chain for quinoline-based therapeutics.
Q: What are the advantages of this silver-catalyzed method over traditional Minisci reactions?
A: Unlike traditional Minisci reactions that often require harsh conditions and produce significant by-products, this method utilizes a silver nitrate catalyst and Selectfluor in water. It offers superior selectivity, higher yields (up to 96%), and eliminates the need for complex photocatalysts or thiol additives, significantly simplifying the purification process.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is highly scalable. It uses water as the primary solvent, which is cost-effective and environmentally benign. The reaction operates at moderate temperatures (80°C) and uses commercially available reagents like silver nitrate and Selectfluor, making it ideal for the commercial scale-up of complex pharmaceutical intermediates without requiring specialized high-pressure equipment.
Q: What is the substrate scope for this alkylation reaction?
A: The method demonstrates broad applicability. It tolerates various substituents on the quinoline ring, including halogens (F, Cl), nitro groups, and trifluoromethyl groups. Furthermore, it accepts a wide range of carboxylic acids, from simple acetic acid to bulky cyclohexanecarboxylic acid, allowing for the synthesis of diverse 4-alkylated derivatives essential for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Position Alkylated 2-Methylquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity quinoline derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to implement the silver-catalyzed aqueous process described in CN114380741B allows us to offer clients a superior balance of quality, cost, and speed.
We invite potential partners to engage with our technical procurement team to discuss how this innovative route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your target molecule. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
