Optimized Industrial Synthesis of Baloxavir Marboxil for Global Pharmaceutical Supply Chains
Optimized Industrial Synthesis of Baloxavir Marboxil for Global Pharmaceutical Supply Chains
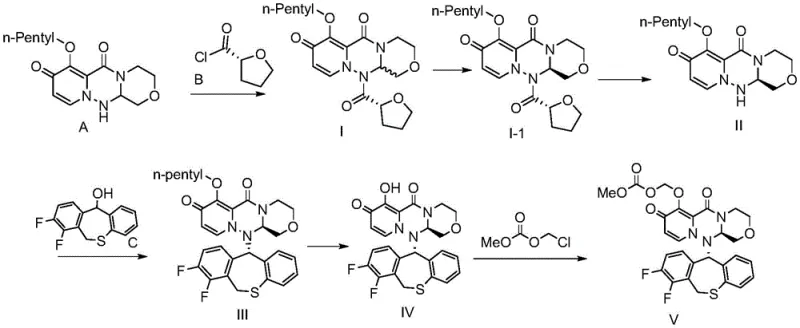
The global demand for effective antiviral therapeutics has necessitated the development of robust, scalable, and cost-efficient manufacturing routes for key active pharmaceutical ingredients. Patent CN114539285A introduces a groundbreaking preparation method for Baloxavir Marboxil, a potent cap-dependent endonuclease inhibitor used in the treatment of influenza A and B. This technical innovation addresses critical bottlenecks in the conventional synthesis pathways by employing a stable n-pentyl protection strategy for the phenolic hydroxyl group. By stabilizing the intermediate structure, the process achieves high-yield condensation with (R)-(+)-tetrahydrofuran-2-formyl chloride while drastically minimizing the formation of difficult-to-remove byproducts associated with protecting group deprotection. This advancement represents a significant leap forward for reliable Baloxavir Marboxil supplier capabilities, offering a pathway that is not only chemically superior but also commercially viable for large-scale production.
Furthermore, the methodology described in the patent eliminates the reliance on controlled reagents such as 1-propylphosphoric cyclic anhydride during the initial condensation phases, which simplifies the regulatory landscape for manufacturing facilities. The subsequent docking with the C fragment is optimized through precise adjustments in solvent polarity, facilitating easy product precipitation and enhancing conversion rates. This solves the longstanding issue of low conversion rates and difficult purification reported in prior literature, ensuring that the final product meets stringent purity specifications required by global health authorities. The integration of these improvements results in a reaction yield increase of approximately 10% compared to previous methods, establishing a new benchmark for cost reduction in antiviral API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Baloxavir Marboxil has been plagued by significant chemical inefficiencies that hinder industrial scalability and cost-effectiveness. Conventional routes, such as those disclosed in earlier patents like WO2016175224, often rely on benzyl protecting groups which are prone to premature deprotection under the rigorous conditions required for coupling the dibenzothiophene and oxazine pyridine triazine ring structures. This instability leads to the generation of epimers and side products that possess physical properties remarkably similar to the target compound, making their removal extremely difficult and resource-intensive. The refining procedures required to achieve acceptable purity levels in these traditional methods greatly increase production time and waste, resulting in reaction yields that often stagnate around 53% for critical coupling steps. Additionally, the use of expensive and regulated reagents like T3P in early stages adds unnecessary complexity to the supply chain, creating vulnerabilities in raw material availability and increasing the overall cost of goods sold.
The Novel Approach
The novel approach detailed in the recent patent data fundamentally reengineers the synthesis pathway to overcome these inherent chemical limitations through strategic molecular design. By replacing the traditional benzyl protecting group with an n-pentyl group, the new method creates a stable intermediate that withstands the reaction conditions without undergoing unwanted side reactions or epimerization. This stability allows for a high-yield condensation process that significantly reduces the burden on downstream purification units, thereby streamlining the entire manufacturing workflow. The process further innovates by adjusting the polarity of the solvent system during the docking phase, which induces the product to precipitate easily, enhancing both conversion rates and separation efficiency. This methodological shift not only improves the reaction yield by about 10% but also simplifies the operational protocol, making it a feasible process for industrial scale-up and implementation without compromising on the quality of the high-purity Baloxavir Marboxil produced.
Mechanistic Insights into n-Pentyl Protected Intermediate Coupling
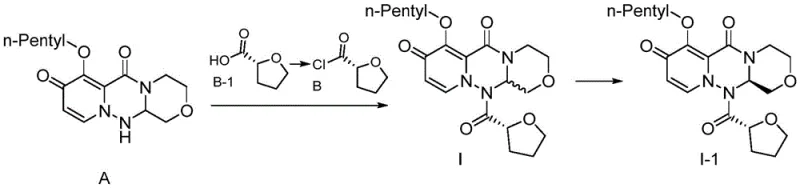
The core of this technological breakthrough lies in the meticulous manipulation of protecting group chemistry to control stereochemistry and reactivity throughout the synthesis. The n-pentyl group serves as a robust shield for the phenolic hydroxyl moiety, preventing it from participating in nucleophilic attacks or elimination reactions that typically lead to impurity profiles in conventional routes. During the condensation with (R)-(+)-tetrahydrofuran-2-formyl chloride, the steric bulk and electronic properties of the n-pentyl chain ensure that the reaction proceeds with high regioselectivity, minimizing the formation of isomeric byproducts that are notoriously difficult to separate. The mechanism involves a precise sequence where the protected intermediate is condensed under mild acid-binding conditions, followed by a controlled deprotection step using alkali metal halides and triethylamine. This sequence ensures that the chiral center at the oxazine pyridine triazine ring remains intact, preserving the biological activity of the final pharmaceutical intermediate.
Impurity control is further enhanced by the strategic selection of solvents and reagents that favor the formation of the desired crystalline form of the product. In the coupling step with the dibenzothiophene fragment, the use of a mixed solvent system comprising ethyl acetate and n-hexane allows for the fine-tuning of solubility parameters. This adjustment ensures that the target compound precipitates out of the solution while impurities remain dissolved, effectively acting as an in-situ purification step that reduces the need for extensive chromatographic separation. The removal of the n-pentyl group is subsequently achieved under mild alkaline conditions that do not compromise the integrity of the sensitive ester linkages in the molecule. This mechanistic precision results in a final product with isomer purity not lower than 99.2%, demonstrating the superior capability of this route to deliver commercial scale-up of complex pharmaceutical intermediates with consistent quality.
How to Synthesize Baloxavir Marboxil Efficiently
The implementation of this synthesis route requires a disciplined approach to reaction conditions and parameter control to fully realize the efficiency gains promised by the patent. The process begins with the preparation of the key n-pentyl protected intermediate, which serves as the foundation for the subsequent high-yield coupling reactions. Operators must adhere to strict temperature controls during the chlorination and condensation steps to prevent the formation of thermal degradation products. The detailed standardized synthesis steps involve precise molar ratios of reagents, such as the 1:0.6 to 0.8 ratio of tetrahydrofuran-2-formic acid to oxalyl chloride, ensuring optimal conversion without excess waste. Following the initial coupling, the deprotection and final docking steps require careful monitoring of pH and solvent composition to induce the desired crystallization behavior. For the complete technical execution, the detailed standardized synthesis steps are provided in the guide below.
- Protect the phenolic hydroxyl group with an n-pentyl group to form a stable intermediate, preventing side reactions during subsequent coupling steps.
- Condense the protected intermediate with (R)-(+)-tetrahydrofuran-2-formyl chloride using mild acid-binding agents to ensure high stereochemical purity.
- Execute the final coupling with the dibenzothiophene fragment in a polarity-adjusted solvent system to facilitate easy product precipitation and purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this optimized synthesis route offers substantial advantages that directly impact the bottom line and supply chain resilience for pharmaceutical manufacturers. The elimination of controlled reagents like 1-propylphosphoric cyclic anhydride from the early stages of synthesis removes a significant regulatory hurdle, simplifying the sourcing process and reducing the administrative burden associated with handling restricted chemicals. This change not only mitigates the risk of supply disruptions caused by regulatory compliance issues but also lowers the overall cost of raw material acquisition. Furthermore, the simplified operational protocol reduces the requirement for specialized equipment and extensive operator training, leading to lower overhead costs and faster technology transfer times between manufacturing sites. The ability to produce high-purity intermediates with fewer purification steps translates into significant cost savings in terms of solvent consumption, waste disposal, and energy usage, making the process economically superior to legacy methods.
- Cost Reduction in Manufacturing: The streamlined process eliminates expensive and regulated reagents, drastically simplifying the bill of materials and reducing the direct cost of goods. By avoiding the use of T3P in critical early steps, the method removes the need for costly removal processes for heavy metal or phosphorus residues, which traditionally require additional scavenging resins and filtration steps. The higher reaction yield means that less starting material is required to produce the same amount of final API, effectively lowering the material cost per kilogram of output. Additionally, the reduced need for complex chromatographic purification lowers the consumption of high-grade solvents and silica gel, contributing to a leaner and more cost-effective manufacturing operation that maximizes profit margins.
- Enhanced Supply Chain Reliability: The reliance on commercially available and non-controlled reagents enhances the stability of the supply chain by diversifying the pool of potential vendors. Unlike processes dependent on single-source or highly regulated chemicals, this route allows procurement teams to source raw materials from multiple suppliers, reducing the risk of bottlenecks and price volatility. The robustness of the n-pentyl protection strategy ensures consistent batch-to-batch quality, minimizing the risk of production failures that could lead to delays in delivery schedules. This reliability is crucial for maintaining continuous supply to downstream formulation partners, ensuring that market demand for antiviral medications is met without interruption even during periods of global supply chain stress.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing solvent systems and reaction conditions that are easily transferable from pilot plant to commercial production scales. The ability to induce product precipitation through solvent polarity adjustment reduces the volume of liquid waste generated, aligning with increasingly stringent environmental regulations and sustainability goals. Fewer purification steps mean a lower environmental footprint in terms of energy consumption and chemical waste disposal, making the facility more compliant with green chemistry principles. This scalability ensures that production capacity can be rapidly expanded to meet surges in demand without the need for significant capital investment in new purification infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis method. These answers are derived directly from the patent specifications and are intended to provide clarity on the operational benefits and chemical mechanisms involved. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their own manufacturing portfolios. The responses highlight the specific advantages in terms of purity, yield, and regulatory compliance that distinguish this method from conventional approaches.
Q: How does the n-pentyl protection strategy improve yield compared to conventional benzyl protection?
A: The n-pentyl group provides superior steric and electronic stability compared to benzyl groups, significantly reducing epimerization and de-protection side reactions during the critical coupling phase, thereby enhancing overall process yield.
Q: Does this synthesis route eliminate the need for controlled reagents like T3P in early stages?
A: Yes, the optimized route strategically removes the requirement for 1-propylphosphoric cyclic anhydride (T3P) in the initial condensation steps, simplifying regulatory compliance and reducing raw material sourcing complexity.
Q: What are the scalability advantages of the solvent adjustment method described in the patent?
A: By adjusting solvent polarity to induce product precipitation, the method eliminates complex chromatographic purification steps, making the process highly amenable to large-scale industrial crystallization and filtration operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Baloxavir Marboxil Supplier
The technical potential of this optimized synthesis route is immense, offering a pathway to produce high-quality antiviral APIs with unprecedented efficiency and consistency. NINGBO INNO PHARMCHEM, as a seasoned CDMO expert, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative process to life. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, ensuring that every batch of Baloxavir Marboxil meets the exacting standards of the global pharmaceutical industry. We understand the critical nature of supply chain continuity and are committed to leveraging this advanced chemistry to provide our partners with a reliable and cost-effective source of this vital medication.
We invite you to engage with our technical procurement team to discuss how this synthesis route can be integrated into your supply chain to achieve significant operational improvements. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits specific to your production volume and requirements. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will enhance your competitive position in the market. Our team is ready to support your journey from development to commercialization with the highest level of technical expertise and service.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
