Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles: A Breakthrough in Cost-Effective Pharmaceutical Intermediate Manufacturing
Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles: A Breakthrough in Cost-Effective Pharmaceutical Intermediate Manufacturing
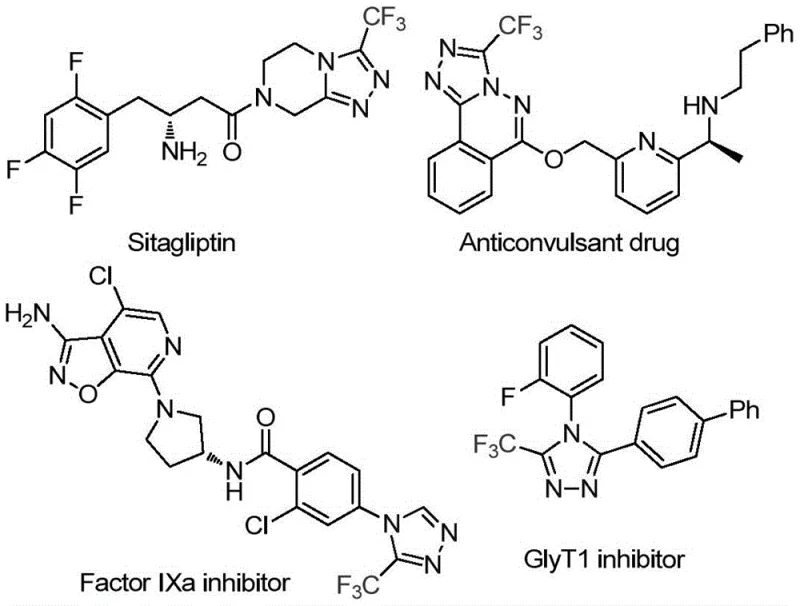
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the urgent need for more sustainable, cost-effective, and operationally simple synthetic routes. A pivotal advancement in this domain is detailed in patent CN114920707B, which discloses a novel preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. This technology is particularly relevant for the production of high-value active pharmaceutical ingredients (APIs), as the 1,2,4-triazole scaffold is a privileged structure found in numerous bioactive molecules, including prominent drugs like Sitagliptin and various factor IXa inhibitors. The strategic incorporation of the trifluoromethyl group further enhances the metabolic stability and lipophilicity of these drug candidates, making efficient access to this chemical motif a critical priority for R&D teams globally.
This patented methodology distinguishes itself by utilizing the ubiquitous organic solvent N,N-dimethylformamide (DMF) not merely as a reaction medium, but as a vital carbon source. By leveraging the inherent reactivity of DMF's methyl and formyl groups, the process achieves a tandem cyclization promoted by molecular iodine under remarkably mild conditions. For procurement managers and supply chain directors, this represents a significant opportunity for cost reduction in API manufacturing, as it replaces expensive, specialized reagents with commodity chemicals while eliminating the need for complex inert atmosphere setups. The following analysis provides a deep technical and commercial evaluation of this transformative synthetic approach.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of trifluoromethyl-substituted heterocycles has been fraught with synthetic challenges that hinder large-scale production. Conventional routes often rely on multi-step sequences involving hazardous reagents, stringent anhydrous conditions, and expensive transition metal catalysts that require rigorous removal to meet pharmaceutical purity standards. Many existing protocols necessitate the use of specialized trifluoromethylating agents or pre-functionalized hydrazines that are costly and difficult to source in bulk quantities. Furthermore, the requirement for oxygen-free environments typically demands sophisticated equipment like gloveboxes or Schlenk lines, which increases capital expenditure and limits the throughput of manufacturing facilities. These factors collectively contribute to extended lead times and inflated production costs, creating bottlenecks for reliable pharmaceutical intermediate suppliers aiming to deliver high-purity materials efficiently.
The Novel Approach
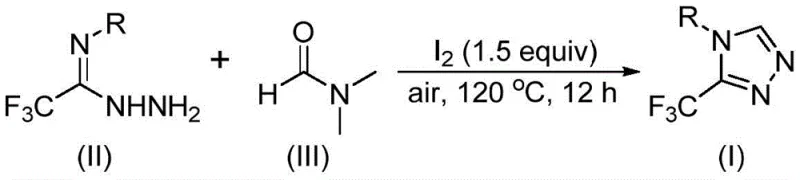
In stark contrast, the method described in patent CN114920707B offers a streamlined, one-pot solution that fundamentally alters the economic and operational dynamics of triazole synthesis. The core innovation lies in the dual role of DMF, which serves as both the solvent and the C1 synthon, thereby simplifying the reaction stoichiometry and minimizing waste generation. The reaction utilizes molecular iodine as a promoter, a readily available and inexpensive halogen source that facilitates the activation of DMF and the subsequent cyclization with trifluoroethyliminohydrazide. Crucially, this transformation proceeds efficiently under an air atmosphere at temperatures between 110°C and 130°C, removing the logistical burden of maintaining inert gas lines. This robustness allows for the commercial scale-up of complex pharmaceutical intermediates with significantly reduced operational overhead, making it an ideal candidate for green chemistry initiatives and continuous flow processing adaptations.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
From a mechanistic perspective, this reaction showcases a fascinating interplay between the solvent and the promoter. The process initiates with the activation of DMF by molecular iodine, which can proceed through two potential pathways depending on which functional group of DMF participates as the carbon source. In one pathway, the formyl group of DMF undergoes condensation with the trifluoroethyliminohydrazide to form a hydrazone intermediate. This is followed by an intramolecular cyclization that eliminates a molecule of dimethylamine, directly yielding the 3-trifluoromethyl-substituted 1,2,4-triazole core. Alternatively, the N-methyl group of DMF can act as the carbon donor, where iodine facilitates the formation of an amine salt. Subsequent nucleophilic addition and elimination steps generate an azadiene intermediate, which then undergoes oxidative aromatization to furnish the final heterocyclic product. Understanding these pathways is essential for R&D directors focused on impurity control, as it highlights the specific byproducts (such as dimethylamine or iodine salts) that must be managed during downstream processing.
The versatility of this mechanism is further evidenced by its tolerance to a wide array of electronic environments on the substrate. The patent data indicates that the reaction accommodates various substituents on the aromatic ring of the starting hydrazide, including electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as halogens and trifluoromethyl moieties. This broad substrate scope suggests that the rate-determining step is likely robust against steric and electronic variations, ensuring consistent yields across different derivatives. For process chemists, this predictability is invaluable, as it reduces the need for extensive condition screening when adapting the route to new analogues. The ability to synthesize diverse 4-substituted 3-trifluoromethyl-1,2,4-triazoles through simple substrate design underscores the practical utility of this method in medicinal chemistry campaigns aimed at structure-activity relationship (SAR) studies.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazoles Efficiently
The implementation of this synthesis protocol is designed for maximum operational simplicity, allowing laboratories and pilot plants to adopt the method with minimal retooling. The procedure involves combining the trifluoroethyliminohydrazide precursor with molecular iodine in DMF, followed by heating under ambient air conditions. The reaction time typically ranges from 10 to 15 hours, providing a balanced trade-off between conversion and throughput. Post-reaction workup is straightforward, involving standard filtration and purification techniques such as column chromatography or recrystallization, which are well-established in industrial settings.
- Combine molecular iodine, trifluoroethyliminohydrazide, and organic solvent DMF in a reaction vessel under an air atmosphere.
- Heat the reaction mixture to a temperature range of 110-130°C and maintain stirring for a duration of 10 to 15 hours to ensure complete conversion.
- Upon completion, perform standard post-treatment procedures including filtration, washing, drying, and purification via column chromatography to isolate the target triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this DMF-based synthesis route offers compelling strategic advantages that extend beyond mere chemical efficiency. The primary driver of value is the drastic simplification of the raw material portfolio. By utilizing DMF, a solvent that is already a staple in almost every chemical inventory, the need to procure specialized, high-cost carbon sources is eliminated. This consolidation of reagents not only lowers the direct material costs but also simplifies logistics and storage requirements, reducing the risk of supply chain disruptions associated with niche chemicals. Furthermore, the use of molecular iodine, a commodity halogen, ensures that the promoter remains affordable and widely available from multiple global vendors, enhancing supply security.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound due to the elimination of expensive catalysts and the reduction of processing steps. Traditional methods often require precious metal catalysts that necessitate costly scavenging processes to meet residual metal specifications for APIs. By replacing these with molecular iodine and a solvent-as-reagent strategy, the process inherently lowers the cost of goods sold (COGS). Additionally, the absence of strict anhydrous requirements means that energy consumption for solvent drying and nitrogen purging is significantly curtailed, leading to substantial operational savings over the lifecycle of the product.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions directly translates to improved supply chain resilience. Because the reaction tolerates air and moisture, it is less susceptible to batch failures caused by minor leaks or environmental fluctuations, which are common pain points in large-scale manufacturing. This reliability ensures consistent delivery schedules and reduces the safety stock levels that buyers typically need to maintain. The use of common solvents and reagents also means that alternative suppliers can be qualified quickly, mitigating the risk of single-source dependency and ensuring business continuity even during market volatility.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this method aligns perfectly with modern green chemistry principles. The atom economy is improved by incorporating the solvent into the final product, thereby reducing the overall mass intensity of the process. The simplified workup generates less hazardous waste compared to multi-step organometallic routes, easing the burden on waste treatment facilities and lowering disposal costs. The straightforward nature of the reaction makes it highly amenable to scale-up from kilogram to tonne quantities without the need for specialized high-pressure or cryogenic equipment, facilitating rapid technology transfer from R&D to commercial production.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of this technology for their specific applications, we have compiled a set of frequently asked questions based on the patent specifications. These insights address common concerns regarding reaction conditions, substrate compatibility, and purification strategies.
Q: What is the primary advantage of using DMF in this triazole synthesis?
A: The primary advantage is the dual functionality of DMF, which acts simultaneously as the reaction solvent and the carbon source (C1 synthon). This eliminates the need for separate, expensive carbon-building reagents and simplifies the workup process by reducing the number of components in the reaction mixture.
Q: Does this method require strict anhydrous or anaerobic conditions?
A: No, one of the most significant operational benefits of this patented method is that it proceeds efficiently under an air atmosphere without the need for rigorous anhydrous or oxygen-free conditions. This drastically reduces the equipment requirements and operational complexity compared to traditional organometallic approaches.
Q: What is the scope of substrates compatible with this iodine-promoted cyclization?
A: The method demonstrates broad substrate tolerance, successfully accommodating various substituted aryl groups including those with electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as halogens and trifluoromethyl moieties, making it highly versatile for diverse pharmaceutical intermediate synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes in the development of next-generation therapeutics. Our team of expert process chemists has extensively evaluated the technology disclosed in CN114920707B and possesses the capability to implement this iodine-promoted cyclization at commercial scales. We offer extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-trifluoromethyl-1,2,4-triazole intermediate delivered meets the highest industry standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this innovative synthesis method for your upcoming projects. By partnering with our technical procurement team, you can gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in fine chemical intermediates can accelerate your drug development timeline while optimizing your overall manufacturing budget.
