Advanced Catalytic Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial API Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct nitrogen-containing heterocycles, particularly those incorporating fluorine motifs which are pivotal for enhancing metabolic stability and bioavailability in modern drug candidates. Patent CN113307778A introduces a groundbreaking preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds, addressing critical bottlenecks in current synthetic methodologies. This innovation leverages a synergistic molybdenum and copper co-catalytic system to facilitate the cycloaddition of trifluoroethylimidoyl chloride with functionalized isonitriles. For R&D directors and procurement specialists alike, this represents a significant opportunity to streamline the supply chain for high-value pharmaceutical intermediates. The protocol operates under remarkably mild conditions, utilizing commercially available reagents that reduce both the complexity and the environmental footprint of the synthesis. By enabling the direct construction of the triazole core with high atom economy, this technology supports the rapid development of next-generation therapeutics, including analogs of blockbuster drugs like Sitagliptin.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted 1,2,4-triazoles has been plagued by significant operational challenges that hinder efficient commercial manufacturing. Traditional literature methods predominantly rely on the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinone. These legacy routes often necessitate harsh reaction conditions, multiple synthetic steps, and the use of unstable or difficult-to-handle precursors like diazonium salts or trifluorodiazoethane. Furthermore, many existing copper-catalyzed multi-component reactions suffer from limited substrate scope and poor functional group tolerance, leading to lower overall yields and increased purification burdens. For a procurement manager, these inefficiencies translate directly into higher raw material costs and extended lead times, while for supply chain heads, the reliance on hazardous reagents poses safety and regulatory compliance risks that can disrupt continuous production schedules.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the method disclosed in CN113307778A offers a streamlined, one-pot solution that dramatically simplifies the manufacturing landscape. By employing trifluoroethylimidoyl chloride and functionalized isonitrile (NIITP) as starting materials, the reaction bypasses the need for pre-formed hydrazines or unstable diazo species. The core of this innovation lies in the dual-catalyst system comprising molybdenum hexacarbonyl and cuprous acetate, which activates the isonitrile and promotes the [3+2] cycloaddition with exceptional efficiency. This approach not only tolerates a wide array of functional groups—including halogens, alkyls, and electron-withdrawing groups—but also operates in common aprotic solvents like THF at moderate temperatures of 70°C to 90°C. 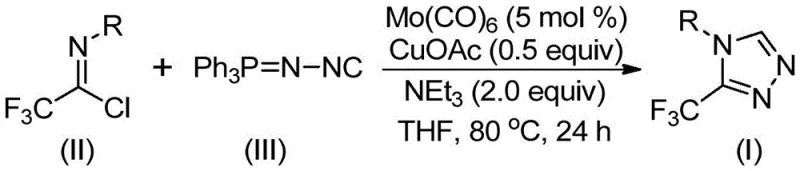
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific API intermediates. The reaction initiates with the coordination of molybdenum hexacarbonyl to the functionalized isonitrile, forming a reactive metal-isocyanide complex that enhances the nucleophilicity of the carbon center. Subsequently, the cuprous acetate acts as a promoter, facilitating the attack of this activated species onto the electrophilic carbon of the trifluoroethylimidoyl chloride. This interaction triggers a concerted [3+2] cycloaddition event, constructing the five-membered 1,2,4-triazole ring intermediate with high regioselectivity. The presence of triethylamine serves to neutralize the hydrochloric acid byproduct generated during the cyclization, driving the equilibrium forward and preventing catalyst deactivation. Finally, the elimination of triphenylphosphine oxide, derived from the isonitrile precursor, occurs under the reaction conditions, yielding the stable 3-trifluoromethyl-substituted triazole product. This elegant mechanism avoids the formation of toxic byproducts associated with older hydrazine-based routes.
From an impurity control perspective, this mechanism offers distinct advantages for ensuring high-purity pharmaceutical intermediates. The mild thermal window of 70°C to 90°C minimizes thermal decomposition pathways that often generate complex impurity profiles in high-temperature cyclizations. Moreover, the specificity of the Mo/Cu catalytic pair reduces the likelihood of side reactions such as polymerization of the isonitrile or hydrolysis of the imidoyl chloride. The use of molecular sieves in the reaction mixture further scavenges trace moisture, protecting the sensitive imidoyl chloride starting material and ensuring consistent batch-to-batch reproducibility. For quality assurance teams, this means a cleaner crude reaction profile, which significantly reduces the load on downstream purification units like column chromatography or crystallization. The ability to tune the R-group on the imidoyl chloride without compromising the core cyclization efficiency allows for the precise engineering of impurity profiles, ensuring that the final active pharmaceutical ingredient meets stringent global regulatory standards for residual solvents and heavy metals.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific stoichiometric ratios and environmental controls to maximize yield and safety. The protocol dictates a precise molar ratio of trifluoroethylimidoyl chloride to functionalized isonitriles, typically ranging from 1:1 to 1:2, with an optimal preference for 1:1.5 to ensure complete consumption of the limiting reagent. The catalyst loading is remarkably low, with molybdenum hexacarbonyl used at 2-8 mol% and cuprous acetate at 0.2-0.8 equivalents, making the process economically viable even for expensive substrates. The reaction is conducted in anhydrous THF, which provides the ideal solubility balance for both organic reactants and inorganic catalysts. Operators must maintain the reaction temperature strictly between 70°C and 90°C for a duration of 18 to 30 hours, monitoring progress via TLC or HPLC to determine the exact endpoint.
- Combine molybdenum hexacarbonyl, cuprous acetate, triethylamine, molecular sieves, trifluoroethylimidoyl chloride, and functionalized isonitrile in an organic solvent such as THF.
- Heat the reaction mixture to a temperature range of 70°C to 90°C and maintain stirring for a duration of 18 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and perform column chromatography purification to isolate the target 3-trifluoromethyl-1,2,4-triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented methodology translates into tangible strategic benefits that extend beyond mere chemical curiosity. The primary advantage lies in the substantial cost reduction in pharmaceutical intermediates manufacturing driven by the use of commodity-grade starting materials. Trifluoroethylimidoyl chloride and functionalized isonitriles are commercially available in bulk quantities, eliminating the need for custom synthesis of exotic precursors that often bottleneck supply chains. Furthermore, the elimination of hazardous reagents like diazonium salts reduces the regulatory burden and insurance costs associated with handling explosive or highly toxic substances. The simplified workup procedure, which involves basic filtration and standard chromatography, lowers the operational expenditure on specialized equipment and waste treatment facilities. This efficiency allows for a more agile response to market demands, ensuring that critical API intermediates can be sourced reliably without the volatility associated with complex multi-step syntheses.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of inexpensive catalysts such as cuprous acetate and triethylamine, which are fraction of the cost of noble metal catalysts like palladium or rhodium often used in cross-coupling reactions. By avoiding the use of expensive transition metals, the downstream purification process is simplified, as there is no need for costly metal scavenging resins to meet strict ppm limits for heavy metals in drug substances. Additionally, the high reaction efficiency and conversion rates minimize the loss of valuable fluorinated starting materials, which are typically among the most expensive components in the bill of materials. This cumulative effect results in a significantly lower cost of goods sold (COGS) for the final triazole intermediate, providing a competitive pricing advantage in the global marketplace.
- Enhanced Supply Chain Reliability: Supply chain continuity is critically dependent on the availability of raw materials, and this method excels by utilizing reagents that are stable and widely stocked by chemical suppliers globally. Unlike methods relying on freshly prepared diazo compounds which have short shelf-lives and require immediate use, the starting materials here can be stored and transported safely, reducing the risk of production stoppages due to reagent degradation. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in utility supplies, such as cooling water temperature fluctuations, ensuring consistent output quality. This reliability allows supply chain heads to forecast inventory levels with greater accuracy and reduce the need for excessive safety stock, thereby optimizing working capital.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant often reveals hidden pitfalls, but this methodology is designed with scalability in mind, having been validated from gram-level to potential ton-scale production. The use of THF as a solvent is advantageous as it is a standard industrial solvent with well-established recovery and recycling protocols, aligning with green chemistry principles and reducing volatile organic compound (VOC) emissions. The absence of corrosive acids or bases in the main reaction step minimizes equipment corrosion, extending the lifespan of reactor vessels and reducing maintenance downtime. Furthermore, the high atom economy of the cycloaddition reaction ensures that less waste is generated per kilogram of product, simplifying waste disposal compliance and lowering environmental fees, which is increasingly important for maintaining a social license to operate in regulated jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethyl triazole synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of what partners can expect when integrating this route into their manufacturing portfolio. Understanding these details is essential for making informed decisions about process adoption and vendor selection.
Q: What are the primary advantages of this Mo/Cu co-catalyzed method over traditional triazole synthesis?
A: Unlike traditional methods that often require harsh conditions or complex multi-step sequences involving trifluoroacetyl hydrazine, this novel approach utilizes readily available functionalized isonitriles and trifluoroethylimidoyl chloride. The reaction proceeds under mild thermal conditions (70-90°C) with high efficiency and broad substrate tolerance, significantly simplifying the operational workflow for process chemists.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the patent data explicitly confirms that the method can be extended from gram-level reactions to larger scales. The use of inexpensive catalysts like cuprous acetate and common solvents like THF, combined with a straightforward post-treatment process involving filtration and chromatography, supports robust commercial scale-up potential for API intermediate manufacturing.
Q: What is the scope of substituents compatible with this trifluoromethyl triazole synthesis?
A: The methodology demonstrates excellent functional group tolerance. It successfully accommodates various aryl groups including phenyl, substituted phenyls (with methyl, methoxy, fluoro, chloro, nitro groups), and naphthyl groups. This versatility allows for the design and synthesis of diverse 4-substituted 1,2,4-triazole derivatives tailored to specific drug discovery needs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a patented laboratory method to a robust commercial supply chain requires deep technical expertise and unwavering commitment to quality. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from development to market. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications for complex fluorinated heterocycles. We understand that the integrity of your final drug product depends on the quality of the intermediates, which is why we adhere to the highest standards of GMP compliance and process safety. By leveraging our expertise in Mo/Cu catalytic systems, we can offer you a secure, cost-effective source of 3-trifluoromethyl-1,2,4-triazoles that meets the demanding requirements of the global pharmaceutical industry.
We invite you to collaborate with us to unlock the full potential of this innovative synthesis route for your specific application needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and purity targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments for your upcoming projects. Let us handle the complexities of chemical manufacturing so you can focus on delivering life-saving therapies to patients worldwide with confidence and speed.
