Scalable Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazine Compounds for Pharma
Scalable Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazine Compounds for Pharma
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for nitrogen-containing heterocycles due to their prevalence in bioactive molecules. Patent CN116253692A discloses a groundbreaking preparation method for trifluoromethyl substituted 1,2,4-triazine compounds, addressing critical bottlenecks in current manufacturing protocols. This technology leverages a novel [3+3] cycloaddition strategy that operates under exceptionally mild conditions, specifically at temperatures between 20°C and 40°C in an ambient air atmosphere. The significance of this development lies in its ability to introduce the trifluoromethyl moiety, a pharmacophore known to drastically improve metabolic stability and lipophilicity, without the logistical burden of inert gas protection or expensive transition metal catalysts. For R&D teams focused on oncology and antifungal agents, this platform offers a versatile entry point to high-value scaffolds.
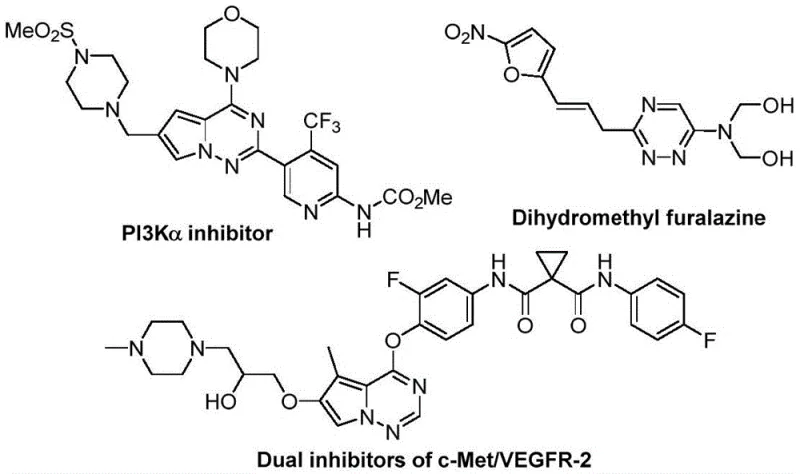
As a reliable pharmaceutical intermediates supplier, we recognize that the structural integrity and purity of these heterocycles are paramount for downstream drug development. The disclosed method utilizes chlorohydrazones and trifluoroacetyl sulfur ylides as key building blocks, reacting them in the presence of potassium carbonate. This approach not only simplifies the supply chain by avoiding exotic reagents but also ensures that the resulting products maintain high purity profiles essential for regulatory compliance. The versatility of the substituents allowed on the triazine ring—ranging from alkyl and phenyl to naphthyl and furyl groups—provides medicinal chemists with a broad chemical space to explore structure-activity relationships (SAR) efficiently.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,4-triazine derivatives has relied on condensation reactions between amidrazones and 1,2-diketones or alkynes, often requiring elevated temperatures and prolonged reaction times. Traditional multicomponent reactions involving hydrazides and dicarbonyl compounds frequently suffer from poor atom economy and generate significant amounts of waste, complicating the purification process. Furthermore, many existing protocols necessitate the use of sensitive reagents that degrade upon exposure to moisture or oxygen, mandating the use of gloveboxes or Schlenk lines which increases operational costs. These conventional pathways often lack the structural diversity required for modern drug discovery, limiting the ability to rapidly generate analog libraries for screening purposes. The reliance on harsh conditions can also lead to the decomposition of sensitive functional groups, thereby reducing overall yield and increasing the complexity of the impurity profile.
The Novel Approach
In stark contrast, the methodology described in CN116253692A represents a paradigm shift towards green and sustainable chemistry. By employing a synergistic [3+3] cycloaddition between a nitrile imine intermediate and a sulfur ylide, this process constructs the triazine core with remarkable efficiency. The reaction proceeds smoothly in common organic solvents like tetrahydrofuran (THF) at room temperature, eliminating the energy costs associated with heating or cooling. Crucially, the system is tolerant to air and moisture, removing the need for rigorous exclusion of atmospheric components. This operational simplicity translates directly into cost reduction in pharma manufacturing, as it reduces the requirement for specialized equipment and highly trained personnel. The use of inexpensive potassium carbonate as a promoter further enhances the economic viability of this route compared to methods utilizing precious metal catalysts.
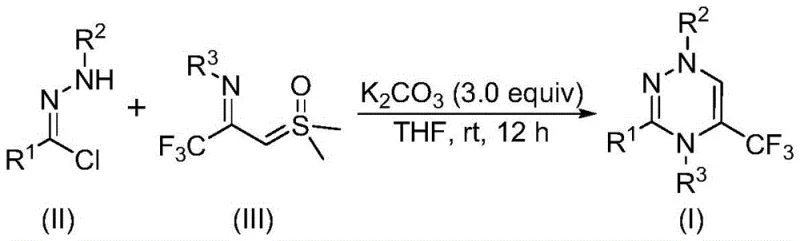
Mechanistic Insights into [3+3] Cycloaddition and Nitrile Imine Formation
The core of this synthetic innovation lies in the in situ generation of a reactive nitrile imine species. Under the promotion of potassium carbonate, the chlorohydrazone precursor undergoes dehydrohalogenation, eliminating a molecule of hydrogen chloride to form the nitrile imine intermediate. This highly reactive 1,3-dipole then engages in a concerted [3+3] cycloaddition with the trifluoroacetyl sulfur ylide. This mechanistic pathway is distinct from traditional stepwise nucleophilic substitutions, offering a more direct route to the six-membered heterocyclic ring. The subsequent elimination of dimethyl sulfoxide (DMSO) drives the reaction forward, ensuring high conversion rates. Understanding this mechanism is vital for process chemists aiming to optimize reaction parameters, as the stability of the nitrile imine and the reactivity of the ylide are key determinants of the final yield.
From an impurity control perspective, the mildness of this reaction conditions plays a pivotal role. Harsh acidic or basic conditions often lead to the hydrolysis of sensitive esters or amides present in complex drug candidates; however, this neutral to mildly basic environment preserves such functionalities. The absence of heavy metals means there is no risk of metal leaching into the final product, a critical consideration for API intermediates where residual metal limits are strictly regulated by agencies like the FDA and EMA. The clean reaction profile minimizes the formation of side products, simplifying the downstream purification process. This results in high-purity trifluoromethyl triazines that require less intensive chromatographic separation, thereby reducing solvent consumption and waste generation.
How to Synthesize Trifluoromethyl 1,2,4-Triazine Efficiently
To implement this synthesis effectively, precise control over stoichiometry and reaction time is essential. The patent data suggests a molar ratio of chlorohydrazone to trifluoroacetyl sulfur ylide to potassium carbonate of approximately 1:2:3 for optimal results. The reaction is typically conducted in THF, which effectively dissolves both organic precursors and the inorganic base, facilitating homogeneous mixing. While the reaction can complete within 10 hours, extending the time to 14 hours ensures full conversion of the starting materials, particularly for substrates with bulky steric groups. Detailed standardized synthesis steps follow below to guide your laboratory execution.
- Prepare the reaction mixture by adding potassium carbonate, chlorohydrazone, and trifluoroacetyl sulfur ylide into an organic solvent such as tetrahydrofuran (THF).
- Stir the mixture at room temperature (20-40°C) in an air atmosphere for 10 to 14 hours to allow the [3+3] cycloaddition to proceed.
- Upon completion, filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final trifluoromethyl substituted 1,2,4-triazine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this metal-free synthesis offers tangible strategic benefits beyond mere technical feasibility. The reliance on commodity chemicals such as acyl chlorides, hydrazines, and potassium carbonate ensures a stable and resilient supply chain, mitigating the risks associated with sourcing specialized catalysts that may have long lead times or geopolitical supply constraints. The ability to run reactions in air significantly reduces the capital expenditure required for reactor infrastructure, as nitrogen blanketing systems become unnecessary. This flexibility allows for rapid scale-up from gram-scale discovery to multi-kilogram production without the need for re-engineering the process.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts removes a significant cost driver from the bill of materials. Furthermore, the absence of metals negates the need for costly scavenging resins or additional purification steps designed to lower metal residues to ppm levels. The use of cheap, non-toxic potassium carbonate instead of strong organic bases or cryogenic conditions further lowers utility and raw material costs. These factors combined result in substantial cost savings that can be passed down the value chain, making the final API more competitive in the marketplace.
- Enhanced Supply Chain Reliability: The starting materials, including various substituted chlorohydrazones and sulfur ylides, are derived from widely available bulk chemicals. This broad availability reduces the risk of supply disruptions caused by single-source dependencies. Additionally, the robustness of the reaction to atmospheric conditions means that production schedules are less vulnerable to delays caused by equipment maintenance related to inert gas systems. This reliability ensures consistent delivery of high-purity intermediates, supporting uninterrupted downstream synthesis operations.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, primarily consisting of benign salts and organic solvents that can be readily recovered and recycled. The avoidance of heavy metals aligns with increasingly stringent environmental regulations regarding waste disposal and effluent treatment. The simplicity of the work-up procedure, involving filtration and standard chromatography, facilitates easy scale-up to industrial reactors. This scalability ensures that the method remains viable and efficient whether producing kilograms for clinical trials or tons for commercial launch.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethyl triazine synthesis platform. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering a clear picture of the technology's capabilities and limitations for potential adopters.
Q: What are the key advantages of this synthesis method over traditional routes?
A: This method eliminates the need for expensive heavy metal catalysts and harsh reaction conditions. It operates at room temperature in air, uses cheap inorganic bases like potassium carbonate, and offers high structural diversity for drug discovery.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process is highly scalable. The use of readily available starting materials, mild conditions (20-40°C), and simple post-treatment (filtration and chromatography) makes it ideal for commercial scale-up without specialized inert atmosphere equipment.
Q: What is the role of the trifluoromethyl group in these compounds?
A: The incorporation of the trifluoromethyl group significantly enhances the physicochemical properties of the parent compound, including improved metabolic stability, lipophilicity, and bioavailability, which are critical for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl 1,2,4-Triazine Supplier
At NINGBO INNO PHARMCHEM, we understand that translating a promising patent into a commercial reality requires more than just a recipe; it demands deep process engineering expertise. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to plant floor is seamless. We are committed to delivering trifluoromethyl 1,2,4-triazine compounds that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex heterocyclic chemistry positions us as a strategic partner for your long-term drug development projects.
We invite you to collaborate with us to leverage this advanced synthetic technology for your pipeline. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing processes can accelerate your time to market while maximizing value.
