Advanced Molybdenum-Copper Catalysis for Scalable 3-Trifluoromethyl-1,2,4-Triazole Production
Advanced Molybdenum-Copper Catalysis for Scalable 3-Trifluoromethyl-1,2,4-Triazole Production
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those incorporating fluorine atoms which are pivotal for enhancing metabolic stability and bioavailability. A groundbreaking development in this domain is detailed in Chinese Patent CN113307778A, which discloses a highly efficient preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. This technology represents a significant leap forward for manufacturers acting as a reliable pharmaceutical intermediate supplier, offering a pathway to synthesize complex heterocyclic scaffolds under remarkably mild conditions. The structural motif of 1,2,4-triazole is ubiquitous in modern medicinal chemistry, found in blockbuster drugs such as Sitagliptin and various antifungal agents, making the ability to install a trifluoromethyl group at the 3-position a critical capability for any advanced CDMO.
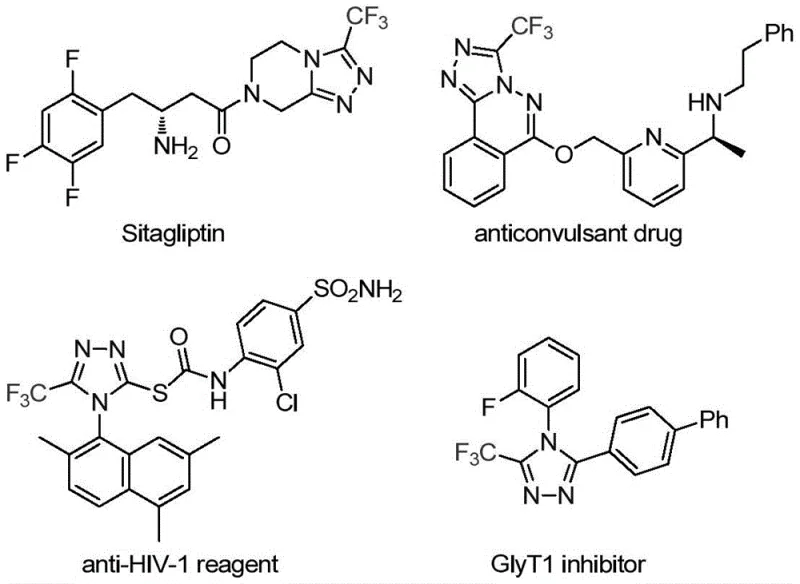
The significance of this patent lies not only in the novelty of the catalyst system but also in its practical applicability for cost reduction in API manufacturing. By utilizing a dual catalytic system comprising molybdenum hexacarbonyl and cuprous acetate, the process avoids the extreme temperatures and hazardous reagents often associated with traditional triazole synthesis. For R&D directors and process chemists, this translates to a safer, more controllable reaction profile that minimizes the formation of difficult-to-remove impurities. Furthermore, the method's compatibility with a wide range of functional groups ensures that it can be adapted for the synthesis of diverse analogues, thereby accelerating the drug discovery timeline for potential new chemical entities targeting various therapeutic areas.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has been fraught with synthetic challenges that hinder large-scale production. Traditional literature reports predominantly rely on the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones. These classical routes often suffer from poor atom economy, require the handling of unstable hydrazine derivatives, and frequently necessitate harsh reaction conditions that can degrade sensitive functional groups on the substrate. Additionally, alternative multi-component reactions involving diazonium salts and trifluorodiazoethane pose significant safety risks due to the explosive nature of diazo compounds, creating substantial bottlenecks for commercial scale-up of complex polymer additives or pharmaceutical intermediates. The reliance on such hazardous materials increases operational costs related to safety infrastructure and waste disposal, making these legacy methods less attractive for modern green chemistry initiatives.
The Novel Approach
In stark contrast, the methodology described in CN113307778A introduces a transformative strategy that leverages the cycloaddition of trifluoroethylimidoyl chloride with functionalized isonitriles. This novel approach eliminates the need for dangerous diazo reagents and unstable hydrazines, replacing them with stable, commercially available starting materials. The reaction proceeds smoothly in common organic solvents like THF at moderate temperatures ranging from 70°C to 90°C. As illustrated in the reaction scheme below, the transformation is driven by a synergistic catalytic system that ensures high conversion rates and excellent yields, with some substrates achieving yields as high as 99% (e.g., Example 8). This shift towards milder, safer reagents directly addresses the pain points of procurement managers looking for reducing lead time for high-purity pharmaceutical intermediates by simplifying the supply chain for raw materials.
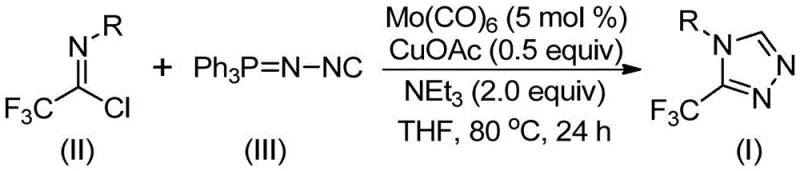
Mechanistic Insights into Mo/Cu Co-catalyzed Cycloaddition
From a mechanistic perspective, the success of this transformation hinges on the unique interplay between the molybdenum and copper catalysts. The process initiates with the activation of the functionalized isonitrile (Ph3P=N-NC) by molybdenum hexacarbonyl, forming a reactive metal-isocyanide complex. This activation step is crucial as it enhances the nucleophilicity of the isonitrile carbon, preparing it for the subsequent cycloaddition event. Simultaneously, the cuprous acetate acts as a promoter for the [3+2] cycloaddition reaction between the activated isonitrile species and the trifluoroethylimidoyl chloride. This dual-catalyst mechanism effectively lowers the activation energy barrier, allowing the formation of the five-membered triazole ring to proceed efficiently at 80°C. The presence of triethylamine serves as a base to neutralize the hydrochloric acid byproduct generated during the cyclization, driving the equilibrium towards the product side and preventing acid-catalyzed decomposition of the sensitive intermediates.
Following the initial ring closure, the reaction mixture undergoes a hydrolysis step where water present in the system facilitates the removal of the triphenylphosphine oxide moiety. This elimination step is vital for aromatization, yielding the final stable 3-trifluoromethyl-substituted 1,2,4-triazole compound. The robustness of this mechanism is evidenced by its tolerance to various electronic environments on the aromatic ring of the imidoyl chloride. Whether the substrate bears electron-donating groups like methyl or methoxy, or electron-withdrawing groups like fluoro or nitro, the catalytic cycle remains effective. This broad substrate scope is a testament to the versatility of the Mo/Cu system, providing process chemists with a powerful tool for generating diverse chemical libraries without the need for extensive re-optimization of reaction conditions for each new analogue.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The operational simplicity of this synthesis makes it highly attractive for both laboratory-scale optimization and pilot plant production. The standard protocol involves charging a reaction vessel with the catalysts, base, and solvent, followed by the addition of the key coupling partners. The reaction is then heated and monitored until completion, typically within 24 hours. Post-reaction processing is straightforward, involving simple filtration to remove metal residues and molecular sieves, followed by standard purification techniques. For detailed procedural specifics regarding stoichiometry, solvent volumes, and purification parameters, please refer to the standardized synthesis guide below.
- Combine molybdenum hexacarbonyl (5 mol %), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), and molecular sieves in an organic solvent such as THF.
- Add trifluoroethylimidoyl chloride (Formula II) and functionalized isonitrile (Formula III, Ph3P=N-NC) to the reaction mixture under inert atmosphere.
- Heat the reaction mixture to 70-90°C (optimally 80°C) and stir for 18-30 hours until completion, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers tangible strategic benefits beyond mere chemical efficiency. The shift away from hazardous diazonium salts and complex hydrazine precursors significantly de-risks the manufacturing process, leading to lower insurance premiums and reduced regulatory compliance burdens. Furthermore, the use of commodity chemicals like triethylamine and cuprous acetate, alongside readily synthesized imidoyl chlorides, ensures a stable and resilient supply chain that is less susceptible to market volatility compared to specialized reagents required by older methods.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of inexpensive and abundant catalysts. Molybdenum hexacarbonyl and cuprous acetate are significantly cheaper than many precious metal catalysts (such as palladium or rhodium) often used in heterocycle synthesis. Additionally, the high reaction efficiency and yields observed across a broad range of substrates minimize raw material waste. The simplified workup procedure, which avoids complex extraction sequences or specialized scavenging resins, further reduces operational expenditures related to labor and consumables, driving down the overall cost of goods sold for the final triazole intermediate.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, specifically the trifluoroethylimidoyl chlorides and the functionalized isonitrile, are either commercially available or can be prepared in high yields from basic feedstocks like aromatic amines and trifluoroacetic acid. This accessibility means that production schedules are less likely to be disrupted by raw material shortages. The robustness of the reaction conditions also implies that the process can be transferred between different manufacturing sites with minimal loss of efficiency, providing supply chain leaders with the flexibility to diversify their manufacturing footprint and mitigate geopolitical or logistical risks.
- Scalability and Environmental Compliance: The method's compatibility with gram-scale reactions, as demonstrated in the patent examples, indicates a clear path towards kilogram and ton-scale production. The reaction operates in THF, a solvent that is widely accepted in pharmaceutical manufacturing and can be efficiently recovered and recycled. Moreover, the absence of heavy metal waste streams associated with stoichiometric oxidants or toxic diazo byproducts simplifies wastewater treatment protocols. This alignment with green chemistry principles not only reduces environmental impact but also streamlines the regulatory approval process for new drug applications that utilize these intermediates.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common inquiries regarding the reaction scope, safety profile, and scalability. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, ensuring accuracy and relevance for process development discussions.
Q: What are the primary advantages of this Mo/Cu catalyzed method over traditional triazole synthesis?
A: Unlike traditional methods requiring harsh hydrazinolysis or unstable diazonium salts, this novel approach utilizes mild conditions (70-90°C) and commercially available starting materials like trifluoroethylimidoyl chloride, resulting in significantly higher reaction efficiency and easier post-treatment.
Q: Can this synthesis method be scaled for industrial production of API intermediates?
A: Yes, the patent explicitly demonstrates that the method is scalable from milligram to gram levels with high conversion rates. The use of robust catalysts like Mo(CO)6 and CuOAc, along with simple workup procedures involving filtration and silica gel mixing, facilitates straightforward commercial scale-up.
Q: What is the substrate scope regarding the R group in the trifluoroethylimidoyl chloride?
A: The method exhibits excellent functional group tolerance. It successfully accommodates various aryl substituents including electron-donating groups (methyl, methoxy, t-butyl) and electron-withdrawing groups (fluoro, chloro, nitro), as well as phenethyl groups, allowing for the diverse design of triazole libraries.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
The technological advancements detailed in CN113307778A represent a significant opportunity for pharmaceutical companies to optimize their supply chains for critical heterocyclic building blocks. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to leverage this novel Mo/Cu co-catalyzed methodology for your specific project needs. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the pre-clinical phase or preparing for market launch. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-trifluoromethyl-1,2,4-triazole delivered meets the highest industry standards for quality and consistency.
We invite you to engage with our technical procurement team to discuss how this efficient synthesis route can be integrated into your manufacturing strategy. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume and purity requirements. We encourage you to contact us today to request specific COA data for our existing inventory or to initiate a dialogue regarding route feasibility assessments for your proprietary analogues. Let us help you secure a reliable, cost-effective, and scalable supply of this essential pharmaceutical intermediate.
