Revolutionizing Coenzyme Q10 Production: A Technical Deep Dive into Olefin Metathesis Scale-Up
The chemical industry is witnessing a paradigm shift in the production of high-value nutraceutical and pharmaceutical intermediates, exemplified by the innovative methodology disclosed in patent CN101139274B. This seminal work introduces a robust strategy for synthesizing Coenzyme Q10 (Ubiquinone-50) by leveraging olefin metathesis, a transformation that fundamentally alters the economic and safety landscape of large-scale manufacturing. Unlike traditional extraction or fermentation methods which suffer from limited scalability and high costs, this chemical synthesis route offers a deterministic path to high purity and consistent supply. The core innovation lies in the strategic disconnection of the molecule into two manageable fragments that are joined via a ruthenium-catalyzed carbon-carbon double bond formation. This approach not only shortens the synthetic lineage but also bypasses the severe safety constraints associated with earlier organometallic protocols. For global procurement leaders, this represents a critical opportunity to secure a more resilient supply chain for this essential mitochondrial cofactor.
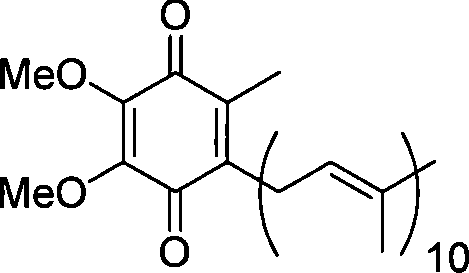
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Coenzyme Q10 has been plagued by complex multi-step sequences that rely on hazardous reagents and tedious purification protocols. Prominent prior art, such as the methods developed by Sato et al., necessitated extensive protection and deprotection strategies to manage the reactivity of the quinone ring, resulting in elongated process times and diminished overall yields often hovering around 28%. Furthermore, the Lipshutz route, while shorter, depended heavily on the use of trimethylaluminum (Me3Al) and zirconocene catalysts for carboalumination steps. These reagents are notoriously pyrophoric, requiring specialized handling equipment, inert atmosphere rigor, and generating significant aluminum waste streams that complicate environmental compliance. The operational risks associated with scaling these exothermic and moisture-sensitive reactions have traditionally acted as a bottleneck for manufacturers seeking to increase capacity without compromising safety standards or inflating insurance and containment costs.
The Novel Approach
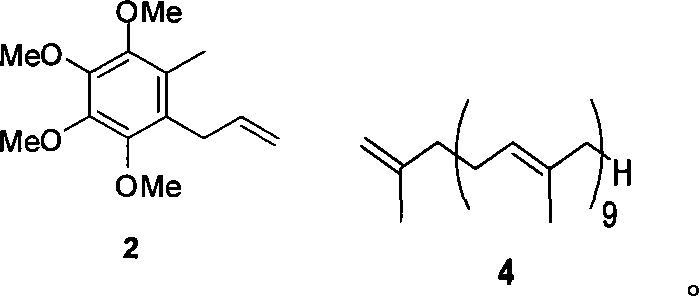
The methodology presented in CN101139274B circumvents these historical impediments by employing a convergent synthesis centered on olefin metathesis. By utilizing 2,3,4,5-tetramethoxytoluene and solanesol as readily available starting materials, the process constructs the critical carbon skeleton through a highly efficient coupling of Compound 2 and Compound 4. This strategy effectively eliminates the need for harsh organoaluminum reagents, replacing them with stable and commercially accessible Grubbs catalysts that operate under much milder conditions. The result is a streamlined six-step sequence that achieves a total yield exceeding 51%, a substantial improvement over legacy technologies. This reduction in step count directly correlates to reduced solvent consumption, lower energy requirements for heating and cooling cycles, and a significantly smaller physical footprint for production facilities. Consequently, this novel approach provides a reliable pathway for the commercial scale-up of complex pharmaceutical intermediates with enhanced economic viability.
Mechanistic Insights into Grubbs-Catalyzed Olefin Metathesis
At the heart of this technological advancement is the application of second-generation Grubbs catalysts to facilitate the union of the aromatic head group and the polyisoprenoid tail. The mechanism proceeds through a [2+2] cycloaddition between the ruthenium carbene species and the terminal olefins present on both coupling partners, forming a metallacyclobutane intermediate. This transient four-membered ring subsequently undergoes cycloreversion to release ethylene gas as a benign byproduct while forging the new internal double bond that links the two fragments. The tolerance of these ruthenium complexes towards various functional groups, including the methoxy substituents on the aromatic ring, is crucial for maintaining high selectivity and preventing side reactions such as isomerization or polymerization of the sensitive solanesol chain. Understanding this catalytic cycle allows process chemists to fine-tune reaction parameters, such as temperature and catalyst loading, to maximize the conversion efficiency while minimizing the formation of homocoupling impurities.
Impurity control in this synthesis is inherently superior due to the avoidance of aggressive Lewis acids and strong nucleophiles that typically degrade the polyene chain. In conventional routes, the use of Me3Al often leads to over-alkylation or decomposition of the sensitive isoprenoid units, creating difficult-to-remove byproducts that compromise the final API purity. In contrast, the metathesis pathway operates with a level of chemoselectivity that preserves the integrity of the ten isoprene units found in the solanesol derivative. The subsequent oxidation step using ammonium cerium nitrate (CAN) is also highly specific, converting the hydroquinone precursor to the target benzoquinone without affecting the alkene geometry. This precision in molecular construction ensures that the final Coenzyme Q10 product meets stringent pharmacopeial specifications for related substances, thereby reducing the burden on downstream purification processes like crystallization or chromatography.
How to Synthesize Coenzyme Q10 Efficiently
The implementation of this synthesis route requires careful attention to the preparation of the two key coupling fragments before the pivotal metathesis event. The process begins with the bromination of 2,3,4,5-tetramethoxytoluene followed by a Grignard reaction to install the allyl group, yielding Compound 2 with high fidelity. Simultaneously, solanesol is activated via bromination and subsequent coupling with methallyl magnesium chloride to produce the terminal olefin side chain, Compound 4. These precursors are then brought together in a dichloromethane solvent system under an inert nitrogen atmosphere, where the Grubbs catalyst mediates the bond formation. Detailed standard operating procedures for each reaction stage, including precise stoichiometry, temperature profiles, and workup protocols, are essential for reproducing the reported yields in a pilot or plant setting. The following guide outlines the critical operational parameters derived from the patent examples to ensure successful technology transfer.
- Bromination of 2,3,4,5-tetramethoxytoluene followed by Grignard reaction with allyl chloride to form Compound 2.
- Conversion of solanesol to solanesyl bromide, followed by reaction with methallyl magnesium chloride to generate Compound 4.
- Coupling of Compound 2 and Compound 4 using Grubbs 2nd Generation catalyst to form the pre-quinone skeleton.
- Final oxidation of the coupled intermediate using ammonium cerium nitrate to yield high-purity Coenzyme Q10.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metathesis-based technology offers profound strategic benefits beyond mere technical elegance. The primary advantage lies in the drastic simplification of the supply chain risk profile by eliminating dependence on hazardous, specialty organometallic reagents that are subject to strict transportation regulations and volatile pricing. By shifting to a catalyst system that is more stable and easier to handle, manufacturers can reduce the costs associated with specialized storage infrastructure and hazardous waste disposal. Furthermore, the reliance on domestically produced raw materials such as 2,3,4,5-tetramethoxytoluene and solanesol insulates the production process from geopolitical supply disruptions that often affect imported fine chemicals. This localization of the supply base enhances the continuity of supply, ensuring that downstream customers in the pharmaceutical and nutraceutical sectors receive their orders without interruption.
- Cost Reduction in Manufacturing: The elimination of expensive and dangerous reagents like trimethylaluminum directly translates to significant cost savings in raw material procurement and safety management. Without the need for complex protection and deprotection steps, the consumption of solvents and auxiliary chemicals is substantially reduced, leading to a leaner and more cost-effective manufacturing process. Additionally, the higher overall yield means that less starting material is wasted, improving the atom economy and lowering the cost of goods sold per kilogram of final product. These efficiencies compound over large production volumes, offering a competitive pricing advantage in the global market for Coenzyme Q10.
- Enhanced Supply Chain Reliability: Utilizing widely available industrial feedstocks ensures that production schedules are not held hostage by the scarcity of niche reagents. The robustness of the Grubbs catalyst system allows for more flexible scheduling and reduced downtime caused by safety incidents or reagent quality issues. This reliability is critical for maintaining long-term contracts with major pharmaceutical clients who require guaranteed delivery timelines. Moreover, the simplified process flow reduces the number of potential failure points in the manufacturing line, further stabilizing the output volume and ensuring consistent availability of high-purity intermediates.
- Scalability and Environmental Compliance: The mild reaction conditions and reduced toxicity of the reagents make this process inherently easier to scale from laboratory to industrial tonnage. Environmental compliance is significantly improved as the process generates less hazardous waste, particularly aluminum salts, which are costly to treat and dispose of. This aligns with increasingly stringent global environmental regulations and corporate sustainability goals, making the facility more attractive to eco-conscious investors and partners. The ability to scale up without proportional increases in environmental liability provides a sustainable long-term growth trajectory for manufacturers adopting this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this olefin metathesis route for Coenzyme Q10 production. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical aspects of the technology. Understanding these details is vital for R&D teams evaluating the feasibility of technology transfer and for procurement officers assessing the long-term viability of the supply source. The answers reflect a commitment to transparency and technical accuracy, ensuring that all stakeholders have a clear understanding of the process capabilities and limitations.
Q: What are the primary safety advantages of this metathesis route over traditional methods?
A: This method eliminates the use of highly pyrophoric and toxic reagents such as trimethylaluminum (Me3Al) and zirconocene catalysts, which were required in previous Lipshutz-type syntheses. By utilizing Grubbs catalysts under milder conditions, the process significantly reduces operational hazards and simplifies waste treatment protocols.
Q: How does this synthetic route impact the overall yield and cost structure?
A: The patent reports a total yield exceeding 51%, which is achieved through a shortened synthetic sequence that minimizes protection and deprotection steps. The use of domestically available raw materials like 2,3,4,5-tetramethoxytoluene and solanesol further stabilizes the cost structure against global supply fluctuations.
Q: Which specific catalysts are recommended for the key coupling step?
A: The technology specifies the use of second-generation Grubbs catalysts or Hoveyda-Grubbs catalysts, with optimal loading typically around 10 mol%. These catalysts provide the necessary stability and activity to couple the sterically hindered side chain efficiently without degrading the sensitive polyene structure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Coenzyme Q10 Supplier
As the global demand for high-quality Coenzyme Q10 continues to rise across the pharmaceutical and functional food sectors, partnering with a manufacturer that possesses advanced synthetic capabilities is more critical than ever. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging cutting-edge technologies like the olefin metathesis route to deliver superior products. Our team boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Coenzyme Q10 meets the highest international standards for safety and efficacy. Our commitment to continuous improvement means we are constantly optimizing our processes to enhance yield and reduce environmental impact.
We invite you to collaborate with us to explore how this advanced synthesis method can benefit your specific product portfolio. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating the tangible economic advantages of switching to our supply chain. Please contact us today to request specific COA data and route feasibility assessments for your next project. By choosing NINGBO INNO PHARMCHEM, you are securing a partnership built on technical excellence, reliability, and a shared vision for a healthier future.
