Advanced Metal-Free Synthesis of Trifluoromethyl Triazoles for Commercial Pharmaceutical Applications
Advanced Metal-Free Synthesis of Trifluoromethyl Triazoles for Commercial Pharmaceutical Applications
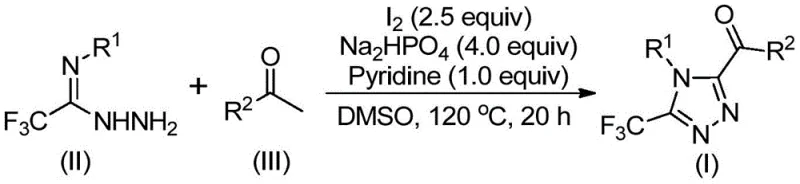
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more sustainable, cost-effective, and scalable synthetic routes. A pivotal development in this domain is disclosed in Chinese Patent CN113105402B, which details a robust preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds. These heterocyclic scaffolds are critical structural motifs found in numerous high-value active pharmaceutical ingredients (APIs), including notable drugs such as Maraviroc, Sitagliptin, and Deferasirox. The introduction of a trifluoromethyl group into these heterocycles is particularly desirable as it enhances metabolic stability, lipophilicity, and bioavailability. However, traditional synthetic pathways often suffer from reliance on scarce resources or complex operational parameters. This patent presents a transformative approach that leverages non-metallic iodine promotion in dimethyl sulfoxide (DMSO), offering a streamlined alternative that addresses both economic and environmental concerns in fine chemical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted 1,2,4-triazole rings, especially those bearing both trifluoromethyl and acyl functionalities, has presented significant challenges for process chemists. Conventional methodologies frequently depend on transition metal catalysts, which introduce substantial complications regarding residual metal removal—a critical quality attribute for any pharmaceutical intermediate intended for human consumption. Furthermore, many existing protocols necessitate stringent reaction conditions, such as strictly anhydrous and oxygen-free environments, which demand specialized equipment like Schlenk lines or gloveboxes, thereby inflating capital expenditure and operational complexity. The use of expensive ligands or exotic reagents further exacerbates the cost structure, making the commercial scale-up of complex heterocycles economically prohibitive for many manufacturers. Additionally, the limited substrate scope of older methods often restricts the ability to introduce diverse functional groups at the 3, 4, and 5 positions, limiting the chemical space available for drug discovery teams.
The Novel Approach
In stark contrast to these legacy techniques, the methodology outlined in patent CN113105402B utilizes a metal-free, iodine-promoted system that operates under remarkably mild and practical conditions. The core innovation lies in the tandem use of iodine and DMSO to facilitate a Kornblum oxidation of aryl ethyl ketones, generating reactive aryl diketone intermediates in situ. These intermediates subsequently undergo condensation with trifluoroethylimide hydrazides followed by oxidative cyclization. This strategy completely bypasses the need for heavy metal catalysts, effectively eliminating the risk of metal contamination and the associated costly purification steps. The reaction tolerates a wide range of functional groups and does not require inert atmosphere protection, significantly lowering the barrier to entry for industrial implementation. By utilizing cheap and readily available starting materials like aryl ketones and elemental iodine, this process offers a highly attractive value proposition for a reliable pharmaceutical intermediate supplier seeking to optimize their production portfolio.
Mechanistic Insights into Iodine-Promoted Cyclization
To fully appreciate the technical elegance of this synthesis, one must delve into the mechanistic pathway that drives the formation of the triazole ring. The reaction initiates with the iodination of the aryl ethyl ketone in the presence of DMSO, which acts as both the solvent and an oxidant. This step generates an alpha-iodo ketone or an aryl diketone species via a Kornblum-type oxidation mechanism. This activated carbonyl species is highly electrophilic and readily reacts with the nucleophilic nitrogen of the trifluoroethylimide hydrazide. The subsequent dehydration leads to the formation of a hydrazone intermediate, which is the precursor to the final heterocyclic ring. The presence of base, specifically sodium dihydrogen phosphate and pyridine, along with additional iodine, promotes the intramolecular cyclization and final aromatization to yield the 3,4,5-trisubstituted 1,2,4-triazole. This cascade sequence is highly efficient, minimizing the accumulation of stable intermediates that could otherwise lead to impurity profiles difficult to separate.
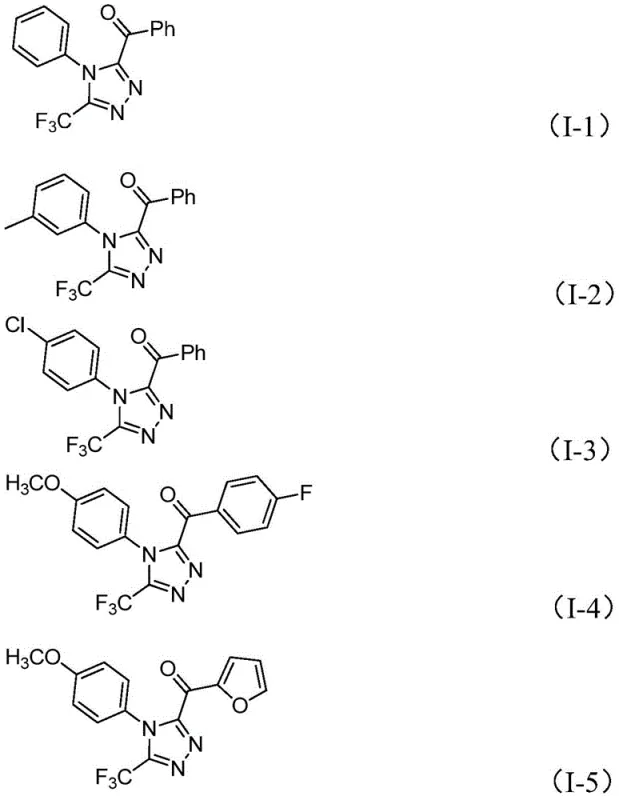
From an impurity control perspective, the choice of reagents plays a pivotal role in ensuring high purity specifications. The use of elemental iodine, while a strong oxidant, is selective enough under the optimized temperature range of 110-130°C to drive the cyclization without causing excessive degradation of the sensitive trifluoromethyl group or the aromatic rings. The patent data indicates that varying the substituents on the aryl rings (R1 and R2) has minimal impact on the reaction efficiency, suggesting a robust mechanism that tolerates electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups like chloro and fluoro. This broad tolerance is essential for a high-purity pharmaceutical intermediate, as it allows for the synthesis of diverse analogues without needing to re-optimize the entire process for each new derivative. The mechanism ensures that side reactions, such as over-oxidation or polymerization, are suppressed, leading to cleaner crude reaction mixtures that are easier to purify via standard column chromatography or crystallization.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific stoichiometric ratios and thermal profiles to maximize yield and minimize byproduct formation. The process is designed as a sequential one-pot reaction, which reduces solvent usage and handling time compared to multi-step isolations. The initial oxidation phase is critical; heating the aryl ketone and iodine in DMSO to 90-110°C for 4-6 hours ensures complete conversion to the activated diketone species before the addition of the hydrazine component. Premature addition of the hydrazine could lead to competing reactions or incomplete oxidation. Following this, the addition of the base system (Na2HPO4 and Pyridine) and the hydrazide triggers the cyclization phase at a slightly elevated temperature of 110-130°C. Detailed standardized synthesis steps see the guide below.
- Mix aryl ethyl ketone and iodine in DMSO, heating to 90-110°C for 4-6 hours to initiate Kornblum oxidation.
- Add trifluoroethylimide hydrazide, sodium dihydrogen phosphate, pyridine, and additional iodine to the mixture.
- Heat the reaction to 110-130°C for 12-20 hours, then filter and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift towards this iodine-promoted methodology represents a strategic opportunity to enhance margin stability and supply security. The primary driver for cost optimization in this process is the elimination of transition metal catalysts. Traditional methods often rely on palladium, copper, or rhodium complexes, which are not only expensive to purchase but also require sophisticated scavenging resins or extensive washing protocols to meet regulatory limits for residual metals. By removing this requirement entirely, the new method drastically simplifies the downstream processing workflow. This reduction in unit operations translates directly into lower labor costs, reduced solvent consumption, and shorter cycle times, all of which contribute to significant cost reduction in API manufacturing without compromising on quality standards.
- Cost Reduction in Manufacturing: The economic benefits extend beyond just the catalyst cost. The raw materials specified in the patent, such as aryl ethyl ketones and elemental iodine, are commodity chemicals produced on a massive global scale. This abundance ensures that price volatility is minimized compared to specialized organometallic reagents. Furthermore, the reaction does not require expensive anhydrous solvents or inert gas blankets (nitrogen/argon), allowing the use of standard stainless steel reactors rather than glass-lined or Hastelloy vessels dedicated to sensitive chemistry. The combination of cheap inputs and simplified infrastructure requirements creates a lean manufacturing model that improves overall gross margins for the final triazole intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the availability of starting materials. Since the key precursors are widely available from multiple chemical vendors globally, the risk of single-source bottlenecks is significantly mitigated. The robustness of the reaction conditions—specifically the tolerance to moisture and oxygen—means that the process is less susceptible to failures caused by minor deviations in plant operations or utility fluctuations. This reliability ensures consistent delivery schedules for downstream customers, reducing the need for safety stock and enabling a more agile just-in-time inventory strategy for pharmaceutical clients who depend on these intermediates for their own production timelines.
- Scalability and Environmental Compliance: Scaling chemical processes from gram to ton scale often introduces unforeseen thermal and mixing challenges. However, the patent explicitly notes that this method can be easily expanded to the gram level and implies suitability for larger scales due to the absence of hazardous reagents. The use of DMSO, while requiring careful recovery, is a standard solvent in the industry with established recycling protocols. Moreover, avoiding heavy metals aligns perfectly with increasingly stringent environmental regulations regarding waste disposal and effluent treatment. This environmental compliance reduces the liability and cost associated with hazardous waste management, making the process more sustainable and future-proof against tightening regulatory frameworks in both domestic and international markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this triazole synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating this route for adoption. Understanding these nuances is critical for assessing the feasibility of integrating this method into existing production lines.
Q: Why is this iodine-promoted method superior to transition metal catalysis?
A: This method eliminates the need for expensive and toxic heavy metal catalysts, significantly simplifying downstream purification and reducing residual metal risks in pharmaceutical intermediates.
Q: What are the key raw materials required for this synthesis?
A: The process utilizes commercially available aryl ethyl ketones and trifluoroethylimide hydrazides, which are cheap and easy to source, ensuring supply chain stability.
Q: Does this reaction require strict anhydrous or oxygen-free conditions?
A: No, one of the major operational advantages of this patent technology is that it proceeds efficiently without the need for rigorous anhydrous or oxygen-free environments, lowering equipment costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of next-generation therapeutics. Our technical team has extensively analyzed emerging synthetic technologies, including the metal-free iodine-promoted route, to ensure we remain at the forefront of process innovation. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you need material for clinical trials or full-scale commercial launch, our capacity is aligned with your needs. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of triazole derivative meets the exacting standards required by global regulatory bodies.
We invite potential partners to engage with us to explore how this advanced synthesis method can be tailored to your specific project requirements. By leveraging our expertise in process optimization, we can help you achieve a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to this metal-free protocol. We encourage you to contact our technical procurement team to request specific COA data for our available triazole intermediates and to discuss route feasibility assessments for your target molecules. Together, we can accelerate your drug development timeline while optimizing your supply chain economics.
