Scalable Metal-Free Synthesis of Trifluoromethyl Triazoles for Advanced Pharmaceutical Intermediates
Scalable Metal-Free Synthesis of Trifluoromethyl Triazoles for Advanced Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust synthetic routes for nitrogen-containing heterocycles, particularly those incorporating fluorine atoms to enhance metabolic stability and bioavailability. Patent CN113105402A introduces a groundbreaking preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds, a structural motif prevalent in high-value drugs such as Maraviroc, Sitagliptin, and Deferasirox. This innovation addresses the critical need for efficient, metal-free methodologies that can be seamlessly integrated into complex API manufacturing pipelines without the burden of heavy metal contamination. By leveraging a unique iodine-promoted cascade reaction in dimethyl sulfoxide (DMSO), this technology offers a streamlined pathway to access diverse trifluoromethylated triazole scaffolds, positioning it as a vital asset for any reliable pharmaceutical intermediate supplier aiming to optimize their portfolio.
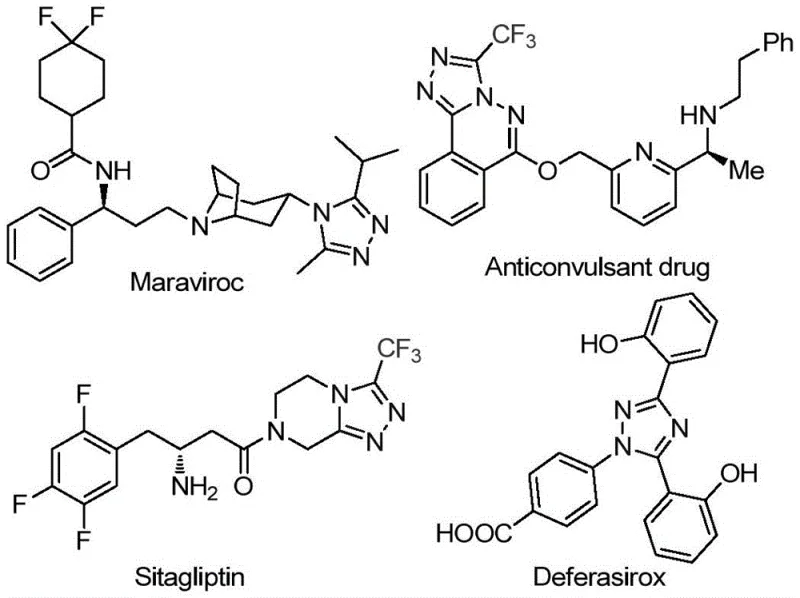
The significance of this patent lies in its ability to bypass traditional limitations associated with triazole synthesis. Historically, constructing the 1,2,4-triazole ring with specific substitution patterns, especially at the 3, 4, and 5 positions simultaneously, has been synthetically challenging. The presence of the trifluoromethyl group further complicates matters due to the electron-withdrawing nature of the fluorine atoms. However, the disclosed method utilizes readily available arylethanones and trifluoroacetimidoyl hydrazides as starting materials. This strategic selection of precursors not only lowers the raw material costs but also expands the chemical space available for medicinal chemists to explore structure-activity relationships (SAR) in drug discovery programs targeting various therapeutic areas.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for polysubstituted 1,2,4-triazoles often rely heavily on transition metal catalysis, such as copper or palladium complexes, which introduce significant downstream processing challenges. The removal of trace heavy metals from the final active pharmaceutical ingredient (API) is a rigorous and costly requirement mandated by regulatory bodies like the FDA and EMA. Furthermore, many existing methods necessitate stringent reaction conditions, including strictly anhydrous and oxygen-free environments, which demand specialized equipment and increase operational complexity. These factors collectively contribute to higher production costs and longer lead times, creating bottlenecks in the supply chain for high-purity pharmaceutical intermediates. Additionally, the tolerance for functional groups in conventional methods is often limited, restricting the diversity of substituents that can be introduced onto the triazole core.
The Novel Approach
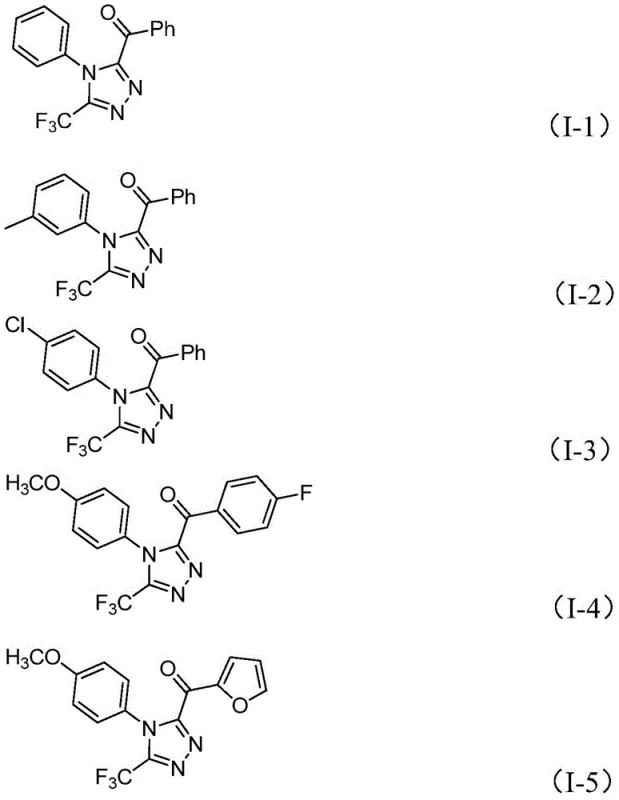
In stark contrast, the method described in CN113105402A employs a non-metallic iodine promotion system that operates effectively under ambient atmospheric conditions. The process initiates with the iodination and subsequent Kornblum oxidation of arylethanones in DMSO to generate reactive alpha-diketone intermediates in situ. These intermediates then undergo a tandem condensation and cyclization with trifluoroacetimidoyl hydrazides. This one-pot strategy eliminates the need for isolating unstable intermediates and avoids the use of toxic heavy metals entirely. The reaction demonstrates excellent functional group tolerance, accommodating various substituents such as halogens, alkoxy groups, and alkyl chains on the aromatic rings. This robustness ensures consistent quality and yield across a broad substrate scope, making it an ideal candidate for cost reduction in API manufacturing where versatility and purity are paramount.
Mechanistic Insights into Iodine-Promoted Cyclization
The core of this synthetic breakthrough involves a sophisticated interplay between iodine, DMSO, and the organic substrates. Initially, elemental iodine acts as an electrophilic promoter, facilitating the alpha-iodination of the arylethanone. In the presence of DMSO, this species undergoes a Kornblum-type oxidation to form an alpha-dicarbonyl compound. This oxidative transformation is crucial as it activates the methyl group of the ketone for subsequent nucleophilic attack. The generated alpha-diketone then reacts with the hydrazide moiety of the trifluoroacetimidoyl hydrazide through a dehydration condensation step, forming a hydrazone intermediate. This step is thermodynamically driven by the release of water and the formation of a stable conjugated system.
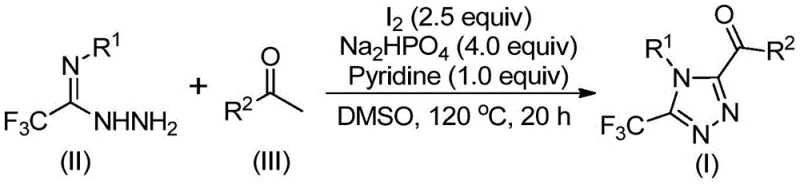
Following the formation of the hydrazone, the reaction proceeds to the final cyclization stage under the combined influence of iodine and the base system (sodium dihydrogen phosphate and pyridine). The base deprotonates the hydrazone nitrogen, enhancing its nucleophilicity, which allows for an intramolecular attack on the adjacent imine carbon. This cyclization closes the five-membered triazole ring, establishing the characteristic 1,2,4-triazole architecture. The presence of the trifluoromethyl group at the 3-position is retained throughout this process, imparting the desired lipophilic and electronic properties to the final molecule. The use of pyridine serves a dual purpose: it acts as a base to neutralize the hydrogen iodide generated during the reaction and as a ligand that may stabilize transient iodine species, thereby maintaining the catalytic cycle efficiency without the need for external transition metals.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
The operational simplicity of this protocol makes it highly attractive for process chemistry teams looking to implement new routes quickly. The procedure involves a sequential addition of reagents in a single vessel, minimizing unit operations and solvent usage. Detailed standard operating procedures regarding stoichiometry, temperature ramping, and work-up protocols are essential for maximizing yield and purity. For R&D professionals seeking to replicate or adapt this chemistry, understanding the precise timing of the second heating stage is critical to ensure complete conversion of the hydrazone intermediate to the final triazole product.
- Mix arylethanone and iodine in DMSO, heating to 90-110°C for 4-6 hours to initiate oxidation.
- Add sodium dihydrogen phosphate, pyridine, and trifluoroethylimine hydrazide to the reaction mixture.
- Heat the solution to 110-130°C for 12-20 hours to complete the cyclization, followed by filtration and chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift towards metal-free synthesis represents a substantial opportunity for cost optimization and risk mitigation. The reliance on elemental iodine, sodium dihydrogen phosphate, and pyridine replaces expensive and supply-constrained transition metal catalysts. These reagents are commodity chemicals with stable global supply chains, ensuring consistent availability and pricing. Moreover, the elimination of heavy metals removes the necessity for specialized scavenging resins or complex extraction processes typically required to meet strict residual metal specifications. This simplification of the purification train directly translates to reduced manufacturing costs and shorter batch cycle times, enhancing the overall economic viability of producing these complex heterocyclic intermediates.
- Cost Reduction in Manufacturing: The absence of precious metal catalysts such as palladium or rhodium drastically lowers the direct material costs associated with the synthesis. Furthermore, the simplified post-reaction work-up, which avoids tedious metal removal steps, reduces the consumption of auxiliary materials like silica gel and specialized chelating agents. The high atom economy of the tandem reaction also means less waste generation per kilogram of product, contributing to lower waste disposal fees and a smaller environmental footprint. These cumulative efficiencies allow for a more competitive pricing structure for the final pharmaceutical intermediates without compromising on quality standards.
- Enhanced Supply Chain Reliability: Utilizing widely available starting materials like arylethanones and hydrazides mitigates the risk of supply disruptions often associated with specialized organometallic reagents. The robustness of the reaction conditions, which do not require inert gas protection or ultra-dry solvents, allows for execution in standard multipurpose reactors found in most chemical manufacturing facilities. This flexibility enables manufacturers to scale production rapidly in response to market demand fluctuations, ensuring a steady flow of critical intermediates to downstream API producers. The ability to source raw materials from multiple vendors further strengthens supply chain resilience against geopolitical or logistical shocks.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to proceed efficiently from gram to multi-gram scales with consistent yields. The use of DMSO, a high-boiling polar aprotic solvent, facilitates high-temperature reactions while allowing for potential solvent recovery and recycling strategies. Since the method avoids toxic heavy metals, the resulting waste streams are easier to treat and dispose of in compliance with increasingly stringent environmental regulations. This alignment with green chemistry principles not only reduces regulatory burdens but also enhances the sustainability profile of the manufacturing process, a key consideration for modern pharmaceutical supply chains aiming for carbon neutrality.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for stakeholders evaluating its adoption. The following questions address common concerns regarding reaction specificity, substrate scope, and practical implementation. These insights are derived directly from the experimental data and mechanistic studies presented in the patent documentation, providing a clear picture of the technology's capabilities and limitations in a real-world manufacturing context.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the patented method utilizes elemental iodine as a promoter instead of toxic or expensive heavy metals like palladium or copper, significantly simplifying downstream purification and reducing heavy metal residue risks in the final API.
Q: What are the critical reaction conditions for high yield?
A: The process requires a two-stage heating protocol: an initial oxidation phase at 90-110°C followed by a cyclization phase at 110-130°C in DMSO, utilizing a specific molar ratio of sodium dihydrogen phosphate to pyridine to optimize the reaction environment.
Q: Is the method suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the reaction does not require stringent anhydrous or oxygen-free conditions and uses cheap, commercially available starting materials, making it highly amenable to commercial scale-up from gram to multi-ton levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free heterocycle synthesis in modern drug development. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN113105402A can be successfully translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 3,4,5-trisubstituted 1,2,4-triazole intermediate meets the exacting standards required by global regulatory authorities. Our commitment to quality assurance ensures that our clients receive materials that are ready for immediate use in subsequent synthetic steps.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced synthetic technology for their pipeline projects. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you accelerate your drug development timeline with reliable, high-quality intermediates produced via efficient and sustainable manufacturing processes.
