Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles: A Breakthrough in Pharmaceutical Intermediate Manufacturing
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing nitrogen-containing heterocycles, particularly those bearing fluorinated motifs which are known to enhance metabolic stability and bioavailability. A significant advancement in this domain is detailed in Chinese Patent CN114920707B, which discloses a highly efficient preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. This patent introduces a novel synthetic strategy that utilizes the ubiquitous organic solvent N,N-dimethylformamide (DMF) not merely as a medium, but as a critical carbon source for the ring construction. The significance of this chemical scaffold cannot be overstated, as the 1,2,4-triazole core is a privileged structure found in numerous bioactive molecules, including prominent drugs like Sitagliptin and various Factor IXa inhibitors. 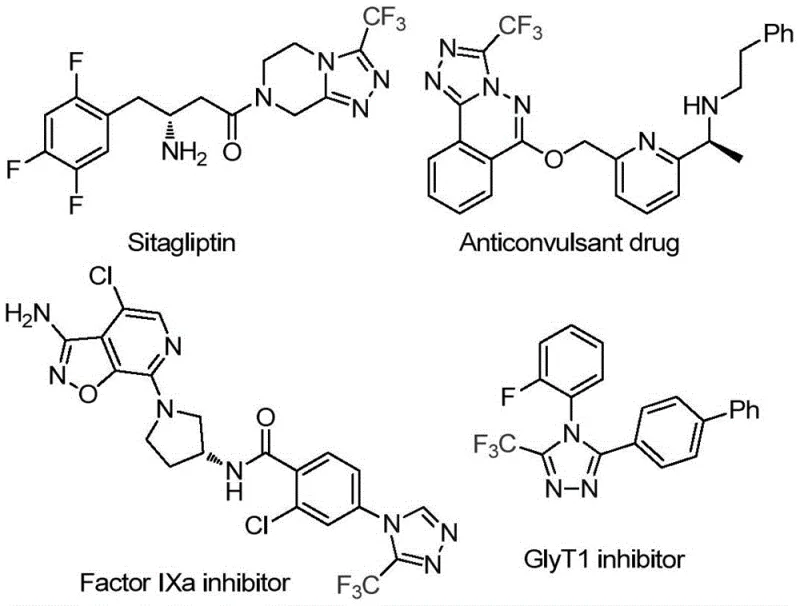 . By leveraging a molecular iodine-promoted tandem cyclization under aerobic conditions, this technology offers a streamlined pathway that addresses many of the logistical and economic pain points associated with traditional heterocyclic synthesis, positioning it as a vital tool for modern process chemistry.
. By leveraging a molecular iodine-promoted tandem cyclization under aerobic conditions, this technology offers a streamlined pathway that addresses many of the logistical and economic pain points associated with traditional heterocyclic synthesis, positioning it as a vital tool for modern process chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has been fraught with synthetic challenges that hinder large-scale production and increase overall manufacturing costs. Conventional routes often rely on the condensation of hydrazides with nitriles or amidines, which frequently necessitate the use of harsh reaction conditions, such as extremely high temperatures or strong acidic/basic environments that can degrade sensitive functional groups. Furthermore, many established protocols require strictly anhydrous and oxygen-free conditions, demanding the use of expensive inert gases like nitrogen or argon and specialized equipment like gloveboxes or Schlenk lines, which drastically increases the capital expenditure and operational complexity for chemical plants. Additionally, traditional methods often employ stoichiometric amounts of expensive coupling reagents or toxic heavy metal catalysts that leave behind difficult-to-remove impurities, complicating the purification process and posing significant environmental disposal challenges. The reliance on specialized C1 sources, distinct from the solvent, further adds to the raw material costs and atom inefficiency of these legacy processes, making them less attractive for the production of commodity pharmaceutical intermediates where margin compression is a constant pressure.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the technology described in patent CN114920707B presents a paradigm shift by utilizing molecular iodine as a promoter and DMF as a dual-purpose solvent and reactant. This approach eliminates the need for external carbon sources or expensive catalysts, as the DMF molecule itself provides the necessary carbon atom for the triazole ring closure through its formyl or methyl group. The reaction proceeds smoothly under an air atmosphere at moderate temperatures ranging from 110°C to 130°C, removing the stringent requirement for inert gas protection and allowing for simpler reactor configurations. 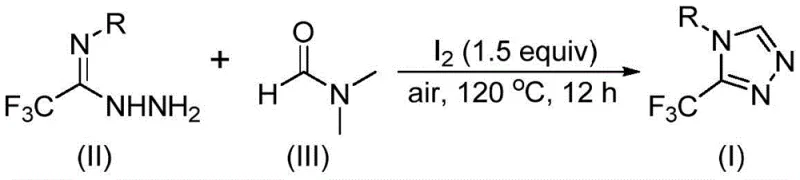 . This simplicity extends to the substrate scope, as the method tolerates a wide array of functional groups on the aromatic ring, including halogens, alkyls, and alkoxy groups, without the need for extensive protecting group strategies. The result is a operationally simple, one-pot procedure that significantly reduces the number of unit operations, minimizes waste generation, and enhances the overall safety profile of the manufacturing process by avoiding pyrophoric reagents.
. This simplicity extends to the substrate scope, as the method tolerates a wide array of functional groups on the aromatic ring, including halogens, alkyls, and alkoxy groups, without the need for extensive protecting group strategies. The result is a operationally simple, one-pot procedure that significantly reduces the number of unit operations, minimizes waste generation, and enhances the overall safety profile of the manufacturing process by avoiding pyrophoric reagents.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The mechanistic elegance of this transformation lies in the activation of the stable DMF molecule by molecular iodine, which facilitates the cleavage of C-N or C-H bonds to generate reactive intermediates capable of cyclizing with the trifluoroethyliminohydrazide. Detailed studies suggest that the reaction can proceed via two potential pathways depending on which part of the DMF molecule acts as the carbon synthon. In one pathway, the formyl group of DMF undergoes a condensation reaction with the hydrazide to form a hydrazone intermediate, which subsequently experiences intramolecular cyclization accompanied by the elimination of dimethylamine to yield the target triazole. Alternatively, the N-methyl group of DMF can be activated by iodine to form an amine salt, which then participates in a nucleophilic addition with the hydrazide. This pathway involves the elimination of N-methylformamide to generate an azadiene intermediate, followed by intramolecular nucleophilic attack and oxidative aromatization to finalize the heterocyclic core.  . The presence of iodine is crucial not only for activating the solvent but also for driving the oxidative aromatization step, ensuring the formation of the fully conjugated triazole system. This dual-pathway capability underscores the robustness of the method, allowing it to accommodate slight variations in reaction conditions while maintaining high efficiency and selectivity for the desired 3-trifluoromethyl product.
. The presence of iodine is crucial not only for activating the solvent but also for driving the oxidative aromatization step, ensuring the formation of the fully conjugated triazole system. This dual-pathway capability underscores the robustness of the method, allowing it to accommodate slight variations in reaction conditions while maintaining high efficiency and selectivity for the desired 3-trifluoromethyl product.
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed alternatives. Since the promoter is molecular iodine, which is volatile and easily removed during workup, the risk of heavy metal contamination in the final Active Pharmaceutical Ingredient (API) is virtually non-existent. The primary byproducts, such as dimethylamine or N-methylformamide, are small, polar molecules that are readily separated from the hydrophobic triazole product during aqueous workup or silica gel chromatography. The reaction's tolerance to air suggests that radical species may play a role in the oxidative steps, but the controlled nature of the iodine mediation prevents uncontrolled polymerization or degradation of the sensitive trifluoromethyl group. This clean reaction profile translates directly to a simplified purification train, reducing the need for multiple recrystallizations or complex preparative HPLC steps that often bottleneck production timelines in fine chemical manufacturing.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical implementation of this synthesis is designed for ease of execution in standard laboratory and pilot plant settings. The protocol involves charging a reaction vessel with the trifluoroethyliminohydrazide precursor, molecular iodine, and DMF, followed by heating under open air conditions. The molar ratio of hydrazide to iodine is typically optimized around 1:1.5 to ensure complete conversion without excessive reagent waste. Reaction times generally span 10 to 15 hours at temperatures between 110°C and 130°C, providing a balance between reaction rate and energy consumption. Upon completion, the mixture is cooled, and the product is isolated through filtration and standard chromatographic techniques.
- Combine molecular iodine, trifluoroethyliminohydrazide, and organic solvent DMF in a reaction vessel under air atmosphere.
- Heat the reaction mixture to a temperature range of 110-130°C and maintain stirring for 10-15 hours to ensure complete conversion.
- Upon completion, perform standard post-treatment including filtration, washing, and column chromatography to isolate the pure triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this DMF-based cyclization technology represents a strategic opportunity to optimize the cost structure and reliability of the supply chain for trifluoromethyl-triazole intermediates. The most immediate impact is seen in raw material costs, as the process utilizes DMF, a commodity chemical available in bulk quantities at very low prices, effectively replacing expensive specialized C1 building blocks. This substitution leads to substantial cost savings in pharmaceutical intermediate manufacturing by reducing the Bill of Materials (BOM) cost per kilogram of the final product. Furthermore, the elimination of inert gas requirements and the ability to run the reaction under air atmosphere significantly lowers the utility costs and infrastructure investment needed for production facilities, as there is no longer a dependency on continuous nitrogen purging or specialized anaerobic reactors.
- Cost Reduction in Manufacturing: The economic benefits extend beyond just raw materials; the simplified workup procedure reduces the consumption of solvents and silica gel during purification, directly lowering the variable costs associated with each batch. By avoiding precious metal catalysts, the process also eliminates the costly and time-consuming steps required for metal scavenging and residual metal testing, which are mandatory for GMP compliance in API production. The high atom economy of using the solvent as a reactant means less chemical waste is generated per unit of product, which in turn reduces the expenses related to hazardous waste disposal and environmental compliance fees. These cumulative efficiencies create a leaner manufacturing process that is more resilient to fluctuations in the pricing of specialty reagents.
- Enhanced Supply Chain Reliability: From a supply chain continuity perspective, the reliance on widely available, commodity-grade starting materials like DMF and iodine mitigates the risk of supply disruptions that often plague processes dependent on niche, single-source reagents. The robustness of the reaction conditions, specifically the tolerance to moisture and oxygen, allows for greater flexibility in manufacturing scheduling and reduces the likelihood of batch failures due to minor deviations in environmental controls. This reliability ensures consistent lead times for customers, enabling pharmaceutical companies to maintain tighter inventory controls and reduce the need for large safety stocks of critical intermediates. The scalability of the method from gram to multi-kilogram scales without significant re-optimization further supports a agile supply chain capable of responding quickly to changes in market demand.
- Scalability and Environmental Compliance: The environmental profile of this synthesis aligns well with modern green chemistry principles, which is increasingly becoming a criterion for supplier selection by major multinational corporations. The use of DMF, while requiring careful handling, is well-understood in industrial hygiene, and the absence of heavy metals simplifies the effluent treatment process. The reaction's high efficiency and selectivity minimize the formation of complex byproduct mixtures, making the waste stream easier to treat and dispose of in accordance with strict environmental regulations. As regulatory pressures on pharmaceutical manufacturing continue to intensify, adopting a cleaner, metal-free synthesis route provides a competitive advantage by future-proofing the supply chain against evolving environmental standards and reducing the carbon footprint associated with the production of these valuable heterocyclic building blocks.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These answers are derived directly from the experimental data and claims within patent CN114920707B, providing clarity on reaction parameters and substrate compatibility. Understanding these details is crucial for process chemists evaluating the feasibility of this route for their specific project needs.
Q: What is the primary advantage of using DMF in this synthesis?
A: DMF serves a dual function as both the reaction solvent and the carbon source (C1 synthon), eliminating the need for separate, expensive formylating agents and simplifying the reaction setup significantly.
Q: Does this reaction require inert gas protection?
A: No, unlike many traditional heterocyclic syntheses that demand strict anhydrous and anaerobic conditions, this iodine-promoted method proceeds efficiently under ambient air atmosphere, reducing operational complexity.
Q: What is the substrate scope for the R group in the hydrazide?
A: The method demonstrates broad compatibility, successfully accommodating various substituted aryl groups including those with electron-donating (methyl, methoxy) and electron-withdrawing (fluoro, chloro, trifluoromethyl) substituents at ortho, meta, and para positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of new therapeutic agents. Our team of expert process chemists has extensively evaluated the methodology described in CN114920707B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this efficient synthesis to an industrial level. We are committed to delivering high-purity 3-trifluoromethyl-1,2,4-triazole derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to ensure every batch complies with global regulatory standards. Our facility is designed to handle fluorinated chemistry safely and efficiently, ensuring that the unique challenges of handling trifluoromethyl precursors are managed with the highest degree of professional care and technical expertise.
We invite R&D directors and procurement specialists to collaborate with us to leverage this advanced synthetic technology for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for custom synthesis projects. Let us help you accelerate your drug development timeline with reliable, cost-effective, and scalable chemical solutions.
