Advanced Molybdenum-Copper Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial Scale-Up
Advanced Molybdenum-Copper Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing nitrogen-containing heterocycles, particularly those incorporating fluorine motifs which are pivotal for enhancing metabolic stability and bioavailability. A significant breakthrough in this domain is detailed in Chinese Patent CN113307778A, which discloses a novel preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. This technology leverages a synergistic molybdenum and copper co-catalytic system to facilitate the cycloaddition of trifluoroethylimidoyl chloride with functionalized isonitriles. The significance of this development cannot be overstated, as 1,2,4-triazole scaffolds are ubiquitous in high-value active pharmaceutical ingredients (APIs), including blockbuster drugs like Sitagliptin, as well as in agrochemical and ligand chemistry applications. By enabling the efficient construction of these complex heterocycles under relatively mild conditions, this patent offers a transformative pathway for reliable pharmaceutical intermediate supplier networks aiming to optimize their synthetic portfolios.
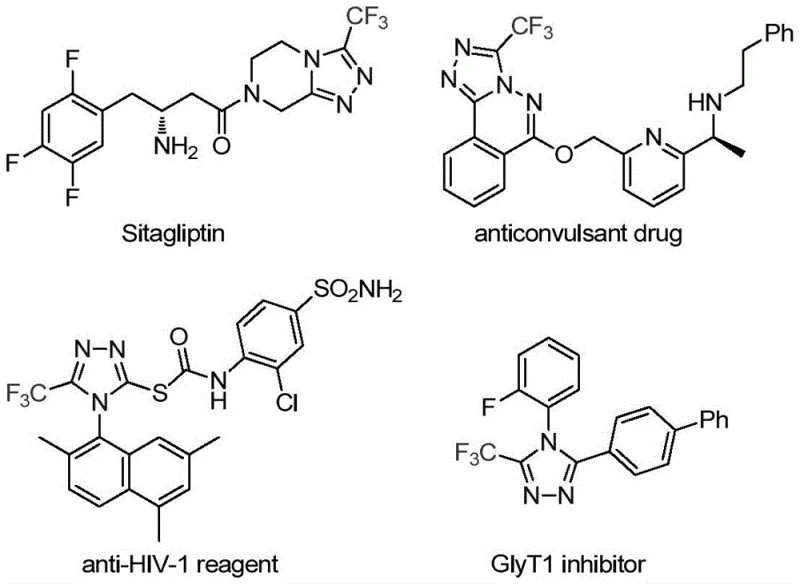
The strategic value of this synthesis lies in its ability to introduce the trifluoromethyl group directly during the ring formation, thereby streamlining what was traditionally a multi-step, labor-intensive process. For R&D directors and process chemists, the ability to access diverse 3,4-disubstituted 1,2,4-triazoles through a single, tunable protocol represents a major advancement in library synthesis and lead optimization. Furthermore, the use of commercially available starting materials and standard laboratory equipment lowers the barrier to entry for adoption, making it an attractive candidate for cost reduction in API manufacturing. As we delve deeper into the technical specifics, it becomes clear that this methodology addresses several critical pain points associated with conventional heterocycle synthesis, offering a cleaner, more efficient route that aligns with modern green chemistry principles and supply chain reliability requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted 1,2,4-triazoles has been fraught with challenges that hinder efficient commercial production. Traditional literature methods often rely on the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones. These classical routes frequently suffer from harsh reaction conditions, requiring extreme temperatures or highly reactive reagents that pose safety risks and complicate process control. Additionally, alternative approaches involving copper-catalyzed multi-component reactions with diazonium salts and trifluorodiazoethane introduce significant hazards due to the explosive nature of diazo compounds, necessitating specialized containment and handling protocols that drive up operational costs. Another common pathway involves the cyclization of trifluoroethylimidoyl chloride with aldehyde hydrazones and hydrazides, which often results in poor atom economy and generates substantial amounts of difficult-to-remove byproducts. These legacy methods typically lack the versatility required to easily modify the substitution pattern on the triazole ring, limiting their utility in the rapid synthesis of diverse analog libraries needed for drug discovery.
The Novel Approach
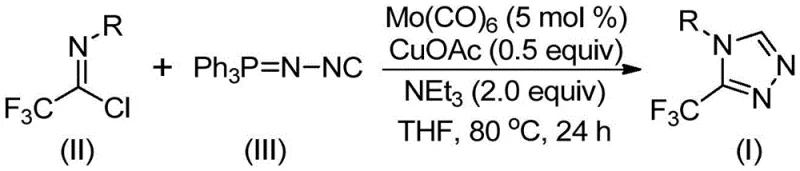
In stark contrast to these cumbersome traditional techniques, the method disclosed in CN113307778A introduces a streamlined, one-pot cycloaddition strategy that fundamentally reshapes the synthetic landscape for these valuable intermediates. By utilizing trifluoroethylimidoyl chloride and functionalized isonitrile (NIITP) as the primary building blocks, this novel approach bypasses the need for hazardous diazo reagents or unstable hydrazine derivatives. The core innovation lies in the dual-catalyst system comprising molybdenum hexacarbonyl and cuprous acetate, which activates the isonitrile and promotes the [3+2] cycloaddition with remarkable efficiency. This methodology operates under significantly milder conditions, typically between 70°C and 90°C, which not only enhances safety but also improves the thermal stability of sensitive functional groups on the substrate. The reaction demonstrates exceptional tolerance for a wide range of substituents, including electron-donating and electron-withdrawing groups on the aryl ring, allowing for the precise design of molecular architectures tailored to specific biological targets. This shift from hazardous, multi-step sequences to a direct, catalytic assembly represents a paradigm shift in how these critical heterocyclic cores are manufactured.
Mechanistic Insights into Mo-Cu Co-Catalyzed Cycloaddition
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams looking to implement this technology at scale. The reaction mechanism is believed to initiate with the coordination of molybdenum hexacarbonyl to the functionalized isonitrile, forming a reactive metal-isocyanide complex that lowers the activation energy for subsequent bond formation. Simultaneously, the cuprous acetate acts as a Lewis acid promoter, facilitating the nucleophilic attack of the activated isonitrile species onto the electrophilic carbon of the trifluoroethylimidoyl chloride. This cooperative catalysis drives a [3+2] cycloaddition event, constructing the five-membered triazole ring intermediate with high regioselectivity. Following the ring closure, the system undergoes a hydrolysis or elimination step, potentially aided by trace water or the basic environment provided by triethylamine, to remove the triphenylphosphine oxide moiety and yield the final 3-trifluoromethyl-substituted 1,2,4-triazole product. This intricate dance between the molybdenum activator and the copper catalyst ensures that the reaction proceeds smoothly without the accumulation of toxic intermediates, providing a clean reaction profile that simplifies downstream processing.
From an impurity control perspective, the mildness of this catalytic system offers distinct advantages over thermal cyclization methods. High-temperature processes often lead to decomposition of the trifluoromethyl group or polymerization of the isonitrile, generating complex impurity profiles that are difficult to separate. In this Mo-Cu catalyzed protocol, the moderate temperature range of 70-90°C minimizes thermal degradation pathways, resulting in a crude reaction mixture with higher purity. The use of triethylamine as a base not only neutralizes the HCl byproduct generated during the reaction but also helps maintain the catalyst in its active oxidation state, preventing the formation of inactive metal precipitates that could contaminate the product. Furthermore, the choice of tetrahydrofuran (THF) as the preferred solvent ensures excellent solubility for both the organic substrates and the metal catalysts, promoting homogeneous reaction kinetics that reduce the likelihood of localized hot spots and side reactions. This level of control over the reaction environment is essential for producing high-purity pharmaceutical intermediates that meet stringent regulatory specifications.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and reaction parameters to maximize yield and reproducibility. The patent outlines a robust protocol where the molar ratio of trifluoroethylimidoyl chloride to functionalized isonitrile is optimized, typically using a slight excess of the isonitrile to drive the reaction to completion. The catalyst loading is kept relatively low, with molybdenum hexacarbonyl used at approximately 5 mol% and cuprous acetate at 0.5 equivalents, balancing cost efficiency with catalytic activity. The reaction is conducted in anhydrous THF under an inert atmosphere to prevent moisture sensitivity issues, although the system shows some tolerance to water during the workup phase. Detailed standardized synthesis steps see the guide below.
- Combine molybdenum hexacarbonyl (5 mol %), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), and molecular sieves in an organic solvent such as THF.
- Add trifluoroethylimidoyl chloride and functionalized isonitrile (NIITP) to the reaction mixture under inert atmosphere.
- Heat the reaction mixture to 70-90°C for 18-30 hours, then filter and purify via column chromatography to isolate the target triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Mo-Cu catalyzed synthesis offers compelling economic and logistical benefits that extend beyond mere chemical elegance. The primary advantage lies in the significant cost reduction in manufacturing driven by the use of inexpensive, commodity-grade reagents. Unlike methods requiring exotic ligands or precious metal catalysts like palladium or rhodium, this process utilizes copper and molybdenum salts which are abundant and cost-effective, drastically lowering the raw material cost per kilogram of product. Additionally, the elimination of hazardous diazo compounds removes the need for expensive safety infrastructure and specialized waste disposal protocols, further reducing the total cost of ownership. The simplicity of the workup procedure, which involves basic filtration and column chromatography, means that production cycles are shorter and labor requirements are minimized, contributing to overall operational efficiency.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the accessibility of its starting materials. Trifluoroethylimidoyl chloride and functionalized isonitriles are readily available from global chemical suppliers, ensuring a stable supply chain that is not vulnerable to the bottlenecks often seen with custom-synthesized precursors. The high atom economy of the cycloaddition reaction means that a larger proportion of the input mass is converted into the desired product, reducing waste disposal costs and maximizing yield. Furthermore, the ability to run the reaction at moderate temperatures reduces energy consumption compared to high-temperature reflux methods, aligning with sustainability goals and lowering utility expenses. These factors combine to create a highly competitive cost structure for producing 3-trifluoromethyl-1,2,4-triazoles, making it an ideal choice for large-volume commercial production.
- Enhanced Supply Chain Reliability: Supply chain resilience is critical for maintaining continuous API production, and this methodology supports that goal through its reliance on stable, non-hazardous reagents. The avoidance of explosive diazonium salts or unstable hydrazines eliminates the risk of shipment delays due to hazardous material transport regulations. The robustness of the reaction conditions allows for flexibility in manufacturing locations, as it does not require highly specialized reactor setups or extreme pressure capabilities. This decentralization potential enables companies to diversify their manufacturing base, reducing the risk of supply disruptions caused by regional instabilities or single-source dependencies. Moreover, the scalability of the process from gram to kilogram levels ensures that supply can be ramped up quickly to meet market demand without the need for extensive process re-engineering.
- Scalability and Environmental Compliance: As regulatory scrutiny on chemical manufacturing intensifies, the environmental profile of a synthesis route becomes a key decision factor. This Mo-Cu catalyzed method generates minimal hazardous waste, primarily consisting of benign metal salts and organic solvents that can be recycled or treated using standard protocols. The absence of heavy metal contaminants like palladium in the final product simplifies the purification process and ensures compliance with strict limits on residual metals in pharmaceuticals. The use of THF, a common solvent with established recovery systems, further facilitates solvent recycling, reducing the environmental footprint of the operation. These attributes make the process not only scalable but also sustainable, positioning it favorably for long-term commercial adoption in an increasingly regulated global market.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology, we have compiled answers to common questions regarding the reaction scope, optimization, and practical implementation. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a realistic view of what can be achieved with this methodology. Understanding these nuances is vital for project planning and risk assessment when integrating new synthetic routes into existing workflows.
Q: What are the optimal reaction conditions for this triazole synthesis?
A: The patent specifies reacting at 70-90°C for 18-30 hours in THF solvent, utilizing Mo(CO)6 and CuOAc as co-catalysts with triethylamine as the base.
Q: Can this method tolerate diverse functional groups on the substrate?
A: Yes, the method demonstrates high substrate tolerance, successfully synthesizing derivatives with methyl, methoxy, fluoro, chloro, and nitro substituents on the aryl ring.
Q: Is this process suitable for large-scale manufacturing?
A: The patent explicitly states the method can be expanded to gram-level reactions with simple post-treatment, indicating strong potential for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
The technological advancements described in CN113307778A highlight the immense potential of 3-trifluoromethyl-1,2,4-triazoles as versatile building blocks for next-generation therapeutics and agrochemicals. At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating such innovative academic and patent research into reliable commercial reality. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of Mo-Cu catalyzed reactions, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest industry standards. We are committed to delivering high-purity pharmaceutical intermediates that empower your R&D and production teams to succeed.
We invite you to explore the possibilities of this advanced synthesis route for your upcoming projects. Whether you require custom synthesis of specific triazole derivatives or bulk supply of standard intermediates, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume and quality needs. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise can optimize your supply chain and accelerate your time to market. Let us be your partner in turning complex chemical challenges into commercial opportunities.
