Advanced Rhodium-Catalyzed Synthesis of Polysubstituted Isoindoles for Commercial Pharmaceutical Applications
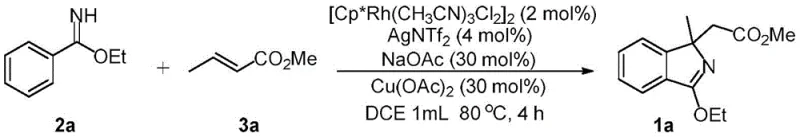
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access complex nitrogen-containing heterocycles, which serve as critical scaffolds for bioactive molecules. Patent CN109879792B introduces a groundbreaking methodology for the preparation of polysubstituted isoindole compounds, addressing significant limitations in existing synthetic routes. This innovation leverages a trivalent rhodium-catalyzed oxidative Heck reaction, enabling the direct construction of isoindole skeletons from readily available imidates and olefin esters. By overcoming the technical bottlenecks of low bonding diversity and limited product types associated with prior art, this technology offers a robust platform for generating high-value intermediates. For R&D directors and procurement specialists, understanding this shift towards catalytic C-H activation is essential for securing a competitive edge in the supply of reliable pharmaceutical intermediates supplier networks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic strategies for constructing the isoindole core often rely on multi-step sequences involving harsh reaction conditions that are increasingly untenable for modern green chemistry standards. Classical approaches may utilize tetralin-imines requiring extreme thermal cycling or dibromobenzenes necessitating complex alkylation and high-temperature cyclization steps. These legacy methods frequently suffer from poor atom economy, generating substantial quantities of hazardous waste and by-products that complicate downstream purification. Furthermore, the introduction of quaternary carbon centers, which are vital for enhancing the metabolic stability of drug candidates, remains exceptionally challenging using conventional nucleophilic substitution or Grignard-type reactions. The rigidity of these old protocols limits the structural diversity accessible to medicinal chemists, thereby constraining the exploration of novel chemical space for drug discovery programs.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a transition metal-catalyzed C-H bond activation strategy that dramatically streamlines the synthesis process. By employing a trivalent rhodium catalyst system, the reaction directly couples imidates with olefin esters through an oxidative Heck manifold, effectively bypassing the need for pre-functionalized halide substrates in many instances. This approach not only shortens the synthetic route but also enhances the overall efficiency by operating under relatively mild thermal conditions in the presence of air. The versatility of this system allows for the modular assembly of diverse polysubstituted isoindoles, including those bearing sensitive functional groups that would decompose under traditional harsh conditions. This represents a paradigm shift in cost reduction in pharmaceutical intermediates manufacturing by minimizing unit operations and maximizing yield potential.

Mechanistic Insights into Rh(III)-Catalyzed C-H Activation and Cyclization
The core of this technological advancement lies in the precise orchestration of the rhodium catalytic cycle, which facilitates the selective activation of inert carbon-hydrogen bonds. The trivalent rhodium species, typically generated from dimers like [Cp*RhCl2]2 in the presence of silver salts, coordinates with the nitrogen directing group of the imidate substrate. This coordination directs the metal center to the ortho-position of the aromatic ring, enabling the cleavage of the C-H bond and the formation of a stable rhodacycle intermediate. Subsequent migratory insertion of the olefin ester into the rhodium-carbon bond sets the stage for the construction of the new carbon-carbon bond essential for the isoindole framework. The presence of copper oxidants and silver additives plays a critical role in regenerating the active Rh(III) species from the reduced Rh(I) state, ensuring the catalytic turnover continues efficiently throughout the reaction duration.
Controlling impurity profiles is paramount for any process intended for pharmaceutical applications, and this catalytic system exhibits remarkable chemical and regioselectivity. The mechanism inherently favors the formation of the desired isoindole ring over potential side reactions such as homocoupling of the olefin or non-selective arylation. The use of specific additives like sodium acetate helps to buffer the reaction environment and facilitate the proton transfer steps required during the catalytic cycle. Moreover, the tolerance towards various substituents on the aromatic ring, including electron-withdrawing nitro groups and electron-donating alkyl chains, ensures that the impurity spectrum remains manageable. This high level of control translates directly into simplified purification processes, which is a key factor in reducing lead time for high-purity isoindole intermediates during the scale-up phase.
How to Synthesize Polysubstituted Isoindoles Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry of the catalyst system and the choice of solvent to ensure optimal conversion rates. The standard protocol involves dissolving the imidate and olefin substrates in an inert solvent such as 1,2-dichloroethane, followed by the addition of the rhodium catalyst, silver salt, copper oxidant, and base. The reaction is typically conducted under an air atmosphere at temperatures ranging from 80°C to 120°C, allowing for the seamless formation of the heterocyclic core within a few hours. Detailed standardized synthesis steps see the guide below for specific molar ratios and workup procedures tailored to different substrate combinations.
- Dissolve imidate and olefin ester substrates in an inert solvent such as 1,2-dichloroethane under air atmosphere.
- Add trivalent rhodium catalyst, silver additive, copper oxidant, and sodium acetate base to the reaction mixture.
- Heat the mixture to 80°C-120°C for 4-6 hours, then purify the crude product via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology offers tangible benefits that extend beyond mere chemical elegance. The simplification of the synthetic route directly correlates with a reduction in raw material consumption and energy usage, driving significant operational efficiencies. By eliminating the need for pre-halogenated starting materials in many cases, the supply chain becomes less dependent on volatile commodity markets for specialized halides. This resilience is crucial for maintaining continuity of supply in the face of global logistical disruptions, ensuring that production schedules remain intact without unexpected delays caused by raw material shortages.
- Cost Reduction in Manufacturing: The streamlined one-pot nature of this reaction eliminates multiple isolation and purification steps that are traditionally required in multi-step syntheses. Removing these unit operations significantly lowers labor costs and reduces the consumption of solvents and chromatography media, leading to substantial cost savings. Furthermore, the high atom economy of the oxidative Heck reaction means that a greater proportion of the starting mass is incorporated into the final product, minimizing waste disposal fees. The avoidance of expensive palladium catalysts in favor of rhodium systems, which can be optimized for loading, also contributes to a more favorable cost structure for large-scale production runs.
- Enhanced Supply Chain Reliability: The substrates required for this transformation, such as simple imidates and acrylates, are commercially available in bulk quantities from multiple global suppliers. This commoditization of starting materials reduces the risk of single-source dependency, allowing procurement teams to negotiate better terms and secure inventory more easily. The robustness of the reaction conditions, which tolerate air and moisture better than many organometallic processes, further decreases the likelihood of batch failures due to environmental factors. Consequently, manufacturers can promise more reliable delivery timelines to their downstream clients, strengthening long-term partnerships.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram levels is facilitated by the homogeneous nature of the catalytic system and the absence of highly pyrophoric reagents. The reduced generation of hazardous by-products aligns with increasingly stringent environmental regulations, lowering the compliance burden on manufacturing facilities. Efficient waste management is achieved through the minimized use of heavy metals and toxic solvents, supporting corporate sustainability goals. This environmental compatibility ensures that the production of high-purity isoindole derivatives can be expanded without encountering regulatory roadblocks, securing the long-term viability of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on performance and applicability. Understanding these details helps stakeholders make informed decisions about integrating this chemistry into their existing portfolios.
Q: What are the advantages of this Rh-catalyzed method over traditional isoindole synthesis?
A: Unlike conventional methods requiring harsh conditions or multiple steps, this oxidative Heck reaction operates under milder conditions with high atom economy and excellent regioselectivity, significantly simplifying the production workflow.
Q: Does this process tolerate diverse functional groups on the substrate?
A: Yes, the protocol demonstrates broad substrate scope, successfully accommodating halogens, nitro groups, and various ester functionalities, which is crucial for synthesizing complex pharmaceutical intermediates.
Q: Is this method suitable for large-scale manufacturing?
A: The one-pot nature and use of readily available reagents suggest strong potential for scalability, reducing operational complexity and facilitating commercial scale-up of complex heterocyclic compounds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Isoindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the Rh-catalyzed oxidative Heck reaction for the future of pharmaceutical manufacturing. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative lab-scale discoveries are successfully translated into industrial reality. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the exacting standards required for clinical and commercial applications. We understand that consistency is key, and our infrastructure is designed to support the continuous supply of complex intermediates without compromise.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Our technical team is prepared to provide a Customized Cost-Saving Analysis that evaluates how this route can optimize your current manufacturing expenses. Please contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your target molecules. Together, we can accelerate your development timelines and secure a competitive advantage in the global market through superior chemical innovation and supply chain reliability.
