Advanced Rhodium-Catalyzed Synthesis of Polysubstituted Isoindole Intermediates for Commercial Scale-up
The landscape of organic synthesis for nitrogen-containing heterocycles is continually evolving, driven by the demand for more efficient and environmentally benign processes. A significant advancement in this domain is detailed in patent CN109879792B, which discloses a novel preparation method for polysubstituted isoindole compounds. These structures are pivotal scaffolds found in numerous biologically active natural products, such as staurosporines, and high-performance organic pigments. The core innovation lies in a trivalent rhodium-catalyzed tandem reaction that merges C-H bond activation with an oxidative Heck coupling. This approach circumvents the limitations of traditional multi-step syntheses, offering a streamlined pathway to complex molecular architectures that include challenging quaternary carbon centers. For research and development teams seeking robust routes to high-value intermediates, this technology represents a substantial leap forward in synthetic efficiency.
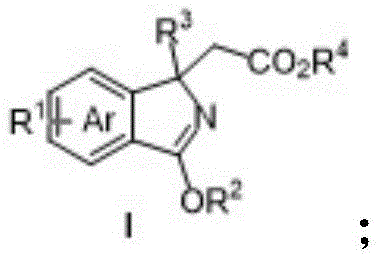
The structural versatility of the resulting isoindole derivatives, as depicted in the general formula, allows for extensive modular customization. The ability to introduce diverse substituents at the R1 position, ranging from halogens and nitro groups to ester and carbonyl functionalities, provides a powerful platform for medicinal chemistry optimization. Furthermore, the presence of ester and alkoxy groups on the isoindole core offers convenient handles for downstream derivatization, enabling the rapid construction of even more complex fused-ring systems. This level of structural diversity is critical for pharmaceutical companies aiming to explore broad chemical space during lead optimization phases without being constrained by synthetic accessibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the isoindole skeleton has relied on methodologies that are often fraught with operational complexities and environmental drawbacks. Classical approaches frequently involve the use of 1,2,3,4-tetralin-1,4-imine precursors which require extreme thermal conditions followed by cryogenic cooling with liquid nitrogen, creating significant energy burdens and safety hazards. Alternative routes utilizing 1,2-dibromobenzene or o-phthalaldehyde often suffer from poor atom economy and generate substantial amounts of toxic by-products that complicate waste management. Moreover, these traditional pathways struggle significantly when attempting to install quaternary carbon centers, a structural feature highly prized in drug design for its metabolic stability and conformational rigidity. The reliance on pre-functionalized halides in many cross-coupling strategies also adds cost and step-count, reducing the overall viability for large-scale manufacturing.
The Novel Approach
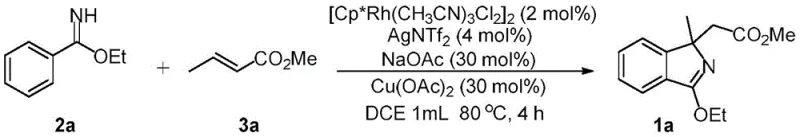
In stark contrast, the methodology described in the patent utilizes a direct oxidative Heck reaction between readily available imidates and olefin esters. This transformative process operates under mild conditions, typically at 80°C in common solvents like 1,2-dichloroethane, and proceeds efficiently under an air atmosphere. By leveraging the power of rhodium catalysis, the reaction achieves direct C-H functionalization, thereby eliminating the need for pre-installed leaving groups on the aromatic ring. This not only simplifies the starting material supply chain but also drastically reduces the generation of salt waste associated with halide displacement. The tandem nature of the reaction builds the heterocyclic ring and installs the side chain in a single operation, showcasing a level of convergence that is rarely achieved in classical heterocycle synthesis.

Mechanistic Insights into Rhodium-Catalyzed C-H Activation and Oxidative Cyclization
The success of this transformation hinges on the precise orchestration of a rhodium catalytic cycle that facilitates sequential C-H activation and migratory insertion. The cycle initiates with the coordination of the trivalent rhodium species to the nitrogen atom of the imidate directing group, which positions the metal center in proximity to the ortho-C-H bond. Following concerted metalation-deprotonation assisted by the acetate base, a stable rhodacycle intermediate is formed. This key species then undergoes migratory insertion with the electron-deficient olefin, establishing the new carbon-carbon bond that will eventually become part of the quaternary center. The subsequent steps involve intramolecular nucleophilic attack and beta-hydride elimination or oxidative reductive elimination pathways, facilitated by the copper oxidant which regenerates the active Rh(III) species. Understanding this mechanism is vital for process chemists to fine-tune reaction parameters for maximum turnover.
From an impurity control perspective, the high regioselectivity inherent in the directed C-H activation mechanism ensures that substitution occurs exclusively at the desired position ortho to the imidate group. This intrinsic selectivity minimizes the formation of regioisomers, which are often difficult to separate and can compromise the purity profile of the final active pharmaceutical ingredient. Furthermore, the compatibility of the catalyst system with various functional groups means that protecting group strategies can often be minimized, reducing the number of unit operations and potential points of failure in the synthesis. The use of copper acetate as a terminal oxidant is particularly advantageous as it is inexpensive and generates manageable by-products compared to stoichiometric amounts of precious metal oxidants often seen in similar transformations.
How to Synthesize Polysubstituted Isoindole Derivatives Efficiently
Implementing this synthesis requires careful attention to the stoichiometry of the oxidant and the choice of the rhodium precursor to ensure high conversion rates. The standard protocol involves combining the imidate and olefin substrates in a Schlenk tube with the catalyst system, ensuring an inert environment is not strictly necessary as the reaction tolerates air. Detailed operational procedures, including specific workup techniques to remove metal residues, are critical for meeting the stringent purity requirements of the pharmaceutical industry. The following guide outlines the generalized steps derived from the patent examples to assist process development teams in replicating this high-efficiency transformation.
- Dissolve the imidate substrate and olefin ester in an inert solvent such as 1,2-dichloroethane within a Schlenk reaction tube under air atmosphere.
- Add the trivalent rhodium catalyst precursor, silver additive, copper oxidant, and sodium acetate base to the reaction mixture.
- Heat the reaction mixture to 80°C for 4 hours, then cool, filter through celite, and purify the crude product via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this rhodium-catalyzed technology offers compelling economic and logistical benefits that extend beyond simple yield improvements. The shift towards a direct C-H functionalization strategy fundamentally alters the raw material landscape, allowing manufacturers to source simpler, commodity-grade starting materials rather than expensive, pre-functionalized building blocks. This simplification of the bill of materials directly contributes to cost reduction in fine chemical manufacturing by lowering the entry price for key intermediates. Additionally, the reduction in synthetic steps translates to shorter production cycles, which enhances the responsiveness of the supply chain to fluctuating market demands and reduces the working capital tied up in work-in-progress inventory.
- Cost Reduction in Manufacturing: The elimination of multiple synthetic steps and the avoidance of cryogenic conditions significantly lower the operational expenditure associated with production. By removing the need for specialized low-temperature equipment and reducing the consumption of energy-intensive reagents, the overall cost of goods sold is substantially decreased. Furthermore, the high atom economy of the oxidative Heck reaction ensures that a greater proportion of the raw material mass is incorporated into the final product, minimizing waste disposal costs and maximizing material efficiency.
- Enhanced Supply Chain Reliability: The reliance on broadly available imidates and acrylate esters mitigates the risk of supply disruptions often associated with niche, custom-synthesized intermediates. Since the reaction tolerates a wide range of functional groups, suppliers can maintain a flexible inventory of common substrates that can be rapidly converted into diverse isoindole derivatives as needed. This modularity ensures a continuous supply of critical intermediates, safeguarding downstream production schedules against raw material shortages and enhancing overall supply chain resilience.
- Scalability and Environmental Compliance: The use of standard solvents like 1,2-dichloroethane and the operation at moderate temperatures make this process highly amenable to scale-up in existing multipurpose reactors. The simplified workup procedure, which involves basic filtration and chromatography, reduces the complexity of purification trains and lowers the burden on wastewater treatment facilities. This alignment with green chemistry principles not only reduces the environmental footprint but also ensures compliance with increasingly stringent regulatory standards regarding industrial emissions and waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These answers are derived directly from the experimental data and specifications provided in the patent documentation to ensure accuracy and relevance for technical decision-makers. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into your current manufacturing portfolio.
Q: What represents the primary technical advantage of this Rhodium-catalyzed method over classical isoindole synthesis?
A: Unlike classical methods requiring harsh conditions like liquid nitrogen cooling or high-temperature heating of tetralin-imines, this method utilizes a mild one-pot oxidative Heck reaction at 80°C. It effectively constructs quaternary carbon centers which are traditionally difficult to access, offering superior atom economy and operational simplicity.
Q: Which catalyst system is preferred for this transformation according to the patent data?
A: The patent specifies the use of trivalent rhodium catalysts, particularly pentamethylcyclopentadienyl rhodium chloride dimers such as [Cp*RhCl2]2 or [Cp*Rh(CH3CN)3Cl2]2. These are used in conjunction with silver additives like AgNTf2 and copper oxidants like Cu(OAc)2 to facilitate the C-H activation cycle.
Q: Is this synthetic route compatible with sensitive functional groups on the aromatic ring?
A: Yes, the methodology demonstrates excellent functional group tolerance. The patent examples confirm successful synthesis with substrates containing electron-withdrawing groups such as nitro and halogens (bromo, iodo), as well as heterocyclic systems like indoles, making it highly versatile for diverse pharmaceutical intermediate production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Isoindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in accelerating drug discovery and development. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of polysubstituted isoindole intermediate delivered meets the highest quality standards required by global regulatory bodies.
We invite you to collaborate with our technical team to explore how this rhodium-catalyzed synthesis can optimize your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this streamlined route. We encourage you to contact our technical procurement team today to obtain specific COA data and comprehensive route feasibility assessments tailored to your target molecules, ensuring a seamless transition from concept to commercial reality.
