Advanced Rhodium-Catalyzed Synthesis of Polysubstituted Isoindoles for Commercial Scale-Up
Introduction to Next-Generation Isoindole Synthesis
The landscape of organic synthesis for nitrogen-containing heterocycles is undergoing a significant transformation, driven by the demand for more efficient and environmentally benign methodologies. Patent CN109879792B introduces a groundbreaking approach to constructing polysubstituted isoindole compounds, a structural motif prevalent in numerous biologically active natural products and pharmaceutical agents. Historically, the synthesis of these valuable scaffolds has been plagued by limitations in bonding diversity and product variety, particularly when attempting to install quaternary carbon centers. This invention addresses these critical bottlenecks by leveraging a trivalent rhodium-catalyzed carbon-hydrogen (C-H) activation strategy coupled with an oxidative Heck reaction. The result is a robust, modular synthetic platform capable of generating complex isoindole derivatives with high precision.
The significance of this technology extends beyond academic curiosity; it offers tangible benefits for the industrial production of high-purity pharmaceutical intermediates. Isoindoles serve as core skeletons for potent protein kinase inhibitors, such as staurosporines, and are integral to the development of advanced organic pigments. By enabling the direct functionalization of unfunctionalized hydrocarbon C-H bonds, this method eliminates the need for pre-functionalized halides often required in classical cross-coupling reactions. This paradigm shift not only streamlines the synthetic route but also enhances the overall sustainability of the manufacturing process, aligning perfectly with the green chemistry initiatives adopted by leading global chemical enterprises.
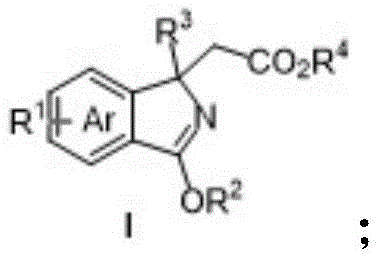
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to isoindole skeletons have long been characterized by their operational complexity and environmental footprint. Classical methods often rely on the thermal decomposition of 1,2,3,4-tetralin-1,4-imines, a process that necessitates extreme temperature fluctuations ranging from high-temperature heating to liquid nitrogen cooling. Such energy-intensive protocols are not only costly to maintain on a large scale but also pose significant safety risks in a production environment. Furthermore, alternative pathways involving the alkylation of 1,2-dibromobenzene followed by high-temperature cyclization generate substantial amounts of halogenated waste, complicating downstream purification and waste treatment procedures.
Another critical drawback of existing technologies is their inability to efficiently construct isoindole compounds featuring quaternary carbon centers. These sterically congested structures are notoriously difficult to access via conventional nucleophilic substitution or condensation reactions, often resulting in low yields and poor selectivity. The reliance on Grignard reagents for subsequent transformations of these quaternary centers introduces additional sensitivity to moisture and air, further restricting the practical utility of these methods in a standard manufacturing setting. Consequently, there has been a persistent need for a more versatile and operationally simple strategy to access these valuable chemical building blocks.
The Novel Approach
The methodology disclosed in patent CN109879792B represents a decisive break from these archaic practices by employing a transition metal-catalyzed tandem reaction sequence. At the heart of this innovation is the use of a trivalent rhodium catalyst to activate inert aryl C-H bonds directly, bypassing the need for pre-installed leaving groups. This C-H activation is seamlessly coupled with an oxidative Heck reaction involving olefin esters, facilitating the simultaneous formation of carbon-carbon and carbon-nitrogen bonds in a single pot. This telescoped approach drastically reduces the number of isolation steps, thereby minimizing material loss and solvent consumption throughout the synthesis.
Moreover, this novel approach exhibits exceptional functional group tolerance, accommodating a wide array of substituents including halogens, esters, nitro groups, and sulfones without compromising reaction efficiency. The ability to preserve sensitive functional groups like aryl bromides and iodides is particularly advantageous for medicinal chemists, as it allows for late-stage diversification via palladium-catalyzed cross-couplings. By shifting the synthetic logic from step-wise construction to a convergent, catalytic assembly, this technology offers a scalable solution for the commercial scale-up of complex heterocyclic scaffolds, ensuring a reliable supply chain for downstream drug development projects.

Mechanistic Insights into Rh(III)-Catalyzed Oxidative Heck Cyclization
The efficacy of this synthesis relies on a sophisticated catalytic cycle initiated by the trivalent rhodium species, typically generated in situ from dimers like [Cp*RhCl2]2. The mechanism begins with the coordination of the imidate nitrogen to the rhodium center, directing the metal to activate the ortho C-H bond of the aromatic ring through a concerted metalation-deprotonation (CMD) pathway facilitated by the acetate base. This step generates a stable five-membered rhodacycle intermediate, which is the pivotal species governing the regioselectivity of the entire transformation. The presence of the silver additive, such as AgNTf2, plays a crucial role in abstracting the chloride ligands from the rhodium precursor, generating a cationic, highly electrophilic rhodium species that is more prone to C-H activation.
Following the C-H activation, the olefin ester coordinates to the rhodium center and undergoes migratory insertion into the Rh-C bond. This step constructs the new carbon-carbon bond and establishes the quaternary center characteristic of the target isoindole structure. Subsequent beta-hydride elimination is suppressed in favor of a reductive elimination or nucleophilic attack by the imidate oxygen/nitrogen, leading to the closure of the five-membered isoindole ring. The catalytic cycle is completed by the re-oxidation of the reduced rhodium(I) species back to the active rhodium(III) state by the copper(II) oxidant, such as Cu(OAc)2, which serves as the terminal oxidant in this aerobic system. This intricate interplay of metals ensures high turnover numbers and sustained catalytic activity throughout the reaction duration.
From an impurity control perspective, the high regioselectivity inherent in the directed C-H activation mechanism ensures that side products arising from non-directed functionalization are minimized. The specific geometry of the rhodacycle intermediate dictates that cyclization occurs exclusively at the ortho position relative to the imidate directing group. This precision is vital for maintaining the purity profile required for pharmaceutical intermediates, where isomeric impurities can be difficult to separate and may impact the biological activity of the final drug substance. The robust nature of the catalyst system also tolerates the presence of electron-withdrawing groups like nitro and cyano moieties, which might otherwise deactivate traditional electrophilic aromatic substitution pathways.
How to Synthesize Polysubstituted Isoindoles Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the stoichiometry of the catalytic components and the choice of solvent. The patent specifies the use of inert solvents such as 1,2-dichloroethane (DCE), which provides an optimal balance of solubility for both organic substrates and inorganic salts while maintaining stability at the required reaction temperature of 80°C. The reaction is notably tolerant to air, eliminating the need for rigorous degassing or glovebox techniques, which simplifies the operational protocol significantly. Typical reaction times range from 4 to 6 hours, allowing for high throughput in a batch processing environment.
- Dissolve the imidate substrate (Formula II) and olefin ester (Formula III) in an inert solvent such as 1,2-dichloroethane (DCE).
- Add the trivalent rhodium catalyst (e.g., [Cp*RhCl2]2), silver additive (AgNTf2), and copper oxidant (Cu(OAc)2) along with sodium acetate base.
- Heat the reaction mixture to 80°C under air atmosphere for 4 hours, then purify the crude product via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this rhodium-catalyzed technology presents a compelling value proposition centered on cost efficiency and supply reliability. By consolidating multiple synthetic steps into a single one-pot operation, the process inherently reduces the consumption of solvents, reagents, and labor hours associated with intermediate isolations. This streamlining of the manufacturing workflow translates directly into cost reduction in fine chemical manufacturing, as fewer unit operations mean lower overheads and reduced capital expenditure on equipment. Furthermore, the avoidance of cryogenic conditions and hazardous reagents like Grignard reagents lowers the safety compliance burden and insurance costs associated with production.
- Cost Reduction in Manufacturing: The elimination of pre-functionalized starting materials, such as aryl halides, in favor of simpler, commodity-grade imidates and olefins significantly lowers the raw material cost basis. Additionally, the high atom economy of the oxidative Heck reaction ensures that a greater proportion of the input mass is converted into the desired product, reducing waste disposal fees. The use of earth-abundant copper salts as the terminal oxidant, rather than expensive stoichiometric oxidants, further optimizes the cost structure of the process.
- Enhanced Supply Chain Reliability: The substrates required for this reaction, including benzimidates and crotonates, are widely available from global chemical suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions, which tolerate air and moisture to a reasonable extent, ensures consistent batch-to-batch reproducibility even in facilities with varying levels of infrastructure. This reliability is crucial for maintaining continuous production schedules and meeting the tight delivery windows demanded by downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The process operates at moderate temperatures (80°C to 100°C) and atmospheric pressure, making it highly amenable to scale-up in standard stainless steel reactors without the need for specialized high-pressure equipment. The reduced generation of halogenated waste and the avoidance of toxic heavy metal residues in the final product simplify the environmental permitting process. This alignment with green chemistry principles facilitates smoother regulatory approvals and enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing a clear understanding of the technology's capabilities and limitations for potential partners and licensees.
Q: What are the key advantages of this Rhodium-catalyzed method over traditional isoindole synthesis?
A: Unlike traditional methods requiring harsh conditions like liquid nitrogen cooling or high-temperature heating of tetralin-imines, this patent describes a mild, one-pot oxidative Heck reaction operating at 80°C under air. It significantly improves atom economy and reduces hazardous waste generation while allowing for diverse functional group tolerance.
Q: Can this synthesis tolerate sensitive functional groups like halogens or nitro groups?
A: Yes, the methodology demonstrates excellent compatibility with various substituents. The patent examples specifically highlight successful synthesis with bromo-, iodo-, and nitro-substituted aromatic rings, preserving these groups for further downstream derivatization, which is critical for medicinal chemistry applications.
Q: Is this process suitable for large-scale manufacturing of pharmaceutical intermediates?
A: The process utilizes readily available starting materials (imidates and olefin esters) and operates under relatively mild conditions (80°C, air atmosphere) without the need for strict inert gas protection in some embodiments. This simplicity, combined with high regioselectivity, makes it highly amenable to commercial scale-up for producing high-purity intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Isoindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of advanced synthetic methodologies in securing a competitive edge in the global market. Our team of expert process chemists has thoroughly evaluated the Rh(III)-catalyzed pathway described in CN109879792B and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications required for GMP-grade intermediates, guaranteeing that every batch meets the highest international standards.
We invite pharmaceutical and agrochemical companies to collaborate with us to leverage this cutting-edge technology for their pipeline projects. By partnering with our technical team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you accelerate your development timelines and optimize your supply chain with our reliable, high-quality polysubstituted isoindole solutions.
