Advanced Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Commercial Pharmaceutical Manufacturing
Advanced Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Commercial Pharmaceutical Manufacturing
The integration of trifluoromethyl groups into heterocyclic scaffolds represents a cornerstone strategy in modern medicinal chemistry, profoundly enhancing the metabolic stability, lipophilicity, and bioavailability of drug candidates. As illustrated in the structural diversity of bioactive molecules such as deferasirox and fluconazole shown below, the 1,2,4-triazole motif is ubiquitous in high-value pharmaceuticals and functional materials. However, the efficient construction of these cores, particularly with specific trifluoromethyl substitution patterns, has historically posed significant synthetic challenges. Patent CN110467579B introduces a transformative preparation method for 5-trifluoromethyl substituted 1,2,4-triazole compounds that addresses these bottlenecks through a novel iodine-promoted cyclization strategy. This technology offers a robust pathway for generating complex nitrogen-containing heterocycles essential for next-generation API development.
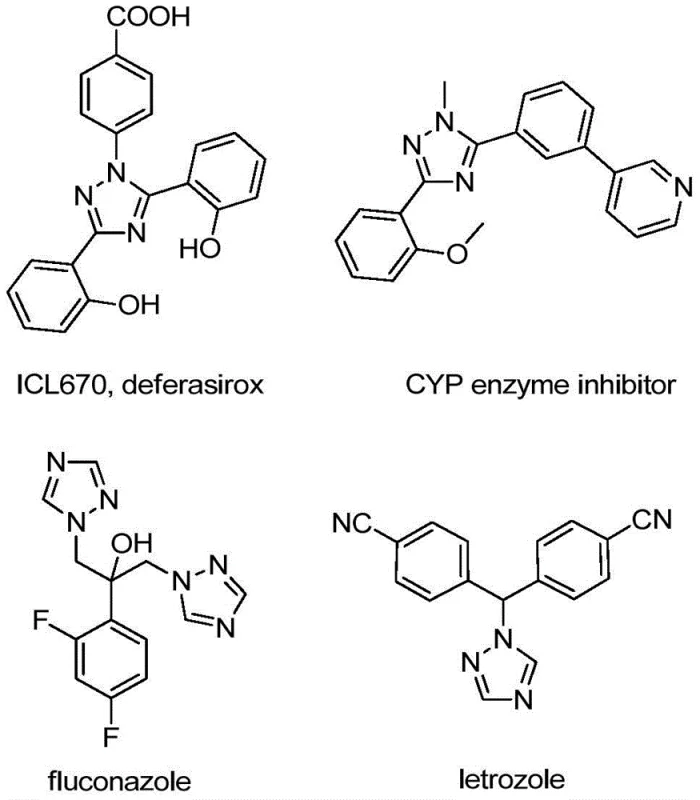
This groundbreaking methodology leverages inexpensive and commercially available hydrazones and trifluoroacetimidoyl chlorides as primary building blocks, circumventing the need for hazardous diazo compounds or costly pre-functionalized heterocycles. By utilizing a simple sodium acetate and elemental iodine system, the process achieves high conversion rates under relatively mild thermal conditions. For procurement managers and supply chain directors, this represents a pivotal shift towards more sustainable and cost-efficient manufacturing protocols. The elimination of stringent anhydrous and anaerobic requirements further simplifies the operational complexity, allowing for seamless translation from laboratory discovery to commercial scale-up without the capital expenditure associated with specialized inert atmosphere reactors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of trifluoromethylated nitrogen heterocycles has relied heavily on two predominant strategies, both of which suffer from significant economic and safety drawbacks in an industrial setting. The first approach involves the direct trifluoromethylation of pre-synthesized 1,2,4-triazole rings using specialized reagents such as Togni or Umemoto reagents. While effective on a small scale, these reagents are prohibitively expensive for large-volume production and often generate substantial stoichiometric waste, complicating downstream purification and waste management. The second conventional route utilizes unstable and potentially explosive synthons like trifluorodiazoethane, which necessitates rigorous safety protocols, specialized equipment, and highly trained personnel to mitigate the risks of thermal runaway or detonation during handling.
Furthermore, many existing catalytic systems depend on precious transition metals such as palladium, copper, or rhodium to facilitate the coupling reactions. For pharmaceutical manufacturers, the use of these heavy metals introduces a critical bottleneck regarding residual metal limits in the final Active Pharmaceutical Ingredient (API). Removing trace metals to meet stringent regulatory standards (often in the parts-per-million range) requires additional purification steps like scavenging or recrystallization, which drastically reduce overall yield and increase the cost of goods sold (COGS). These legacy methods also frequently demand strictly anhydrous and oxygen-free environments, requiring expensive solvents and inert gas manifolds that hinder rapid process optimization and scale-up efficiency.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the method disclosed in patent CN110467579B utilizes a metal-free, iodine-promoted oxidative cyclization that fundamentally simplifies the synthetic landscape. The core innovation lies in the condensation of trifluoroacetimidoyl chloride with hydrazones, followed by an oxidative ring closure mediated by elemental iodine. This reaction pathway, depicted in the general scheme below, proceeds efficiently in common organic solvents like 1,2-dichloroethane (DCE) at moderate temperatures around 80°C. The avoidance of explosive diazo species and the reliance on stable, shelf-stable starting materials dramatically lowers the barrier to entry for safe manufacturing.
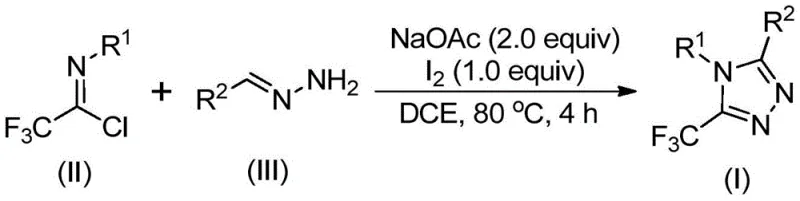
The operational simplicity of this novel approach cannot be overstated for process chemists. The reaction does not require rigorous exclusion of moisture or oxygen, meaning it can be performed in standard glass-lined reactors without the need for complex drying trains or nitrogen purging systems. Moreover, the use of sodium acetate as a mild base ensures compatibility with a wide range of functional groups, allowing for the synthesis of diverse derivatives without protecting group manipulations. This flexibility enables the rapid generation of analog libraries for structure-activity relationship (SAR) studies while maintaining a direct line of sight to commercial viability. The resulting 5-trifluoromethyl-1,2,4-triazoles are obtained in high purity after simple workup procedures, streamlining the path from raw material to finished intermediate.
Mechanistic Insights into Iodine-Promoted Oxidative Cyclization
From a mechanistic perspective, this transformation represents an elegant cascade of bond-forming events driven by the dual role of iodine as both an oxidant and an electrophilic activator. The reaction initiates with a base-promoted intermolecular nucleophilic attack, where the nitrogen of the hydrazone attacks the electrophilic carbon of the trifluoroacetimidoyl chloride. This step forms a trifluoroacetamidine intermediate, establishing the critical carbon-nitrogen backbone of the triazole ring. Subsequent isomerization aligns the reactive centers for cyclization, setting the stage for the key oxidative step that differentiates this method from traditional thermal cyclizations.
The addition of elemental iodine triggers a base-promoted oxidative iodination, generating an iodinated intermediate that serves as a potent electrophile. This species undergoes an intramolecular electrophilic substitution reaction, where the proximal nitrogen atom attacks the iodinated carbon, closing the five-membered ring. The final step involves aromatization and the elimination of hydrogen iodide, driven by the thermodynamic stability of the aromatic triazole system. This mechanism explains the high regioselectivity observed for the 5-trifluoromethyl substitution pattern, as the trifluoromethyl group remains intact throughout the oxidative process. Understanding this pathway allows chemists to fine-tune reaction parameters, such as the stoichiometry of iodine and base, to maximize yield and minimize side products for specific substrate classes.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical execution of this synthesis is designed for reproducibility and ease of operation, making it an ideal candidate for technology transfer between R&D and production units. The protocol involves a straightforward one-pot procedure where reagents are combined sequentially, minimizing unit operations and solvent swaps. Detailed standardized operating procedures for this synthesis, including precise stoichiometric ratios and safety handling guidelines for iodine, are outlined in the technical guide below.
- Mix sodium acetate, trifluoroethylimide chloride, and hydrazone in an organic solvent like DCE.
- Heat the mixture to 80°C and react for 2 to 4 hours to form the intermediate.
- Add elemental iodine to the system and continue reacting for 1 to 2 hours to complete the cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iodine-promoted synthesis offers tangible strategic advantages that extend beyond mere chemical novelty. The primary value driver is the drastic reduction in raw material costs and processing complexity. By replacing expensive transition metal catalysts and hazardous diazo reagents with commodity chemicals like iodine and sodium acetate, the direct material cost of the synthesis is significantly lowered. Furthermore, the absence of heavy metals eliminates the need for costly metal scavenging resins and extensive analytical testing for residual metals, which are major cost centers in API manufacturing. This streamlined process directly contributes to improved margins and a more competitive pricing structure for the final pharmaceutical intermediate.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts such as palladium or copper removes a significant variable cost from the production budget. Additionally, the use of cheap and widely available starting materials like hydrazones and trifluoroacetimidoyl chlorides ensures stable pricing and reduces exposure to volatile commodity markets. The simplified workup procedure, which avoids complex chromatographic separations often required for metal removal, further reduces solvent consumption and labor hours, leading to substantial overall cost savings in the manufacturing process.
- Enhanced Supply Chain Reliability: The robustness of this reaction conditions means that production is less susceptible to disruptions caused by environmental factors or equipment failures. Since the reaction does not require strict anhydrous or anaerobic conditions, it can be run in standard facilities without specialized infrastructure, increasing the number of qualified contract manufacturing organizations (CMOs) capable of producing the material. The high functional group tolerance also means that a single versatile platform can produce a wide array of derivatives, reducing the need for multiple distinct supply chains for different analogs.
- Scalability and Environmental Compliance: The process has been demonstrated to be scalable from gram to multi-kilogram levels with consistent yields, indicating strong potential for tonnage production. From an environmental standpoint, the avoidance of toxic heavy metals and explosive reagents simplifies waste treatment and disposal, ensuring compliance with increasingly stringent environmental regulations. The use of common solvents like DCE, which can be recovered and recycled, further enhances the green chemistry profile of the process, aligning with corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and scope defined in the patent literature, providing a factual basis for feasibility assessments.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the method described in patent CN110467579B utilizes elemental iodine as a promoter instead of expensive heavy metal catalysts like palladium or copper, significantly reducing purification costs and residual metal risks.
Q: What are the reaction conditions for this triazole formation?
A: The reaction operates under mild conditions, typically heating to 80°C in a solvent like dichloroethane (DCE), without the need for strict anhydrous or anaerobic environments.
Q: Is this method suitable for large-scale production?
A: Yes, the process uses cheap and readily available starting materials and avoids sensitive conditions, making it highly scalable for industrial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of novel therapeutic agents depends on access to high-quality, reliably sourced intermediates. Our technical team has extensively evaluated the iodine-promoted cyclization route described in CN110467579B and validated its potential for robust manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 5-trifluoromethyl-1,2,4-triazole meets the exacting standards required for global pharmaceutical registration.
We invite you to collaborate with us to leverage this advanced synthetic technology for your pipeline projects. Whether you require custom synthesis of specific analogs or bulk supply of standard intermediates, our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your development timeline with superior chemical solutions.
