Advanced Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for High-Purity Pharmaceutical Intermediates
Advanced Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for High-Purity Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways to access nitrogen-containing heterocycles, particularly those bearing trifluoromethyl groups which confer superior metabolic stability and lipophilicity to drug candidates. Patent CN110467579B, published in August 2020, discloses a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,4-triazole compounds that addresses many of the historical bottlenecks in heterocyclic synthesis. This technology leverages a non-metallic iodine-promoted cyclization strategy, utilizing inexpensive hydrazones and trifluoroethylimidoyl chloride as key building blocks. The significance of this development cannot be overstated for R&D teams focusing on next-generation APIs and functional materials, as 1,2,4-triazole scaffolds are ubiquitous in high-value therapeutics ranging from antifungal agents to kinase inhibitors. As illustrated in the structural diversity of known bioactive molecules, the integration of this specific heterocyclic core is a critical design element in modern medicinal chemistry.
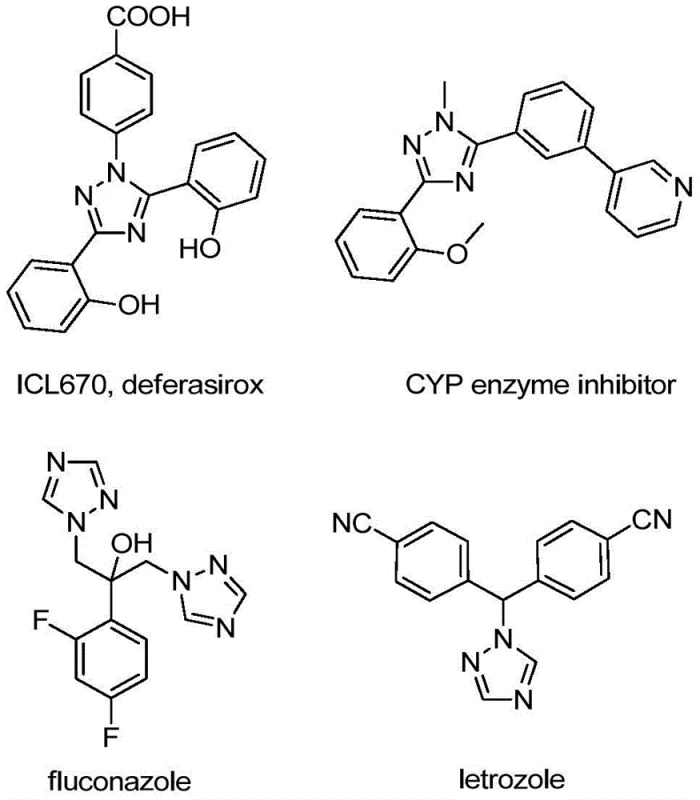
Furthermore, the introduction of the trifluoromethyl moiety at the 5-position of the triazole ring significantly enhances the electronegativity and bioavailability of the resulting compounds, making them highly desirable candidates for lead optimization programs. The patent highlights that these compounds are not only relevant to the pharmaceutical sector but also find applications in organic light-emitting diodes (OLEDs) as ligands for metal coordination. For procurement managers and supply chain directors, the shift towards such efficient synthetic methodologies represents a strategic opportunity to secure reliable sources of complex intermediates while mitigating the risks associated with volatile reagent markets and complex regulatory compliance regarding heavy metal residues.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted nitrogen heterocycles has been plagued by significant operational and economic challenges that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Traditional literature reports primarily describe two dominant strategies, both of which suffer from distinct drawbacks in an industrial setting. The first approach involves the direct trifluoromethylation of pre-synthesized nitrogen-containing heterocycles, a process that invariably requires the use of specialized, often hazardous, and prohibitively expensive trifluoromethylating reagents. These reagents frequently demand strict anhydrous and anaerobic conditions, necessitating costly infrastructure investments in gloveboxes or specialized reactor setups that increase the overall cost of goods sold. The second mainstream method relies on the reaction of synthons bearing trifluoromethyl groups with coupling substrates, yet even here, the most common synthons like trifluorodiazoethane pose severe safety risks due to their explosive nature, while trifluoroethylimide acid halides have seen relatively limited application due to perceived reactivity issues. Consequently, existing manufacturing routes often result in low atom economy, difficult purification profiles, and substantial waste generation, creating friction for supply chain heads aiming for green chemistry compliance.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN110467579B introduces a paradigm shift by employing a simple, efficient, and metal-free cyclization strategy that dramatically simplifies the manufacturing workflow. This novel approach utilizes readily available hydrazones and trifluoroethylimidoyl chloride as starting materials, reacting them in the presence of sodium acetate and elemental iodine within a standard organic solvent system. The elimination of toxic heavy metal catalysts is a pivotal advancement, as it removes the need for expensive and time-consuming metal scavenging steps during downstream processing, thereby directly contributing to cost reduction in API manufacturing. Moreover, the reaction proceeds smoothly under aerobic conditions without the stringent requirement for anhydrous environments, allowing for the use of standard reactor vessels and significantly lowering the barrier to entry for contract manufacturing organizations. The operational simplicity extends to the workup procedure, which involves basic filtration and silica gel treatment followed by column chromatography, ensuring that high-purity 1,2,4-triazole derivatives can be obtained with minimal technical overhead and maximum throughput efficiency.
Mechanistic Insights into Iodine-Promoted Cyclization
To fully appreciate the robustness of this synthetic route for R&D directors evaluating process feasibility, it is essential to dissect the underlying mechanistic pathway proposed in the patent data. The transformation begins with a base-promoted intermolecular carbon-nitrogen bond formation between the trifluoroethylimidoyl chloride and the hydrazone substrate, leading to the generation of a trifluoroacetamidine intermediate. This initial condensation step is critical as it sets the stage for the subsequent ring closure, facilitated by the mild basicity of sodium acetate which neutralizes the HCl byproduct without degrading sensitive functional groups. Following this, the system undergoes an isomerization process that aligns the reactive centers for cyclization. The addition of elemental iodine then triggers a base-promoted oxidative iodination, generating a reactive iodine-containing intermediate that serves as the electrophile for the final ring-closing event. This sequence avoids the high-energy barriers typically associated with direct C-H activation or radical trifluoromethylation, offering a more controlled and predictable reaction profile that is ideal for maintaining consistent impurity profiles across different batches.
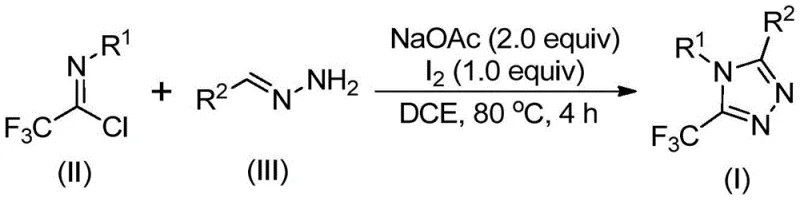
The final stage of the mechanism involves an intramolecular electrophilic substitution reaction followed by aromatization, which drives the equilibrium towards the formation of the stable 5-trifluoromethyl-1,2,4-triazole core. This mechanistic understanding is vital for quality control teams, as it explains the high functional group tolerance observed in the experimental data; the mild nature of the iodine promotion ensures that sensitive substituents such as esters, ethers, and halogens remain intact throughout the synthesis. Furthermore, the use of dichloroethane (DCE) as the preferred solvent optimizes the solubility of all reactants and intermediates, ensuring homogeneous reaction conditions that minimize the formation of side products. By controlling the stoichiometry carefully, specifically maintaining a molar ratio of trifluoroethylimidoyl chloride to hydrazone of approximately 1:2, the process maximizes conversion rates while minimizing the accumulation of unreacted starting materials, thus streamlining the purification process and enhancing the overall yield of the desired high-purity intermediates.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to ensure optimal results and reproducibility. The patent outlines a straightforward protocol where sodium acetate, trifluoroethylimidoyl chloride, and the chosen hydrazone are combined in an organic solvent, typically dichloroethane, and heated to temperatures between 80°C and 100°C. This initial heating phase is maintained for 2 to 4 hours to allow the condensation and isomerization steps to reach completion before the oxidant is introduced. Once the initial reaction period concludes, elemental iodine is added to the mixture, and the heating is continued for an additional 1 to 2 hours to drive the oxidative cyclization to completion. The detailed standardized synthesis steps, including precise stoichiometric ratios and purification techniques, are provided in the guide below to assist technical teams in replicating this high-efficiency process.
- Combine sodium acetate, trifluoroethylimidoyl chloride, and hydrazone in an organic solvent such as dichloroethane.
- Heat the reaction mixture to 80-100°C and maintain stirring for 2 to 4 hours to facilitate initial condensation.
- Introduce elemental iodine to the system, continue heating for 1 to 2 hours, then filter and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this iodine-promoted synthesis route offers tangible strategic benefits that extend far beyond simple chemical transformation. The primary value proposition lies in the drastic simplification of the supply chain for raw materials; hydrazones are easily prepared from widely available aldehydes and hydrazine hydrate, while trifluoroethylimidoyl chloride can be rapidly synthesized from commodity chemicals like aromatic amines and trifluoroacetic acid. This reliance on bulk chemical feedstocks rather than specialized, custom-synthetized reagents significantly de-risks the sourcing strategy, ensuring long-term supply continuity even in volatile market conditions. Additionally, the avoidance of noble metal catalysts or exotic trifluoromethylating agents eliminates a major cost center, allowing for substantial cost savings in the overall production budget without compromising on the quality or purity of the final intermediate.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive catalytic systems and the reduction of downstream processing complexity. By avoiding heavy metal catalysts, manufacturers save significantly on the costs associated with metal scavengers, specialized filtration equipment, and the rigorous testing required to meet residual metal specifications in pharmaceutical products. Furthermore, the ability to run the reaction under ambient atmospheric conditions removes the capital expenditure and operational costs linked to maintaining inert gas lines and anhydrous solvent systems. These cumulative efficiencies translate into a lower cost per kilogram for the final 5-trifluoromethyl-1,2,4-triazole product, providing a competitive edge in pricing negotiations with downstream API producers.
- Enhanced Supply Chain Reliability: From a logistics perspective, the use of stable, non-hazardous starting materials like hydrazones and elemental iodine simplifies storage and transportation requirements compared to explosive diazo compounds or moisture-sensitive reagents. This stability reduces the risk of shipment delays or regulatory hurdles associated with hazardous material transport, ensuring a more reliable flow of materials into the production facility. The robustness of the reaction conditions also means that the process is less susceptible to batch failures caused by minor fluctuations in environmental conditions, thereby improving the predictability of delivery schedules and strengthening the trust relationship between suppliers and their pharmaceutical clients.
- Scalability and Environmental Compliance: The patent explicitly notes the ease of scaling this method to the gram level and beyond, indicating strong potential for multi-kilogram or ton-scale production. The simplified workup procedure, involving basic filtration and chromatography, generates less hazardous waste compared to traditional methods that might require quenching of reactive species or complex extraction protocols. This alignment with green chemistry principles not only reduces waste disposal costs but also aids in meeting increasingly stringent environmental regulations, making the process sustainable for long-term commercial operation and attractive to eco-conscious stakeholders in the global supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel triazole synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating the feasibility of adopting this route for their specific projects. Understanding these nuances is crucial for making informed decisions about process integration and resource allocation.
Q: What are the primary advantages of this iodine-promoted method over traditional trifluoromethylation?
A: Unlike conventional methods requiring expensive trifluoromethylating reagents or harsh anhydrous conditions, this protocol utilizes cheap, readily available hydrazones and trifluoroethylimidoyl chloride under mild, metal-free conditions.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates that the method is easily scalable to the gram level and beyond, utilizing simple post-treatment processes like filtration and column chromatography without needing specialized inert atmosphere equipment.
Q: What is the substrate scope for the R1 and R2 groups in this triazole synthesis?
A: The method exhibits broad functional group tolerance, successfully accommodating substituted aryl groups (methyl, methoxy, bromo, trifluoromethyl), heteroaryl groups (thiophene, pyridine, furan), and alkenyl groups at both the N1 and C3 positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
As the global demand for fluorinated heterocycles continues to surge, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthetic technologies backed by rigorous quality standards. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, allowing us to seamlessly transition promising laboratory methods like the one described in CN110467579B into robust industrial processes. Our state-of-the-art facilities are equipped with stringent purity specifications and rigorous QC labs capable of detecting trace impurities, guaranteeing that every batch of 5-trifluoromethyl-1,2,4-triazole intermediate meets the exacting requirements of top-tier pharmaceutical companies. We understand that consistency is key in drug development, and our commitment to process optimization ensures that your supply remains uninterrupted and compliant with international regulatory standards.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this metal-free methodology for your pipeline. We encourage you to contact us today to obtain specific COA data for our existing triazole inventory or to initiate a dialogue regarding route feasibility assessments for your custom synthesis projects, ensuring that your development timeline remains on track with the highest quality materials available.
