Advanced Four-Step Synthesis of Dexamethasone Intermediate for Scalable API Production
Advanced Four-Step Synthesis of Dexamethasone Intermediate for Scalable API Production
The pharmaceutical industry continuously seeks robust and efficient pathways for the synthesis of critical corticosteroid intermediates, particularly for high-volume drugs like Dexamethasone. A significant breakthrough in this domain is detailed in patent CN102603843A, which discloses a streamlined preparation method for a key dexamethasone intermediate, specifically Compound V. This intermediate is pivotal because it possesses the crucial 21-hydroxyl functional group required for the final synthesis of Dexamethasone BP98, eliminating the need for complex downstream functionalization. The patented route transforms the readily available starting material, 1,4,9-trien-androstane-3,20-dione (Compound I), through a sequence of ethynylation, esterification, oxidation, and rearrangement reactions. This approach not only addresses the yield limitations of prior art but also offers a more direct path to the target molecule, making it highly attractive for manufacturers aiming to optimize their API supply chains.
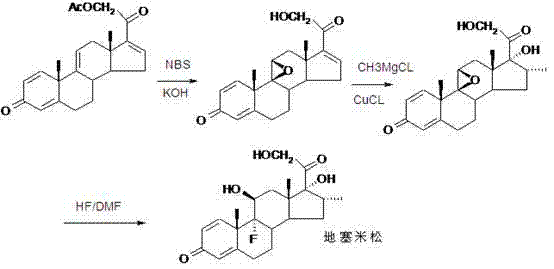
Understanding the structural evolution from the starting ketone to the final intermediate is essential for R&D teams evaluating process feasibility. The transformation involves introducing an acetyl side chain and modifying the steroid backbone to match the pharmacological requirements of glucocorticoids. As illustrated in the downstream conversion pathway, the resulting Compound V serves as a direct precursor that can be hydrolyzed to obtain the active pharmaceutical ingredient. This strategic positioning of the intermediate reduces the total number of synthetic steps required from bulk steroids to the final drug substance. For procurement managers and supply chain heads, this reduction in step count translates directly to reduced operational complexity and potentially lower cost of goods sold (COGS), provided the individual step yields are maintained at commercially viable levels as reported in the patent data.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dexamethasone intermediates has been plagued by inefficient multi-step protocols that hinder large-scale production capabilities. Prior art, such as the methods disclosed in CN101979399A and CN101397320A, typically relies on starting materials like 21-methyl tetraene compounds which themselves require extensive processing. These conventional routes often necessitate a series of chemically demanding reactions, including Grignard additions, epoxidation, fluorination, iodination, and subsequent hydrolysis. Each of these steps introduces potential points of failure, impurity generation, and yield loss. Furthermore, the reliance on Grignard reagents imposes strict anhydrous conditions and cryogenic temperatures, which are energy-intensive and difficult to manage safely on a multi-ton scale. The cumulative effect of these lengthy sequences is a significantly lower overall efficiency of the PCR (Process Critical Reaction) products and extended production lead times, creating bottlenecks in the supply of high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology presented in CN102603843A offers a concise four-step trajectory that bypasses the cumbersome functional group manipulations of the past. By starting directly with 1,4,9-trien-androstane-3,20-dione, the process leverages a direct ethynylation to build the carbon skeleton, followed by a protective esterification and a catalytic oxidation. The culmination of the sequence is a rearrangement reaction that elegantly establishes the necessary side-chain architecture. This novel approach is characterized by its use of relatively mild conditions and accessible reagents, such as acetylene, organic acid anhydrides, and molecular oxygen. The elimination of hazardous halogenation steps and complex organometallic additions simplifies the waste stream and reduces the burden on environmental compliance teams. For a reliable pharmaceutical intermediate supplier, adopting this route means achieving a more robust manufacturing process with fewer unit operations, directly addressing the industry's demand for cost reduction in API manufacturing.
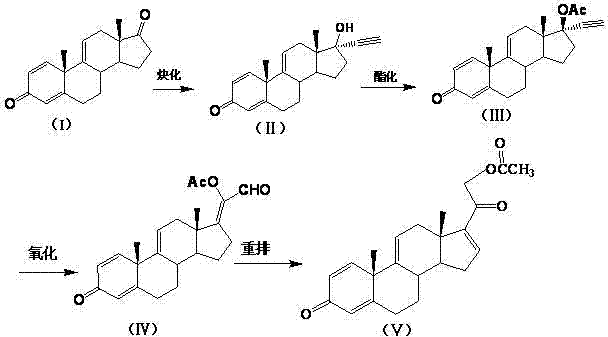
Mechanistic Insights into the Catalytic Oxidation and Rearrangement
The core chemical innovation in this patent lies in the specific execution of the oxidation and rearrangement steps, which dictate the purity and yield of the final intermediate. The oxidation of Compound III to Compound IV is facilitated by a dual-catalyst system comprising a palladium species, such as potassium tetrabromopalladate or palladous chloride, and a cupric chloride cocatalyst. This system operates effectively under an oxygen or air atmosphere at temperatures ranging from 40°C to 70°C. The presence of the copper cocatalyst is critical for regenerating the active palladium species, ensuring a continuous catalytic cycle that minimizes the requirement for expensive noble metals. Following oxidation, the rearrangement of Compound IV to Compound V is triggered by strong organic bases like 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU) or tetrahydropyrimidine. This base-mediated rearrangement proceeds under inert gas protection, preventing unwanted side reactions and ensuring the stereochemical integrity of the steroid nucleus is maintained throughout the transformation.
Impurity control is another critical aspect where this mechanism offers distinct advantages over traditional routes. The ethynylation step, which converts Compound I to Compound II, is performed using potassium hydroxide and acetylene in solvents like THF or ether at controlled low temperatures (-5°C to 10°C). The patent specifies the use of solubility promoters like ethanol to enhance the dissolution of KOH, ensuring a homogeneous reaction environment that minimizes the formation of polymeric byproducts often associated with acetylene chemistry. Furthermore, the workup procedures described, involving pH adjustment and careful precipitation, are designed to remove residual catalysts and unreacted starting materials effectively. By strictly controlling the reaction parameters, such as the weight ratio of Compound I to KOH (1:1 to 1:3) and the feeding rate of acetylene, manufacturers can achieve a product purity of up to 98%, which is essential for meeting the stringent quality standards required for hormonal pharmaceutical intermediates.
How to Synthesize Dexamethasone Intermediate Efficiently
The synthesis of this high-value intermediate requires precise adherence to the reaction conditions outlined in the patent to ensure reproducibility and safety. The process begins with the preparation of potassium ethynylide under inert atmosphere, followed by the sequential addition of the steroid ketone. Subsequent steps involve careful temperature management during esterification and oxidation to prevent degradation of the sensitive diene system. The final rearrangement step is particularly sensitive to base concentration and temperature, requiring close monitoring via TLC or HPLC to determine the optimal endpoint. For process chemists looking to implement this technology, the following guide outlines the standardized operational parameters derived from the patent examples, ensuring a smooth transition from laboratory bench to pilot plant.
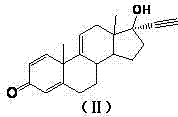
- Perform ethynylation on Compound I using potassium hydroxide and acetylene in an organic solvent under inert gas protection to obtain Compound II.
- Conduct esterification on Compound II with organic acid anhydride and a catalyst such as DMAP or triethylamine to yield Compound III.
- Execute oxidation of Compound III using a palladium catalyst, cupric chloride cocatalyst, and oxygen source to form Compound IV.
- Complete the synthesis via rearrangement of Compound IV using 1,8-diazabicyclo[5.4.0]undec-7-ene or tetrahydropyrimidine to obtain the final intermediate Compound V.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route presents compelling economic and logistical benefits that extend beyond simple yield metrics. The primary advantage lies in the significant simplification of the raw material portfolio. By utilizing 1,4,9-trien-androstane-3,20-dione, which is a commercially available and cost-effective bulk steroid, the process avoids the need for custom-synthesized precursors that often carry high price tags and long lead times. Additionally, the elimination of Grignard reagents and complex fluorination agents reduces the dependency on specialized, hazard-class chemicals that require expensive storage and handling infrastructure. This shift towards more benign reagents not only lowers the direct material costs but also mitigates the risks associated with supply chain disruptions for niche chemical inputs, thereby enhancing the overall reliability of the manufacturing schedule.
- Cost Reduction in Manufacturing: The streamlined four-step sequence inherently reduces the operational expenditure associated with multi-step synthesis. By cutting down the number of isolation and purification stages, the process saves on solvent consumption, energy usage for heating and cooling, and labor hours. The use of air or oxygen as the oxidant in the third step is a prime example of cost-efficient reagent selection, replacing expensive stoichiometric oxidants with a ubiquitous gas. Furthermore, the catalytic nature of the oxidation step means that precious metal loading can be minimized and potentially recovered, contributing to substantial cost savings in the long run without compromising the reaction efficiency.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes directly to supply chain stability. Unlike cryogenic Grignard reactions that are susceptible to failure due to minor temperature fluctuations or moisture ingress, the ethynylation and rearrangement steps in this protocol operate within manageable temperature ranges (0°C to 70°C). This tolerance makes the process more forgiving and easier to control in a standard multipurpose chemical plant, reducing the likelihood of batch failures. Consequently, manufacturers can offer more consistent delivery schedules to their clients, reducing the lead time for high-purity pharmaceutical intermediates and ensuring a steady flow of material for downstream API production.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route is superior to conventional methods. The avoidance of heavy halogenation steps reduces the generation of halogenated waste streams, which are costly to treat and dispose of. The solvents used, such as ethyl acetate, ethanol, and THF, are common industrial solvents with well-established recovery and recycling protocols. This alignment with green chemistry principles facilitates easier regulatory approval and permits for capacity expansion. The ability to scale from kilogram to ton quantities without fundamental changes to the chemistry ensures that the supply can grow in tandem with market demand for dexamethasone, securing the supply chain against future shortages.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dexamethasone intermediate synthesis. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of what partners can expect when integrating this technology into their production lines. Understanding these nuances is vital for making informed decisions about process adoption and vendor qualification.
Q: What are the limitations of conventional dexamethasone intermediate synthesis methods?
A: Conventional methods often rely on multi-step sequences involving Grignard reactions, epoxy reactions, and fluorination, which result in lower overall efficiency, longer reaction times, and complex purification requirements compared to the novel four-step route.
Q: How does the new oxidation step improve process efficiency?
A: The novel oxidation step utilizes a palladium catalyst system with cupric chloride and molecular oxygen or air, allowing for milder reaction conditions (40-70°C) and avoiding harsh reagents, thereby simplifying the workflow and improving safety profiles.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the route is designed for scalability, utilizing readily available starting materials like 1,4,9-trien-androstane-3,20-dione and employing robust reaction conditions that facilitate commercial scale-up while maintaining high product purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dexamethasone Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient intermediate synthesis plays in the global availability of essential medicines. Our technical team has extensively analyzed the route disclosed in CN102603843A and validated its potential for industrial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of dexamethasone intermediate we supply meets the highest international standards for pharmaceutical use.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage this advanced synthesis technology. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to discuss specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both cost and continuity in the competitive landscape of corticosteroid manufacturing.
