Scalable Metal-Free Synthesis of Trifluoromethyl-1,2,4-Triazoles for Advanced Pharmaceutical Applications
Scalable Metal-Free Synthesis of Trifluoromethyl-1,2,4-Triazoles for Advanced Pharmaceutical Applications
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more sustainable, cost-effective, and scalable synthetic routes. A significant breakthrough in this domain is detailed in Chinese Patent CN113105402B, which discloses a novel preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds. These heterocyclic scaffolds are critical structural motifs found in numerous high-value active pharmaceutical ingredients (APIs), including blockbuster drugs such as Maraviroc, Sitagliptin, and Deferasirox. The introduction of a trifluoromethyl group into these heterocycles is particularly desirable as it significantly enhances physicochemical properties such as metabolic stability, lipophilicity, and bioavailability. This patent presents a robust, iodine-promoted methodology that bypasses traditional limitations, offering a streamlined pathway for producing these complex molecules with high efficiency and purity.
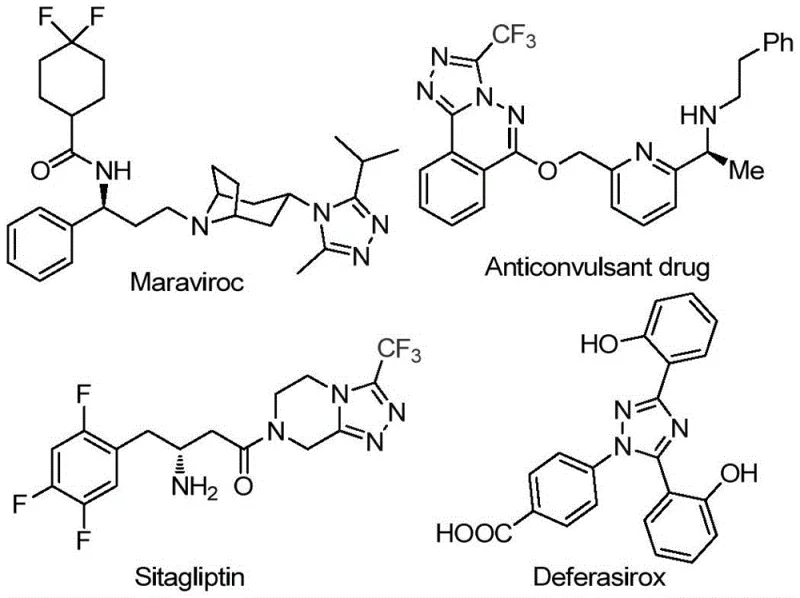
The strategic importance of this technology cannot be overstated for R&D directors and procurement managers alike. By leveraging a metal-free catalytic system, this process eliminates the substantial downstream costs associated with removing heavy metal residues, a common bottleneck in API synthesis. Furthermore, the use of inexpensive, commercially available starting materials like aryl ethyl ketones ensures a stable supply chain, reducing the risk of raw material shortages. As we delve deeper into the technical specifics, it becomes clear that this innovation represents a paradigm shift towards greener, more economical chemical manufacturing, aligning perfectly with the industry's push for sustainable development and cost reduction in pharmaceutical intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted 1,2,4-triazoles, particularly those bearing both trifluoromethyl and acyl groups, has been fraught with challenges. Conventional methodologies often rely heavily on transition metal catalysts, such as copper or palladium complexes, which are not only expensive but also pose significant environmental and safety hazards. The presence of these heavy metals necessitates rigorous purification steps to meet the stringent residual metal limits imposed by regulatory bodies like the FDA and EMA. These additional purification stages, often involving specialized scavengers or repeated chromatography, drastically increase the overall production cost and extend the lead time for high-purity pharmaceutical intermediates. Moreover, many traditional routes require harsh reaction conditions, including strictly anhydrous and oxygen-free environments, which demand specialized equipment and increase operational complexity, making scale-up difficult and risky for commercial production facilities.
The Novel Approach
In stark contrast, the method disclosed in patent CN113105402B offers a refreshing departure from these cumbersome protocols. By utilizing elemental iodine as a non-metal promoter in conjunction with dimethyl sulfoxide (DMSO), this novel approach facilitates a tandem iodination and Kornblum oxidation sequence that proceeds smoothly under aerobic conditions. This eliminates the need for expensive inert gas protection and complex drying procedures, significantly simplifying the operational workflow. The reaction tolerates a wide range of functional groups on both the aryl ketone and the hydrazide substrates, allowing for the facile synthesis of diverse derivatives without the need for protecting group strategies. This versatility, combined with the avoidance of toxic heavy metals, results in a cleaner reaction profile and a much simpler workup procedure, typically requiring only filtration and standard column chromatography to achieve high-purity products suitable for further pharmaceutical development.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
To fully appreciate the elegance of this synthesis, one must understand the underlying mechanistic pathway that drives the formation of the 1,2,4-triazole ring. The reaction initiates with the iodination of the aryl ethyl ketone, mediated by molecular iodine in DMSO. This is followed by a Kornblum oxidation, where the alpha-iodo ketone intermediate is converted into an alpha-diketone species. This oxidative transformation is crucial as it generates the necessary electrophilic center for the subsequent condensation step. The generated alpha-diketone then undergoes a dehydration condensation with trifluoroethylimide hydrazide to form a hydrazone intermediate. This step is facilitated by the basic environment provided by the additives, ensuring efficient nucleophilic attack. Finally, the hydrazone undergoes an intramolecular cyclization, promoted by the continued presence of iodine and base, to close the triazole ring and yield the final 3,4,5-trisubstituted product. This cascade reaction design maximizes atom economy and minimizes waste generation.
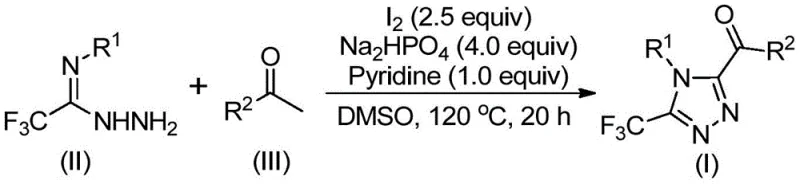
From an impurity control perspective, this mechanism offers distinct advantages. The use of DMSO as both solvent and oxidant ensures a homogeneous reaction medium, which helps in suppressing side reactions that often occur in heterogeneous systems. The specific stoichiometry of reagents, particularly the molar ratio of sodium dihydrogen phosphate to pyridine to iodine (optimized at 4:1:2.5), plays a critical role in buffering the reaction pH and controlling the rate of oxidation versus cyclization. This precise control prevents the over-oxidation of sensitive functional groups and minimizes the formation of polymeric byproducts. For R&D teams, understanding these nuances is vital for troubleshooting and optimizing the process for specific substrates, ensuring that the final API intermediate meets the rigorous quality standards required for clinical trials and commercial launch.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
The practical implementation of this synthesis route is designed for ease of execution, making it highly attractive for process chemistry teams looking to transfer technology from the lab to the pilot plant. The protocol involves a straightforward two-stage heating process in a standard Schlenk tube or reactor. Initially, the aryl ethyl ketone and iodine are heated in DMSO to generate the reactive diketone species. Subsequently, the hydrazide component and buffering agents are introduced to drive the cyclization to completion. The robustness of this method allows for significant flexibility in substrate scope, accommodating various electron-donating and electron-withdrawing groups on the aromatic rings. For detailed operational parameters and specific substrate examples, please refer to the standardized synthesis guide below which outlines the precise steps for reproducibility.
- Mix aryl ethyl ketone and iodine in DMSO, heating to 90-110°C for 4-6 hours to initiate Kornblum oxidation.
- Add sodium dihydrogen phosphate, pyridine, and trifluoroethylimide hydrazide to the reaction mixture.
- Heat the solution to 110-130°C for 12-20 hours to complete the cyclization, followed by filtration and column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iodine-promoted synthesis route translates into tangible strategic benefits that go beyond mere chemical yield. The primary advantage lies in the drastic simplification of the supply chain for raw materials. Unlike methods requiring exotic ligands or precious metal catalysts that are subject to geopolitical supply fluctuations and price volatility, elemental iodine and aryl ketones are commodity chemicals available in bulk quantities from multiple global suppliers. This diversification of the supply base significantly mitigates the risk of production stoppages due to raw material shortages, ensuring a consistent and reliable flow of intermediates for downstream API manufacturing. Furthermore, the elimination of heavy metals removes the need for expensive metal scavenging resins and the associated disposal costs, directly impacting the bottom line through reduced operational expenditures.
- Cost Reduction in Manufacturing: The economic impact of switching to this metal-free protocol is profound. By removing the dependency on transition metal catalysts, manufacturers avoid the high procurement costs associated with palladium or copper salts, which can fluctuate wildly in the global market. Additionally, the simplified purification process means less solvent consumption and reduced labor hours spent on complex workups, leading to substantial cost savings per kilogram of product. The ability to run the reaction under aerobic conditions also reduces utility costs related to nitrogen or argon consumption, further enhancing the overall cost-efficiency of the manufacturing process.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust, shelf-stable reagents that do not require special storage conditions like cryogenic temperatures or strict moisture exclusion. This ease of handling reduces logistics costs and minimizes the risk of material degradation during transport and storage. The scalability of the reaction, demonstrated from gram to multi-gram levels in the patent data, suggests a smooth path to ton-scale production without the need for significant re-engineering of the process. This predictability allows supply chain planners to forecast production timelines with greater accuracy, ensuring timely delivery to pharmaceutical clients.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns well with modern green chemistry principles. The absence of toxic heavy metals simplifies wastewater treatment and waste disposal, reducing the environmental footprint of the manufacturing facility. This compliance with stricter environmental regulations avoids potential fines and shutdowns, ensuring uninterrupted production. The use of DMSO, a high-boiling polar aprotic solvent, also facilitates easier product isolation and solvent recovery, contributing to a more sustainable and circular manufacturing model that appeals to environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the feasibility and advantages of adopting this route for your specific project needs. Understanding these details is crucial for making informed decisions about process development and vendor selection.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the method described in patent CN113105402B utilizes elemental iodine as a non-metal promoter, completely avoiding the need for costly and toxic heavy metal catalysts like palladium or copper.
Q: What are the optimal reaction conditions for this triazole formation?
A: The process operates effectively in dimethyl sulfoxide (DMSO) solvent, utilizing a two-stage heating protocol: initial oxidation at 90-110°C followed by cyclization at 110-130°C under aerobic conditions.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the reaction can be easily expanded to the gram level and beyond, utilizing cheap and readily available starting materials without stringent anhydrous or oxygen-free requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of novel triazole derivatives or reliable supply of established intermediates, our infrastructure is designed to meet the demanding requirements of the global pharmaceutical industry with speed and precision.
We invite you to collaborate with us to leverage this innovative iodine-promoted synthesis technology for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain and accelerate your time-to-market with our superior manufacturing capabilities and dedication to excellence.
