Advanced Iodine-Catalyzed Synthesis of High-Purity Triazole Intermediates for Pharmaceutical Manufacturing
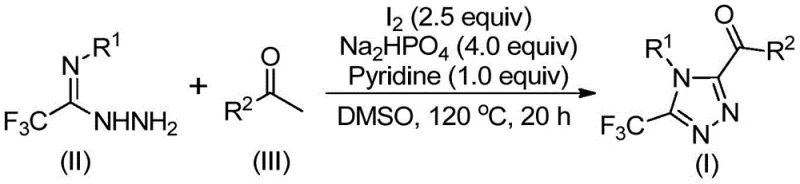
This technical analysis examines Chinese Patent CN113105402B, which discloses an innovative iodine-catalyzed methodology for synthesizing 3,4,5-trisubstituted 1,2,4-triazole compounds containing trifluoromethyl groups. The patent represents a significant advancement in heterocyclic chemistry by providing a streamlined synthetic route that eliminates the need for transition metal catalysts while maintaining high yields and purity levels essential for pharmaceutical applications. This novel approach addresses critical limitations in existing synthetic methodologies by operating under standard atmospheric conditions without requiring anhydrous or oxygen-free environments. The process utilizes readily available starting materials including aryl ethyl ketones and trifluoroethylimide hydrazides, making it economically attractive for commercial scale-up. The elimination of heavy metal catalysts not only reduces costs but also simplifies downstream purification processes required for pharmaceutical intermediates. This patent demonstrates exceptional potential for industrial implementation due to its operational simplicity and robust reaction profile across diverse substrate combinations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for preparing multi-substituted triazole compounds typically require transition metal catalysts such as copper or palladium complexes, which introduce significant challenges in pharmaceutical manufacturing environments. These metal-catalyzed processes necessitate stringent anhydrous and oxygen-free conditions that substantially increase operational complexity and equipment requirements at commercial scale. The presence of heavy metal residues creates substantial purification hurdles that can compromise final product purity and require additional processing steps to meet pharmaceutical quality standards. Furthermore, many conventional methods exhibit limited substrate scope with poor functional group tolerance, restricting their applicability across diverse molecular architectures required in modern drug discovery programs. The high cost of transition metal catalysts and the specialized handling requirements associated with these systems create significant economic barriers for large-scale production of triazole-based pharmaceutical intermediates.
The Novel Approach
The patented methodology overcomes these limitations through an elegant iodine-catalyzed process that operates under standard atmospheric conditions without requiring specialized equipment or handling procedures. By utilizing elemental iodine as a catalyst in dimethyl sulfoxide solvent system, the reaction achieves high efficiency while eliminating concerns about metal contamination in the final product. The process demonstrates remarkable substrate flexibility with excellent functional group tolerance across a wide range of aryl substituents as demonstrated in the patent examples. The reaction conditions are operationally simple and scalable from laboratory to commercial production volumes without requiring significant process modifications. Most significantly, the elimination of transition metal catalysts removes the need for extensive purification steps to remove metal residues, thereby streamlining the manufacturing process and reducing overall production costs while maintaining high product quality standards required for pharmaceutical applications.
Mechanistic Insights into Iodine-Catalyzed Triazole Formation
The reaction mechanism begins with iodine-mediated oxidation of aryl ethyl ketones in DMSO to form aryl diketones through a Kornblum-type oxidation process. This intermediate then undergoes condensation with trifluoroethylimide hydrazide to form a hydrazone intermediate through dehydration. The subsequent cyclization step is facilitated by the iodine catalyst in combination with sodium dihydrogen phosphate and pyridine as co-catalysts under thermal conditions. The phosphate buffer system maintains optimal pH conditions while pyridine acts as both a base and ligand to facilitate the cyclization process. This carefully orchestrated sequence enables the formation of the triazole ring system with precise regioselectivity at the 3,4,5 positions while incorporating the valuable trifluoromethyl group that enhances the pharmacological properties of the final compounds.
The impurity profile of this synthetic route is significantly improved compared to traditional methods due to the absence of transition metal catalysts that typically generate difficult-to-remove impurities. The reaction pathway avoids common side reactions associated with metal-catalyzed processes such as homocoupling or over-reduction products. The phosphate buffer system helps control reaction pH to minimize acid or base-catalyzed decomposition pathways that could generate impurities. The purification process described in the patent utilizes standard column chromatography techniques that effectively separate the desired triazole products from minor byproducts formed during the reaction sequence. This controlled impurity profile is particularly valuable for pharmaceutical applications where strict purity specifications must be met for regulatory approval and clinical safety.
How to Synthesize Triazole Intermediates Efficiently
This patented methodology represents a significant advancement in the synthesis of trifluoromethyl-containing triazole compounds that are critical building blocks in modern pharmaceutical development. The process eliminates traditional barriers associated with metal-catalyzed approaches while maintaining excellent yields and purity profiles across diverse substrate combinations. By operating under standard atmospheric conditions without requiring specialized equipment or handling procedures, this method offers substantial operational advantages for manufacturing environments. The detailed standardized synthesis steps provided below outline the precise methodology for achieving optimal results when implementing this innovative approach in commercial production settings.
- Prepare aryl ethyl ketone solution with iodine in DMSO at 90-110°C for 4-6 hours
- Add trifluoroethylimide hydrazide with sodium dihydrogen phosphate and pyridine
- Heat reaction mixture to 110-130°C for 12-20 hours followed by standard purification
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic route addresses critical pain points in pharmaceutical intermediate supply chains by offering a more sustainable and cost-effective manufacturing solution that aligns with modern procurement priorities. The elimination of transition metal catalysts creates substantial operational advantages while maintaining high product quality standards required for pharmaceutical applications. This methodology provides procurement teams with greater flexibility in supplier selection while ensuring consistent supply chain performance through simplified manufacturing requirements that reduce vulnerability to raw material shortages.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts significantly reduces raw material costs while simultaneously simplifying downstream processing requirements by avoiding metal contamination concerns. The use of readily available starting materials at favorable stoichiometric ratios creates additional cost advantages through reduced material waste and higher atom economy. The simplified purification process eliminates specialized equipment requirements for metal removal, resulting in substantial capital expenditure savings during facility setup and operation.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials with broad supplier networks reduces vulnerability to single-source dependencies that can disrupt production schedules. The process operates under standard atmospheric conditions without requiring specialized handling procedures or controlled environments that could create production bottlenecks during scale-up. This operational simplicity enables more predictable manufacturing timelines and reduces the risk of production delays caused by equipment failures or environmental control issues.
- Scalability and Environmental Compliance: The demonstrated scalability from laboratory to commercial production volumes without requiring significant process modifications provides confidence in reliable supply chain performance at all production scales. The elimination of toxic heavy metals from the manufacturing process substantially reduces environmental impact while simplifying waste treatment requirements. The simplified process flow with fewer unit operations creates opportunities for more efficient resource utilization and reduced energy consumption during commercial manufacturing.
Frequently Asked Questions (FAQ)
The following questions and answers address common technical and commercial concerns regarding the implementation of this patented synthetic methodology for pharmaceutical intermediate production. These insights are derived directly from the technical details and experimental data presented in Chinese Patent CN113105402B to provide accurate information for procurement and technical evaluation teams.
Q: Why is this iodine-catalyzed method superior to traditional metal-catalyzed approaches?
A: This method eliminates the need for expensive and toxic heavy metal catalysts while maintaining high reaction efficiency and purity levels required for pharmaceutical applications.
Q: How does this process ensure consistent high purity for pharmaceutical intermediates?
A: The reaction mechanism avoids metal contamination pathways and the purification process is designed to remove potential impurities through standard column chromatography techniques.
Q: What are the scalability advantages of this synthetic route?
A: The process operates under standard atmospheric conditions without requiring anhydrous or oxygen-free environments, making it readily scalable from laboratory to commercial production volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazole Intermediate Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs and advanced analytical capabilities. This patented iodine-catalyzed methodology represents an ideal candidate for immediate implementation within our manufacturing portfolio due to its operational simplicity and excellent scalability profile that aligns perfectly with our technical capabilities. We have successfully implemented similar transition-metal-free processes across multiple product lines while maintaining consistent quality standards required by global pharmaceutical clients through our comprehensive quality management system.
For procurement teams seeking to optimize their supply chain performance for triazole-based intermediates, we offer a Customized Cost-Saving Analysis that demonstrates how this innovative methodology can reduce total cost of ownership while improving supply chain reliability. Contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your manufacturing requirements and quality specifications.
