Advanced Ruthenium-Catalyzed Synthesis of 3-Acylpyrroles for Pharmaceutical Intermediates
Introduction to Patent CN113636968B: A Breakthrough in Heterocyclic Synthesis
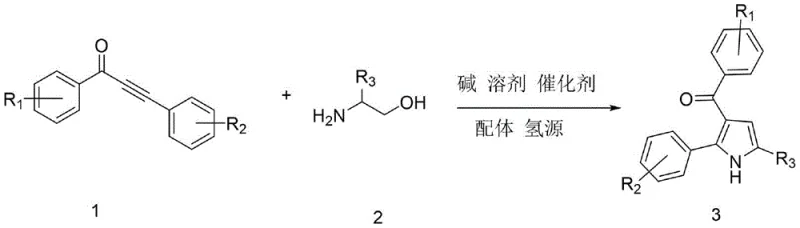
The pharmaceutical industry constantly seeks more efficient pathways to construct privileged scaffolds, and the 3-acylpyrrole motif stands out as a critical building block for bioactive molecules ranging from HDAC inhibitors to HMG-CoA reductase inhibitors. Patent CN113636968B, published in early 2023, introduces a transformative synthetic methodology that addresses long-standing inefficiencies in constructing these valuable nitrogen heterocycles. This innovation leverages a sophisticated ruthenium-catalyzed transfer hydrogenation strategy, converting readily available alpha-amino alcohols and alpha,beta-unsaturated alkynones into high-purity 3-acylpyrroles through a seamless one-pot cascade. For R&D directors and procurement specialists alike, this patent represents a significant leap forward, offering a route that bypasses the need for hazardous oxidants and expensive palladium catalysts, thereby aligning perfectly with modern green chemistry principles and cost-reduction mandates in fine chemical manufacturing.
The core brilliance of this technology lies in its dual functionality: it achieves both the formation of the pyrrole ring and the installation of the acyl group in a single operational sequence. By utilizing ethanol not merely as a solvent but as a sacrificial hydrogen donor, the process achieves exceptional atom economy. This approach fundamentally alters the economic landscape of producing these intermediates, positioning it as a highly attractive option for any organization seeking a reliable pharmaceutical intermediate supplier capable of delivering complex scaffolds with superior purity profiles and reduced environmental footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-acylpyrroles has been plagued by significant operational and economic hurdles that hinder large-scale adoption. Traditional routes often rely on stoichiometric oxidants such as IBX (2-Iodoxybenzoic acid) to mediate intramolecular oxidative cyclization. While effective on a milligram scale, these reagents are prohibitively expensive for tonnage production and generate substantial amounts of iodine-containing waste, creating severe disposal challenges and escalating environmental compliance costs. Furthermore, alternative methods employing palladium catalysis, while powerful, introduce the persistent risk of heavy metal residues in the final active pharmaceutical ingredient (API), necessitating costly and time-consuming purification steps to meet stringent regulatory limits. Additionally, many classical approaches require multi-step sequences involving sensitive intermediates like N-hydroxyalkylenamines or unstable enamines, which demand strict anhydrous conditions and cryogenic temperatures, drastically increasing energy consumption and operational complexity.
The Novel Approach
In stark contrast, the methodology disclosed in CN113636968B offers a streamlined, robust alternative that directly tackles these pain points. By employing a dodecacarbonyl triruthenium catalyst system coupled with a 4-methyl-1,10-phenanthroline ligand, the reaction proceeds via a "borrowing hydrogen" mechanism. This elegant pathway allows the alpha-amino alcohol to undergo dehydrogenation in situ, generating the reactive amino-ketone species which then condenses with the alkynone. The hydrogen removed in the first step is subsequently returned to the system to finalize the aromatization of the pyrrole ring. This internal redox neutrality means no external high-pressure hydrogen gas is required, enhancing process safety. The result is a simplified workflow that operates in a single vessel, significantly reducing solvent usage, labor hours, and the overall carbon footprint associated with the manufacturing of these critical heterocyclic intermediates.
Mechanistic Insights into Ru-Catalyzed Transfer Hydrogenation Cyclization
For the technical team evaluating process feasibility, understanding the mechanistic underpinnings is crucial for troubleshooting and optimization. The reaction initiates with the coordination of the ruthenium catalyst to the hydroxyl group of the alpha-amino alcohol. Under the initial heating phase at 80°C, the catalyst facilitates the dehydrogenation of the alcohol to an amino-ketone, simultaneously forming a ruthenium-hydride species. This activated ketone then undergoes a condensation reaction with the carbonyl group of the alpha,beta-unsaturated alkynone, forming an enamine intermediate. The presence of the base, typically potassium carbonate, is vital here to neutralize the proton released during condensation and to maintain the catalytic cycle. As the temperature is ramped to 150°C, the ruthenium-hydride species delivers the stored hydrogen to the triple bond of the alkynone moiety within the intermediate, triggering a 5-endo-dig cyclization. This step is the rate-determining phase where the pyrrole ring closes, followed by tautomerization to yield the stable aromatic 3-acylpyrrole product.
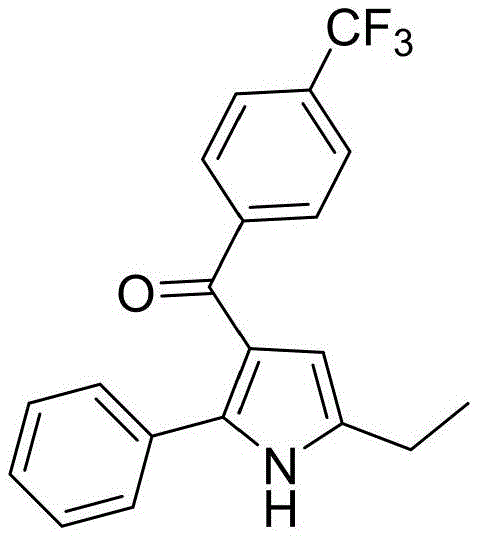
A critical advantage of this mechanism is its exceptional functional group tolerance, which is paramount for synthesizing diverse libraries for drug discovery. The mild basic conditions and the specific electronic properties of the phenanthroline ligand protect sensitive functionalities from degradation. For instance, the system successfully accommodates strong electron-withdrawing groups like trifluoromethyl (-CF3) and halogens without inducing side reactions such as dehalogenation or hydrolysis. This robustness is exemplified by the successful synthesis of derivatives like compound 3d, which bears a para-trifluoromethyl group, demonstrating that the catalyst system does not interfere with electrophilic sites on the aromatic rings. Such versatility ensures that the process can be adapted for a wide array of substrates, making it a universal platform for the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 3-Acylpyrrole Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and minimize impurities. The patent outlines a specific protocol where the molar ratio of the alkynone to the amino alcohol is maintained at 1:1, ensuring complete conversion without excess reagent waste. The catalyst loading is optimized at a mere 2 mol% (0.02:1 ratio), which strikes a balance between reaction rate and cost efficiency. The process begins with a pre-stirring phase at 80°C for 1.5 hours under nitrogen protection, allowing the catalyst to activate and the initial dehydrogenation to occur safely before the system is subjected to the higher thermal stress of 150°C. This two-stage temperature profile is essential; jumping immediately to high temperatures can lead to polymerization of the alkynone or decomposition of the amino alcohol. Following the 18-hour reaction period, the workup is remarkably simple, involving dilution, filtration to remove insoluble salts, and standard column chromatography, avoiding the need for complex aqueous extractions or specialized scavengers.
- Charge reactor with alpha-amino alcohol, alpha,beta-unsaturated alkynone, Ru3(CO)12 catalyst, ligand, base, and solvent under nitrogen.
- Stir mixture at 80°C for 1.5 hours to initiate catalytic activation and pre-reaction equilibration.
- Raise temperature to 150°C and maintain for 18 hours to complete the cyclization and hydrogen transfer, followed by purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented technology offers compelling arguments for supply chain optimization and cost containment. The shift from stoichiometric oxidants to a catalytic hydrogen transfer system fundamentally changes the cost structure of the raw materials. Ethanol, serving as the hydrogen source, is a commodity chemical with stable pricing and global availability, unlike specialized oxidants which are subject to supply volatility. Furthermore, the elimination of heavy metals like palladium removes the necessity for expensive metal scavenging resins and the associated validation testing for residual metals, which are significant hidden costs in API manufacturing. This simplification of the downstream processing directly translates to shorter batch cycles and higher throughput in multipurpose plants, enhancing the overall asset utilization rate for contract manufacturing organizations.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the drastic reduction in reagent costs and waste treatment expenses. By replacing expensive oxidants and precious metal catalysts with a low-loading ruthenium system and cheap ethanol, the direct material cost per kilogram of product is significantly lowered. Additionally, the one-pot nature of the reaction eliminates the isolation and purification of unstable intermediates, reducing solvent consumption and labor intensity. This streamlined process flow allows for substantial cost savings in utility consumption, as there is no need for cryogenic cooling or high-pressure hydrogenation equipment, making it accessible for facilities with standard glass-lined or stainless steel reactors.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of widely available starting materials. Alpha-amino alcohols and alkynones are commodity building blocks produced by numerous chemical suppliers globally, reducing the risk of single-source dependency. The robustness of the reaction conditions—tolerating moisture and oxygen to a reasonable extent after the initial nitrogen purge—means that the process is less prone to batch failures due to minor environmental fluctuations. This reliability ensures consistent delivery schedules, a critical factor for pharmaceutical companies managing tight clinical trial timelines or commercial launch windows where delay penalties can be severe.
- Scalability and Environmental Compliance: Scaling this chemistry from gram to ton scale is straightforward due to the absence of exothermic hazards associated with strong oxidants. The reaction operates at atmospheric pressure, removing the safety risks and capital expenditure associated with high-pressure autoclaves. From an environmental standpoint, the atom economy is superior, and the waste stream is predominantly organic solvents and inorganic salts, which are easier to treat than heavy metal sludge or iodine waste. This alignment with green chemistry metrics facilitates smoother regulatory approvals and supports corporate sustainability goals, adding intangible value to the supply chain partnership.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this synthesis method, we have compiled answers to common inquiries regarding its implementation and scope. These insights are derived directly from the experimental data and claims within the patent documentation, providing a transparent view of what partners can expect when adopting this technology for their pipeline. Understanding these nuances helps in making informed decisions about process integration and vendor selection.
Q: What are the key advantages of this Ru-catalyzed method over traditional oxidative cyclization?
A: Unlike traditional methods requiring stoichiometric oxidants like IBX or harsh acidic conditions, this patent utilizes a borrowing hydrogen strategy with ethanol. This eliminates toxic waste streams, reduces heavy metal contamination risks associated with Palladium, and operates under neutral to mild basic conditions, significantly improving the impurity profile and safety for large-scale production.
Q: Is the catalyst system cost-effective for industrial scale-up?
A: Yes. The process uses Dodecacarbonyl triruthenium (Ru3(CO)12) at a low loading ratio of 0.02:1 relative to the substrate. Combined with the use of inexpensive ethanol as both solvent component and hydrogen source, the overall catalyst and reagent costs are substantially lower than precious metal alternatives, making it highly viable for cost reduction in API manufacturing.
Q: What is the functional group tolerance of this synthesis?
A: The method demonstrates excellent compatibility with diverse substituents including halogens (Fluorine), electron-withdrawing groups (Trifluoromethyl, Cyano), and electron-donating groups (Methyl, Methoxy). This broad scope allows for the direct synthesis of complex, multi-substituted pyrrole scaffolds required for modern drug discovery without needing extensive protecting group strategies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Acylpyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN113636968B for accelerating drug development programs. As a dedicated CDMO partner, we possess the technical expertise to rapidly translate such patent literature into robust, GMP-compliant manufacturing processes. Our engineering team is adept at optimizing catalytic cycles and scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify that every batch of 3-acylpyrrole intermediate meets the exacting standards required for downstream API synthesis.
We invite you to leverage our technical capabilities to evaluate the feasibility of this route for your specific project requirements. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this catalytic method versus your current supply chain. We encourage you to contact our technical procurement team today to request specific COA data for similar heterocyclic compounds and to discuss detailed route feasibility assessments tailored to your molecule’s unique structural challenges.
