Advanced Ruthenium-Catalyzed Synthesis of 3-Acylpyrrole Intermediates for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing complex heterocyclic scaffolds, particularly those serving as critical building blocks for active pharmaceutical ingredients (APIs). Patent CN113636968B introduces a groundbreaking synthetic strategy for 3-acylpyrrole compounds, a privileged structural motif found in numerous bioactive molecules including histone deacetylase inhibitors and HMG-CoA reductase inhibitors. This innovation addresses long-standing challenges in heterocyclic chemistry by employing a ruthenium-catalyzed transfer hydrogenation cascade that merges alpha-amino alcohols and alpha,beta-unsaturated alkynones in a single operational sequence. For R&D directors and process chemists, this patent represents a significant leap forward in step economy, bypassing the need for pre-functionalized intermediates or hazardous oxidizing agents that typically plague traditional pyrrole syntheses. The methodology not only streamlines the synthetic route but also aligns perfectly with modern green chemistry principles, making it an attractive candidate for industrial adoption by reliable pharmaceutical intermediate suppliers aiming to optimize their manufacturing portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 3-acylpyrrole derivatives has relied on multi-step sequences that are often fraught with inefficiencies and safety concerns. Traditional approaches frequently involve the use of stoichiometric amounts of toxic oxidants, such as IBX (2-iodoxybenzoic acid), which pose significant handling risks and generate substantial waste streams that complicate downstream processing. Furthermore, palladium-catalyzed methods, while effective, often require expensive ligands and stringent anhydrous conditions, driving up the cost of goods sold (COGS) and limiting their feasibility for large-scale production. Other classical routes involving beta-amino enones or alpha-amino ketones typically suffer from poor regioselectivity and require harsh acidic or basic conditions that can degrade sensitive functional groups on the substrate. These limitations create bottlenecks in the supply chain, leading to extended lead times and inconsistent batch quality, which are critical pain points for procurement managers overseeing the sourcing of high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN113636968B utilizes a sophisticated yet operationally simple one-pot tandem reaction driven by a triruthenium dodecacarbonyl catalyst system. This novel approach leverages the concept of borrowing hydrogen, where the catalyst temporarily dehydrogenates the amino alcohol to form a reactive amino carbonyl species in situ, which then undergoes condensation and cyclization with the alkynone. The elegance of this method lies in its ability to utilize ethanol not merely as a solvent but as a sacrificial hydrogen donor, thereby regenerating the catalyst and completing the redox cycle without the need for external high-pressure hydrogen gas. This results in a process that is inherently safer, more atom-economical, and significantly easier to scale. By integrating the oxidation and cyclization steps into a single vessel, the method drastically reduces solvent consumption and purification time, offering a compelling value proposition for cost reduction in API manufacturing.
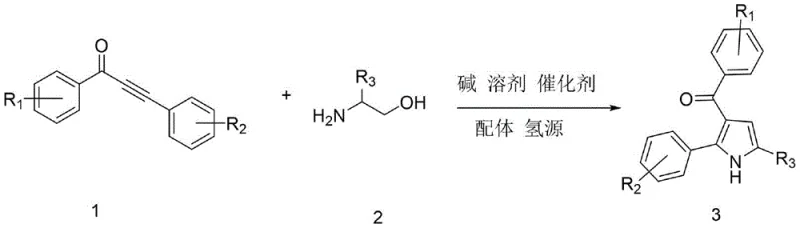
Mechanistic Insights into Ru-Catalyzed Transfer Hydrogenation Cyclization
The core of this synthetic breakthrough is the intricate catalytic cycle mediated by the ruthenium complex, which facilitates the sequential dehydrogenation and hydrogenation events with high fidelity. The mechanism initiates with the coordination of the alpha-amino alcohol to the ruthenium center, followed by beta-hydride elimination to generate an unstable amino-ketone intermediate and a ruthenium-hydride species. This transient amino-ketone immediately reacts with the electron-deficient alkyne moiety of the unsaturated ketone via a Michael-type addition, setting the stage for intramolecular cyclization. The presence of the 4-methyl-1,10-phenanthroline ligand is crucial here, as it stabilizes the active catalytic species and modulates the electronic properties of the metal center to favor the desired cyclization pathway over competing side reactions. This precise control over the reaction trajectory ensures high regioselectivity for the 3-acyl substitution pattern, minimizing the formation of isomeric impurities that are notoriously difficult to separate in pyrrole chemistry.
Furthermore, the final aromatization step is driven by the transfer of hydride from the ruthenium-hydride complex back to the dihydropyrrole intermediate, restoring aromaticity and releasing the final 3-acylpyrrole product. This internal redox balance is what grants the reaction its exceptional atom economy, as no external oxidants or reductants are consumed in the net transformation. From an impurity control perspective, the mild reaction conditions (80°C to 150°C) and the specific ligand environment prevent the degradation of sensitive substituents such as halogens or trifluoromethyl groups, which are common in medicinal chemistry scaffolds. The robustness of this catalytic system allows for a broad substrate scope, accommodating various electronic and steric environments on both the amine and alkyne components, as illustrated by the diverse range of successful examples in the patent data. This mechanistic understanding provides R&D teams with the confidence to adapt the protocol for novel analogues without extensive re-optimization.
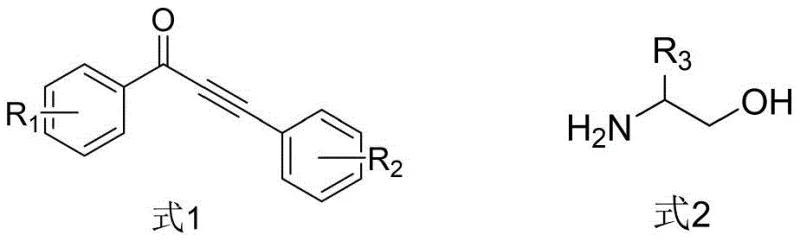
How to Synthesize 3-Acylpyrrole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized parameters defined in the patent to ensure maximum yield and purity. The process begins with the precise charging of reagents into a Schlenk tube or equivalent pressure-rated reactor under an inert nitrogen atmosphere to prevent catalyst deactivation by oxygen. The specific molar ratios, particularly the 1:1 ratio of the alkynone to the amino alcohol and the catalytic loading of the ruthenium complex, are critical for driving the equilibrium towards the product. The temperature profile is equally important, starting with a lower temperature phase to facilitate the initial dehydrogenation and condensation, followed by a higher temperature phase to promote the cyclization and aromatization steps. Detailed standard operating procedures regarding the workup, which involves simple filtration and distillation followed by column chromatography, are essential for reproducibility. For a comprehensive guide on the exact experimental conditions and purification protocols, please refer to the standardized synthesis steps outlined below.
- Combine alpha-amino alcohol, alpha,beta-unsaturated alkynone, triruthenium dodecacarbonyl catalyst, 4-methyl-1,10-phenanthroline ligand, ethanol, base, and solvent in a reactor.
- Stir the mixture at 80°C under nitrogen protection for 1.5 hours, then increase temperature to 150°C and react for 18 hours.
- Cool to room temperature, dilute, filter, distill under reduced pressure, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this ruthenium-catalyzed methodology offers tangible strategic advantages that extend beyond mere technical feasibility. The elimination of expensive and hazardous oxidizing agents like IBX directly translates to a reduction in raw material costs and a simplification of waste disposal logistics, which are often hidden cost drivers in fine chemical manufacturing. Moreover, the use of ethanol as a hydrogen source removes the requirement for specialized high-pressure hydrogenation equipment, allowing the reaction to be performed in standard glass-lined or stainless steel reactors available in most multipurpose facilities. This flexibility significantly enhances supply chain reliability by reducing dependency on specialized infrastructure and enabling faster technology transfer between different manufacturing sites. The simplicity of the workup procedure, which avoids complex aqueous extractions or cryogenic conditions, further contributes to shorter cycle times and improved throughput, ensuring a steady flow of high-purity intermediates to downstream formulation units.
- Cost Reduction in Manufacturing: The economic benefits of this process are primarily derived from the high atom economy and the use of commodity chemicals as reagents. By replacing stoichiometric oxidants with a catalytic system that recycles hydrogen internally, the process minimizes the consumption of expensive reagents and reduces the volume of chemical waste generated per kilogram of product. Additionally, the high efficiency of the ruthenium catalyst means that lower loadings can be used while maintaining excellent conversion rates, further driving down the cost of catalyst consumption. The simplified purification process also reduces the demand for chromatography media and solvents, leading to substantial cost savings in the downstream processing phase. These factors combined create a leaner manufacturing model that is highly resilient to fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as alpha-amino alcohols and substituted alkynones ensures a stable and secure supply chain, as these precursors are produced by multiple global vendors. The robustness of the reaction conditions, which tolerate a wide range of functional groups and moisture levels better than traditional organometallic methods, reduces the risk of batch failures due to minor variations in reagent quality. This tolerance translates to higher first-pass yields and more predictable production schedules, allowing supply chain planners to maintain lower safety stocks without compromising on delivery commitments. Furthermore, the scalability of the one-pot process means that production volumes can be ramped up quickly to meet surges in demand without the need for complex process redesigns or additional unit operations.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this method aligns well with increasingly stringent global standards for sustainable chemical manufacturing. The absence of heavy metal oxidants and the use of ethanol, a green solvent, significantly reduce the environmental footprint of the synthesis, simplifying the permitting process for new production lines. The reduced generation of hazardous waste lowers the costs associated with waste treatment and disposal, while the energy-efficient temperature profile minimizes the carbon footprint of the operation. These sustainability credentials are becoming increasingly important for pharmaceutical companies aiming to meet their corporate social responsibility goals and comply with green chemistry mandates, making this technology a future-proof choice for long-term production partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route, based on the detailed data provided in the patent literature. Understanding these nuances is vital for technical teams evaluating the feasibility of adopting this method for their specific pipeline candidates. The answers reflect the proven capabilities of the technology as demonstrated in the experimental examples, providing a realistic expectation of performance and operational requirements. For further customization or specific data regarding your target molecule, our technical team is prepared to conduct a detailed feasibility assessment.
Q: What is the primary catalyst system used in this synthesis?
A: The process utilizes triruthenium dodecacarbonyl (Ru3(CO)12) as the catalyst, paired with 4-methyl-1,10-phenanthroline as the ligand, facilitating efficient transfer hydrogenation.
Q: What serves as the hydrogen source in this reaction?
A: Ethanol acts as the hydrogen donor in this transfer hydrogenation protocol, eliminating the need for high-pressure external hydrogen gas and enhancing operational safety.
Q: How does this method improve upon traditional pyrrole synthesis?
A: Unlike conventional methods requiring toxic oxidants or harsh conditions, this one-pot approach offers superior atom economy, milder conditions, and easier purification, significantly reducing production costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Acylpyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development and commercialization. Our team of expert process chemists has extensively evaluated the ruthenium-catalyzed transfer hydrogenation route described in CN113636968B and confirmed its potential for robust commercial manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial supply is seamless and risk-free. Our state-of-the-art facilities are equipped with the necessary infrastructure to handle air-sensitive catalytic reactions safely, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of 3-acylpyrrole intermediate meets the highest industry standards. We are committed to delivering not just a chemical product, but a reliable supply solution that supports your long-term strategic goals.
We invite you to collaborate with us to leverage this advanced technology for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data for our existing inventory or to discuss route feasibility assessments for novel derivatives. Let us help you optimize your supply chain and reduce your time-to-market with our proven expertise in complex heterocyclic synthesis.
