Scalable Synthesis of 2-Trifluoromethyl Quinazolinones Using Iron Catalysis for Commercial API Production
Scalable Synthesis of 2-Trifluoromethyl Quinazolinones Using Iron Catalysis for Commercial API Production
The pharmaceutical industry continuously seeks robust and cost-efficient synthetic routes for nitrogen-containing heterocycles, particularly those exhibiting potent biological activities. Patent CN111675662B discloses a groundbreaking preparation method for 2-trifluoromethyl substituted quinazolinone compounds, addressing critical bottlenecks in current manufacturing processes. Quinazolinones are privileged scaffolds found in numerous bioactive molecules, including anticancer, anticonvulsant, and anti-inflammatory agents, where the introduction of a trifluoromethyl group significantly enhances metabolic stability and lipophilicity. This novel approach leverages an iron-catalyzed cyclization strategy that transforms readily available isatin derivatives and trifluoroethylimidoyl chlorides into high-value intermediates. By shifting away from expensive precious metals and harsh conditions, this technology offers a sustainable pathway for producing complex heterocyclic structures essential for modern drug discovery and development pipelines.
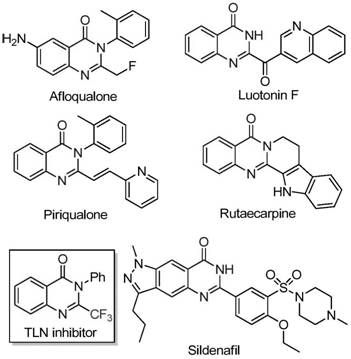
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted quinazolinones has relied heavily on cyclization reactions involving synthons like trifluoroacetic anhydride or ethyl trifluoroacetate with substrates such as anthranilamide or isatoic anhydride. These traditional pathways are often plagued by severe reaction conditions that require stringent anhydrous environments or extremely high temperatures, leading to safety concerns and energy inefficiencies in large-scale operations. Furthermore, the substrate scope in conventional methods is frequently narrow, limiting the ability of medicinal chemists to explore diverse chemical space for structure-activity relationship studies. The reliance on expensive fluorinating reagents and the generation of difficult-to-remove byproducts often result in low overall yields and complicated purification processes, which drastically increase the cost of goods sold for the final active pharmaceutical ingredient.
The Novel Approach
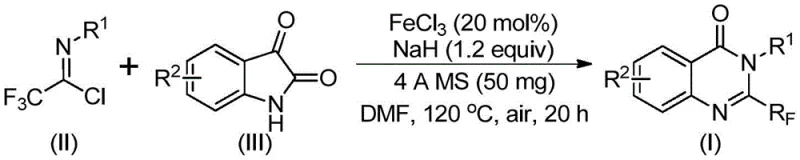
In stark contrast, the methodology described in the patent utilizes a synergistic combination of ferric chloride and sodium hydride to catalyze the reaction between trifluoroethylimidoyl chloride and isatin. This innovative route operates under relatively milder conditions, typically initiating at 40°C before heating to 120°C in dimethylformamide (DMF), which simplifies the thermal management requirements for reactor systems. The use of isatin as a starting material is particularly advantageous due to its commercial availability and low cost compared to specialized anthranilic acid derivatives. Additionally, the reaction demonstrates remarkable tolerance for various functional groups, including halogens and electron-donating groups, enabling the rapid synthesis of a wide library of analogues without the need for protecting group strategies. This flexibility significantly accelerates the lead optimization phase in drug development while maintaining high atom economy.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this transformation lies in the unique ability of the iron catalyst to facilitate a cascade of bond-forming events that construct the quinazolinone core efficiently. Mechanistically, the reaction likely proceeds through an initial alkali-promoted formation of carbon-nitrogen bonds between the trifluoroethylimidoyl chloride and the isatin nitrogen, generating a trifluoroacetamidine intermediate. Subsequently, the ferric chloride species activates the system for a decarbonylation event, which is a critical step that distinguishes this method from traditional condensation reactions. This decarbonylation allows for the extrusion of carbon monoxide and the subsequent cyclization to form the stable six-membered heterocyclic ring. The presence of 4A molecular sieves in the reaction mixture plays a crucial role in scavenging moisture, thereby preventing the hydrolysis of the sensitive imidoyl chloride starting material and ensuring the catalyst remains active throughout the prolonged heating period.
From an impurity control perspective, this mechanism offers distinct advantages over acid-catalyzed alternatives which often lead to polymerization or degradation of the trifluoromethyl group. The specific coordination of the iron center helps direct the regioselectivity of the cyclization, minimizing the formation of isomeric byproducts that are notoriously difficult to separate via crystallization. The use of sodium hydride as a base ensures complete deprotonation of the isatin nitrogen, driving the equilibrium towards the desired product and suppressing side reactions associated with incomplete conversion. Understanding these mechanistic nuances allows process chemists to fine-tune parameters such as catalyst loading and stoichiometry to maximize purity profiles, which is a critical requirement for regulatory compliance in pharmaceutical manufacturing.
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the order of addition and thermal profiling to ensure reproducibility and safety. The protocol dictates mixing ferric chloride, sodium hydride, molecular sieves, and the organic substrates in an aprotic solvent like DMF, followed by a staged heating process that begins at a lower temperature to form intermediates before ramping up for cyclization. Detailed standardized operating procedures regarding the handling of sodium hydride and the workup involving silica gel mixing are essential for consistent results. For a comprehensive guide on the specific molar ratios, solvent volumes, and purification techniques validated in the patent examples, please refer to the technical protocol below.
- Combine ferric chloride (20 mol%), sodium hydride (1.2 equiv), 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin in anhydrous DMF solvent within a Schlenk tube.
- Stir the reaction mixture at 40°C for approximately 8 to 10 hours to initiate the formation of intermediate bonds.
- Increase the temperature to 120°C and continue reacting under air for 18 to 20 hours to complete the cyclization, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iron-catalyzed route presents a compelling value proposition centered on cost stability and raw material security. By eliminating the dependency on scarce and volatile precious metal catalysts, manufacturers can insulate their production costs from fluctuations in the commodities market associated with palladium or rhodium. The utilization of commodity chemicals like isatin and simple aromatic amines ensures a robust supply chain with multiple qualified vendors globally, reducing the risk of single-source bottlenecks that can halt production lines. Furthermore, the simplified downstream processing reduces the consumption of silica gel and solvents during purification, contributing to a leaner manufacturing footprint and lower waste disposal costs.
- Cost Reduction in Manufacturing: The substitution of expensive catalysts with ferric chloride, a ubiquitous and low-cost industrial chemical, drives a substantial decrease in the direct material cost per kilogram of the intermediate. The high conversion rates observed in the patent data suggest that less starting material is wasted, improving the overall mass balance and reducing the burden on waste treatment facilities. Additionally, the ability to run the reaction in air rather than requiring inert gas atmospheres simplifies the reactor setup and lowers utility costs associated with nitrogen or argon purging, leading to significant operational expenditure savings.
- Enhanced Supply Chain Reliability: Sourcing strategies benefit greatly from the use of widely available starting materials that are produced on a multi-ton scale by numerous chemical suppliers worldwide. This abundance ensures that lead times for raw materials remain short and predictable, allowing for more agile inventory management and just-in-time production models. The robustness of the reaction conditions also means that the process is less susceptible to minor variations in reagent quality, providing a buffer against supply chain disruptions and ensuring consistent output quality even when sourcing from different batches of raw materials.
- Scalability and Environmental Compliance: The transition from bench-scale to commercial production is facilitated by the absence of highly toxic reagents and the use of standard organic solvents that are easily recovered and recycled. The process generates fewer hazardous byproducts compared to traditional fluorination methods, aligning with increasingly stringent environmental regulations and corporate sustainability goals. This green chemistry profile not only reduces the cost of environmental compliance but also enhances the marketability of the final product to eco-conscious pharmaceutical partners who prioritize sustainable supply chains in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation to provide clarity on feasibility and performance. Understanding these details is crucial for technical teams evaluating the integration of this route into existing manufacturing portfolios.
Q: What are the primary advantages of using FeCl3 over traditional catalysts for quinazolinone synthesis?
A: The use of ferric chloride (FeCl3) offers significant economic and operational benefits compared to precious metal catalysts. It is inexpensive, readily available commercially, and demonstrates high catalytic efficiency in promoting the decarbonylation and cyclization steps required for forming the quinazolinone core.
Q: Can this synthesis method tolerate diverse functional groups on the aromatic ring?
A: Yes, the protocol exhibits excellent functional group tolerance. The reaction successfully accommodates various substituents such as methyl, halogen (fluorine, bromine, chlorine), methoxy, and nitro groups at ortho-, meta-, or para-positions, allowing for the design of diverse molecular scaffolds.
Q: Is the reaction scalable for industrial production of pharmaceutical intermediates?
A: The method is designed with scalability in mind, utilizing cheap and easily obtained starting materials like isatin and trifluoroethylimidoyl chloride. The simple post-treatment involving filtration and standard column chromatography facilitates potential expansion from gram-level laboratory synthesis to larger commercial batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient heterocycle synthesis in accelerating drug development timelines. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from discovery to market. Our state-of-the-art facilities are equipped to handle the specific thermal and safety requirements of iron-catalyzed reactions, while our rigorous QC labs enforce stringent purity specifications to meet the highest international pharmacopeial standards. We are committed to delivering high-purity quinazolinone derivatives that empower your research and commercial manufacturing needs.
We invite you to engage with our technical team to discuss how this innovative synthesis route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this iron-catalyzed method for your supply chain. Please contact our technical procurement team today to obtain specific COA data for our catalog compounds and to receive detailed route feasibility assessments for your custom synthesis projects.
