Scalable Synthesis of Roxadustat Intermediates via Zinc-Mediated Reduction
Introduction to Advanced Roxadustat Manufacturing
The pharmaceutical landscape for anemia treatment has been significantly reshaped by the advent of Hypoxia Inducible Factor-Prolyl Hydroxylase Inhibitors (HIF-PHIs), with Roxadustat standing as a premier example of this therapeutic class. As global demand for effective erythropoiesis-stimulating agents grows, the imperative for robust, scalable, and cost-efficient synthetic routes becomes paramount for supply chain stability. Patent CN111533691A discloses a groundbreaking preparation method that addresses critical bottlenecks in the traditional synthesis of Roxadustat, offering a pathway that is not only chemically elegant but also commercially superior. This technical insight report analyzes the proprietary methodology which replaces hazardous hydrogenation steps with a mild, zinc-mediated reduction, thereby enhancing both safety profiles and production throughput for reliable API intermediate suppliers.
The core innovation lies in the strategic manipulation of the isoquinoline scaffold. By utilizing a Mannich-type reaction followed by a specific reductive demethylation, the process achieves high regioselectivity and yield. The structural integrity of the final molecule, as depicted below, relies heavily on the precise installation of the methyl group at the 1-position of the isoquinoline ring, a transformation that has historically been fraught with inefficiency.
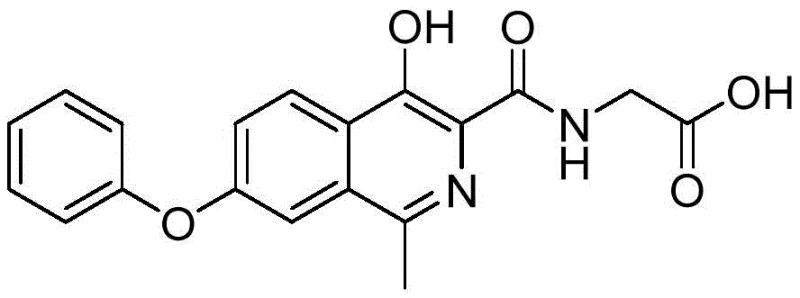
Understanding the molecular architecture is the first step toward mastering its synthesis. The presence of the phenoxy group at the 7-position and the carboxylic acid moiety at the 3-position requires orthogonal protection and deprotection strategies that do not compromise the sensitive heterocyclic nitrogen. The disclosed method navigates these complexities with remarkable efficiency, setting a new benchmark for high-purity pharmaceutical intermediates in the market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthesis routes, such as those disclosed in WO2014/14834, have long relied on catalytic hydrogenation using Palladium on Carbon (Pd/C) to effect the necessary reduction steps. While chemically feasible, this approach presents substantial drawbacks for commercial scale-up of complex pharmaceutical intermediates. The conventional pathway typically suffers from prolonged reaction times, often extending up to 30 hours to reach completion, which severely limits reactor turnover and overall plant capacity. Furthermore, the conversion rates in these traditional methods are frequently suboptimal, hovering around 60-70%, leading to significant material loss and increased waste generation.
Beyond efficiency metrics, the reliance on Pd/C hydrogenation introduces inherent safety and operational risks. The requirement for high-pressure hydrogen gas necessitates specialized equipment and rigorous safety protocols, increasing capital expenditure and operational complexity. Additionally, the removal of trace palladium from the final product to meet stringent regulatory limits adds further downstream processing steps, complicating the purification workflow. These factors collectively contribute to higher manufacturing costs and potential supply chain vulnerabilities.
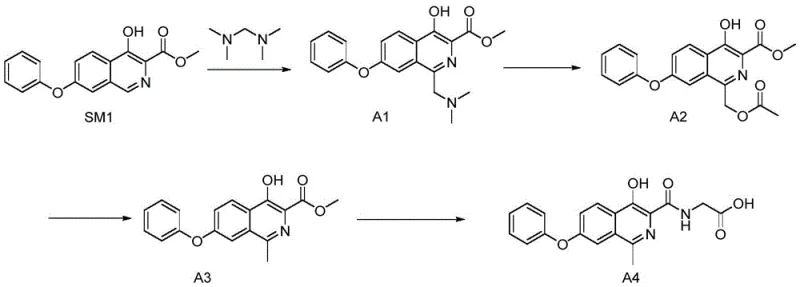
The Novel Approach
In stark contrast, the methodology outlined in CN111533691A introduces a paradigm shift by employing a zinc-mediated reduction in an acidic medium. This novel approach circumvents the need for high-pressure hydrogenation entirely, replacing it with a heterogeneous reduction using zinc powder and hydrochloric acid in glacial acetic acid. This substitution drastically simplifies the operational requirements, allowing the reaction to proceed under atmospheric pressure and mild thermal conditions (50-60°C). The result is a dramatic reduction in reaction time, with the critical reduction step completing in approximately 6 hours, representing a five-fold increase in processing speed compared to legacy methods.
The new route also demonstrates superior atom economy and yield characteristics. By avoiding the harsh conditions of catalytic hydrogenation, the process minimizes side reactions and degradation of the sensitive isoquinoline core. The use of zinc, a plentiful and inexpensive reducing agent, coupled with the simplicity of the workup procedure, translates directly into cost reduction in pharmaceutical intermediates manufacturing. This streamlined process ensures that the production of Roxadustat can be scaled reliably to meet global demand without the bottlenecks associated with traditional catalytic methods.
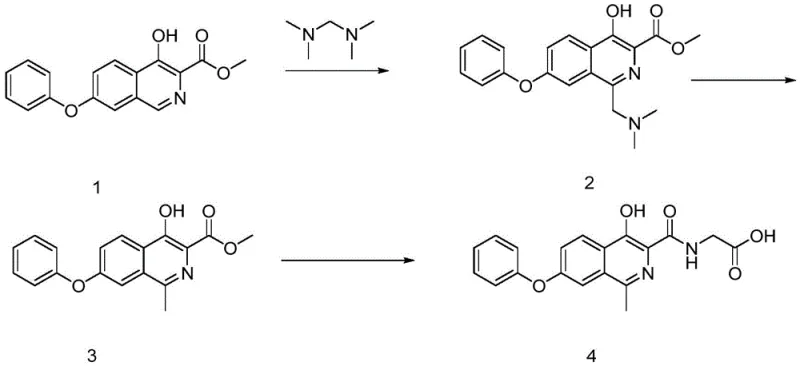
Mechanistic Insights into Zinc-Mediated Reductive Demethylation
The heart of this innovative synthesis lies in the second step: the conversion of the 1-((dimethylamino)methyl) intermediate to the 1-methyl derivative. Mechanistically, this transformation involves the in situ generation of reactive hydrogen species or direct electron transfer from the zinc surface to the iminium ion formed under acidic conditions. The glacial acetic acid serves a dual purpose: it acts as the solvent to solubilize the organic substrate and provides the acidic environment necessary to protonate the dimethylamino group, facilitating its departure as a leaving group. The addition of dilute hydrochloric acid further enhances the reducibility of the zinc by maintaining a steady supply of protons, effectively catalyzing the reduction cycle.
This reductive demethylation is highly selective for the exocyclic C-N bond, leaving the endocyclic aromatic system and the ester functionality intact. The mechanism likely proceeds through a radical or hydride-like transfer from the activated zinc surface to the electrophilic carbon of the iminium intermediate. This specificity is crucial for maintaining the integrity of the 4-hydroxy and 3-carboxylate groups, which are essential for the biological activity of the final API. The ability to achieve this transformation with such high fidelity underscores the sophistication of the reaction design.
Furthermore, the process incorporates a robust purification strategy immediately following the reduction. The reaction mixture is subjected to a liquid-liquid extraction using dichloromethane and water. This step is critical for reducing lead time for high-purity pharmaceutical intermediates by efficiently removing inorganic zinc salts and other polar impurities that could interfere with subsequent steps. By ensuring the intermediate (Formula 3) is of high purity (>96%) before proceeding to the final amidation, the overall yield of the target Roxadustat is maximized, consistently achieving yields in the range of 85-90% for the overall sequence.
How to Synthesize Roxadustat Efficiently
The execution of this synthesis requires precise control over stoichiometry and reaction parameters to ensure reproducibility and safety. The process begins with the formation of the Mannich base, followed by the critical zinc reduction, and concludes with the amidation with glycine. Each step has been optimized to balance reaction kinetics with product stability. For R&D teams looking to implement this technology, understanding the nuances of the zinc activation and the pH control during the reduction phase is vital for success.
- Perform a Mannich reaction on 4-hydroxy-7-phenoxyisoquinoline-3-methyl formate using tetramethylmethanediamine in glacial acetic acid.
- Execute reductive demethylation using Zinc powder and hydrochloric acid in acetic acid to form the methyl-substituted intermediate.
- Conduct amidation with glycine in methanol using sodium methoxide to finalize the Roxadustat structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers tangible strategic benefits beyond mere technical feasibility. The elimination of expensive noble metal catalysts like palladium removes a significant variable cost component and mitigates the risk associated with the volatility of precious metal markets. Moreover, the simplified operational profile reduces the dependency on specialized high-pressure infrastructure, allowing for more flexible manufacturing arrangements and potentially lower capital investment for new production lines.
- Cost Reduction in Manufacturing: The substitution of Pd/C hydrogenation with zinc reduction fundamentally alters the cost structure of the synthesis. Zinc is a commodity metal with a stable and low price point compared to palladium. Additionally, the removal of the hydrogenation step eliminates the need for costly catalyst recovery and regeneration processes, as well as the extensive testing required to ensure residual metal levels comply with ICH Q3D guidelines. This streamlining of the process flow results in substantial cost savings per kilogram of produced intermediate.
- Enhanced Supply Chain Reliability: By relying on widely available reagents such as zinc powder, hydrochloric acid, and acetic acid, the supply chain becomes more resilient. Unlike specialized catalysts which may have long lead times or single-source dependencies, the raw materials for this process are commoditized and globally accessible. This diversification of the supply base ensures continuity of supply even in the face of geopolitical disruptions or logistical constraints, securing the production schedule for downstream API manufacturing.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of high-pressure hydrogen gas make this process inherently safer and easier to scale from pilot plant to commercial tonnage. The waste profile is also improved; zinc salts are generally easier to treat and dispose of than spent noble metal catalysts mixed with organic solvents. This alignment with green chemistry principles not only reduces environmental compliance costs but also enhances the sustainability profile of the final product, a growing priority for multinational pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the technology's capabilities and limitations for potential adopters.
Q: How does the new Zinc-reduction method improve upon traditional Palladium-Carbon hydrogenation?
A: The novel Zinc-mediated reduction eliminates the safety risks and high operational costs associated with high-pressure hydrogenation and expensive Palladium catalysts. It significantly shortens reaction time from approximately 30 hours to roughly 6 hours while improving conversion rates and simplifying the workup procedure.
Q: What represents the critical quality attribute in the intermediate synthesis?
A: The purity of the 4-hydroxy-1-methyl-7-phenoxyisoquinoline-3-carboxylic acid methyl ester intermediate is critical. The patented process utilizes a specific extraction protocol with dichloromethane and water to efficiently remove metal salt inorganic impurities, ensuring the final API meets stringent purity specifications exceeding 99%.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process is explicitly designed for scalability. It operates under mild conditions (50-60°C) without the need for specialized high-pressure equipment, uses readily available reagents like Zinc and acetic acid, and offers a robust yield profile suitable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Roxadustat Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex therapeutics like Roxadustat depends on a partnership grounded in technical excellence and supply chain reliability. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition seamlessly from laboratory bench to full-scale manufacturing. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite you to engage with our technical procurement team to discuss how this advanced zinc-mediated synthesis can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic advantages specific to your volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that your sourcing strategy is built on a foundation of verified quality and proven performance.
