Optimizing Roxadustat Intermediate Production via Safe Halogenation and Methylation Strategies
Optimizing Roxadustat Intermediate Production via Safe Halogenation and Methylation Strategies
The pharmaceutical landscape for treating chronic anemia has been significantly transformed by Hypoxia Inducible Factor Prolyl Hydroxylase Inhibitors (HIF-PHIs), with Roxadustat (FG-4592) standing as a pioneering molecule in this class. As global demand for effective anemia management solutions escalates, the efficiency and safety of supplying high-quality active pharmaceutical ingredients (APIs) and their precursors become paramount. Patent CN110903242A introduces a groundbreaking preparation method for the critical Roxadustat intermediate, 4-hydroxy-1-methyl-7-phenoxy-3-isoquinoline carboxylic ester. This technical disclosure addresses long-standing challenges in heterocyclic synthesis, offering a pathway that balances rigorous safety standards with high-yield production capabilities. By shifting away from hazardous reagents traditionally employed in isoquinoline functionalization, this innovation provides a robust framework for reliable pharmaceutical intermediate supplier networks aiming to secure the supply chain for next-generation therapeutics.
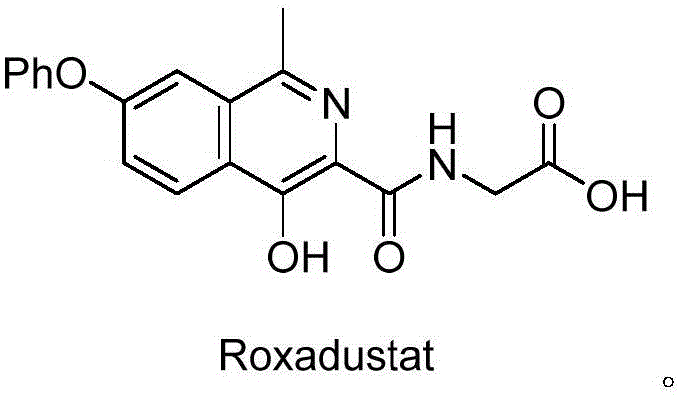
The structural complexity of Roxadustat necessitates precise synthetic control, particularly at the 1-methyl-4-hydroxy-7-phenoxy-3-isoquinoline core. The patent elucidates a refined approach that circumvents the limitations of earlier methodologies, ensuring that the resulting intermediates meet the stringent purity specifications required for clinical applications. For procurement managers and supply chain heads, understanding the underlying chemistry of this patent is crucial, as it directly correlates to reduced operational risks and enhanced cost reduction in API manufacturing. The transition from volatile, dangerous reagents to stable, manageable solids represents a paradigm shift in how complex heterocycles are produced at scale, promising greater stability in lead times and inventory management for downstream drug manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the key intermediate 4-hydroxy-1-methyl-7-phenoxy-3-isoquinoline carboxylic ester has been plagued by significant safety and efficiency hurdles. Prior art methods, such as those disclosed in WO2004108681A1, frequently relied on phosphorus oxybromide (POBr3) for bromination, a reagent known for its extreme corrosivity and violent reaction with water, posing severe safety risks during scale-up production. Furthermore, subsequent methylation steps often utilized n-butyllithium and methyl iodide, requiring cryogenic conditions and presenting pyrophoric hazards that complicate industrial operations. Other routes described in WO2013013609 employed trimethylboron, a substance prone to spontaneous combustion, alongside palladium catalysts in 1,4-dioxane, leading to low yields and substantial safety liabilities. Additionally, methods involving multiple protection and deprotection steps, or the use of expensive oxidants like iodobenzene diacetate, inflated production costs and introduced complex purification burdens that hindered commercial viability.
The Novel Approach
In stark contrast, the methodology presented in CN110903242A offers a streamlined, two-step sequence that prioritizes operator safety and process robustness. The invention replaces hazardous liquid halogenating agents with solid N-halosuccinimides, such as N-bromosuccinimide (NBS), which can be handled safely in standard solvents like dichloromethane or acetonitrile. This halogenation step selectively targets the 1-position of the isoquinoline ring to form the halo-intermediate (Formula V) with high precision. Subsequently, the methylation is achieved through a palladium-catalyzed cross-coupling reaction using stable methylating agents like trimethylboroxine or methylboronic acid. This approach eliminates the need for cryogenic temperatures and pyrophoric reagents, operating instead at moderate temperatures between 90-110°C. The result is a mature and controllable process route that significantly mitigates the safety risks associated with amplified production while delivering superior reaction yields.
Mechanistic Insights into Pd-Catalyzed Methylation and Selective Halogenation
The core of this technological advancement lies in the precise control of electrophilic aromatic substitution and transition metal catalysis. In the initial halogenation phase, the electron-rich isoquinoline ring of Formula IV undergoes selective bromination at the C-1 position. The use of NBS in dichloromethane facilitates a controlled radical or electrophilic mechanism that avoids over-halogenation or degradation of the sensitive phenoxy and ester functionalities. This selectivity is critical for maintaining the integrity of the molecular scaffold, ensuring that the subsequent coupling reaction proceeds without interference from regio-isomers. The patent data indicates that optimizing the molar ratio of the substrate to the halogenating agent (preferably 1:1.05) is essential for maximizing conversion while minimizing impurity formation, a key consideration for R&D directors focused on impurity profiles.
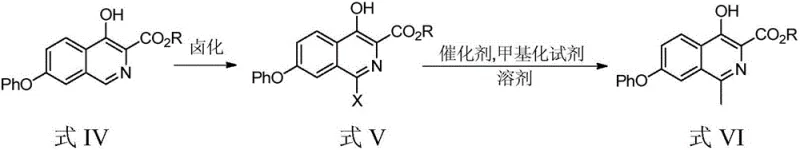
Following halogenation, the transformation of the 1-bromo intermediate (Formula V) to the 1-methyl target (Formula VI) proceeds via a Suzuki-Miyaura type cross-coupling mechanism. The patent highlights the efficacy of using palladium acetate or tetrakis(triphenylphosphine)palladium(0) in conjunction with bulky phosphine ligands like tricyclohexylphosphine. These ligands enhance the oxidative addition of the aryl bromide to the palladium center and facilitate the transmetallation with the organoboron species. The choice of base, such as potassium phosphate heptahydrate or potassium fluoride dihydrate, plays a pivotal role in activating the boron reagent and neutralizing the acid byproduct. This catalytic cycle is highly efficient, as evidenced by yields reaching up to 88.01% with purities exceeding 99.8%. The mechanistic robustness of this system allows for the use of less hazardous solvents like toluene, further enhancing the environmental profile of the synthesis compared to previous methods utilizing dioxane or DMF exclusively.
How to Synthesize 4-hydroxy-1-methyl-7-phenoxy-3-isoquinoline carboxylic ester Efficiently
The practical implementation of this synthesis route involves carefully controlled reaction parameters to ensure reproducibility and safety. The process begins with the dissolution of the isoquinoline precursor in a chlorinated solvent, followed by the slow addition of the solid halogenating agent to manage exotherms. After confirming complete conversion, the intermediate is isolated and subjected to the methylation conditions under an inert nitrogen atmosphere to prevent catalyst deactivation. The detailed standardized synthetic steps, including specific workup procedures involving pH adjustment and extraction protocols, are outlined below to guide process chemists in replicating these high-yield results.
- Perform halogenation on 4-hydroxy-1-hydrogen-7-phenoxy-3-isoquinoline carboxylic ester using N-bromosuccinimide (NBS) in dichloromethane to generate the 1-bromo derivative.
- Conduct methylation reaction on the halo-intermediate using trimethylboroxine or methylboronic acid with a palladium catalyst and phosphate base in toluene.
- Purify the final crude product via extraction and column chromatography to achieve high-purity 4-hydroxy-1-methyl-7-phenoxy-3-isoquinoline carboxylic ester.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for sourcing and logistics, the shift to this novel synthetic route offers profound strategic benefits beyond mere chemical elegance. The elimination of highly regulated, dangerous goods such as n-butyllithium and phosphorus oxybromide simplifies the procurement process and reduces the regulatory burden associated with transporting and storing hazardous materials. This inherently safer chemistry translates directly into enhanced supply chain reliability, as production schedules are less likely to be disrupted by safety incidents or strict handling constraints. Furthermore, the use of commercially available, stable reagents like NBS and trimethylboroxine ensures a consistent supply of raw materials, mitigating the risk of shortages that often plague specialty chemical markets.
- Cost Reduction in Manufacturing: The economic implications of this process are substantial, driven primarily by the simplification of operational requirements and the improvement in overall yield. By removing the need for specialized cryogenic equipment and expensive, hazardous reagents, the capital expenditure and operational costs associated with the manufacturing facility are significantly lowered. The high reaction yield, reported up to 88.01% in optimized examples, means that less raw material is wasted per kilogram of final product, directly improving the cost of goods sold (COGS). Additionally, the simplified purification process, which avoids complex chromatographic separations often needed to remove difficult byproducts from older routes, reduces solvent consumption and waste disposal costs, contributing to substantial cost savings in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: Stability in the supply of critical intermediates is vital for maintaining continuous API production. This method enhances reliability by utilizing reagents that are shelf-stable and widely available from multiple global suppliers, reducing dependency on single-source vendors for exotic chemicals. The robustness of the reaction conditions, which tolerate standard industrial equipment and do not require exotic pressure vessels or extreme temperatures, ensures that the process can be easily transferred between manufacturing sites without significant re-validation. This flexibility allows for a more resilient supply network, capable of adapting to market fluctuations and ensuring reducing lead time for high-purity intermediates needed for clinical and commercial batches.
- Scalability and Environmental Compliance: As the demand for Roxadustat grows, the ability to scale production from kilograms to metric tons without compromising safety or quality is a decisive factor. This process is inherently scalable, having been designed with industrial amplification in mind, avoiding the pitfalls of methods that rely on dangerous oxidants or hydrogenation steps requiring specialized infrastructure. From an environmental perspective, the replacement of toxic solvents and reagents with greener alternatives aligns with increasingly stringent global environmental regulations. The reduction in hazardous waste generation and the potential for solvent recycling contribute to a lower environmental footprint, making this a sustainable choice for commercial scale-up of complex pharmaceutical intermediates.

Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy processes in terms of safety, yield, and operational feasibility.
Q: Why is the new halogenation method safer than traditional POBr3 routes?
A: Traditional methods utilize phosphorus oxybromide (POBr3), which poses significant safety risks due to its corrosive nature and sensitivity to moisture. The patented method employs solid N-bromosuccinimide (NBS) in common solvents like dichloromethane, drastically reducing handling hazards and simplifying waste treatment protocols.
Q: What are the yield improvements in the novel methylation strategy?
A: By utilizing stable methylating agents like trimethylboroxine alongside optimized palladium catalysts and ligands, the process achieves yields up to 88.01% with purity exceeding 99.8%. This contrasts sharply with prior art methods that often suffer from low yields due to side reactions or unstable reagents like trimethylboron.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for industrial scalability. It avoids cryogenic conditions required for n-butyllithium and eliminates the need for specialized hydrogenation equipment. The use of robust reagents and standard temperature ranges (90-110°C) ensures consistent quality and operational safety at the 100 MT scale.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Roxadustat Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of life-saving medications like Roxadustat depends on a partnership grounded in technical excellence and supply security. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory discovery to full-scale manufacturing. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch of 4-hydroxy-1-methyl-7-phenoxy-3-isoquinoline carboxylic ester adheres to the highest industry standards.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals in the competitive pharmaceutical market.
