Advanced Mo/Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial API Production
Advanced Mo/Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial API Production
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for nitrogen-containing heterocycles, particularly those incorporating fluorine motifs which are critical for enhancing metabolic stability and bioavailability. Patent CN113307778A introduces a groundbreaking preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds, a structural scaffold prevalent in numerous high-value drugs such as Sitagliptin and various anticonvulsants. This technology addresses the longstanding challenges in constructing fully substituted triazole rings by utilizing a novel dual-catalyst system involving molybdenum and copper. For R&D directors and procurement specialists, this patent represents a significant opportunity to streamline the supply chain for complex heterocyclic intermediates, offering a pathway that balances high reaction efficiency with operational simplicity.
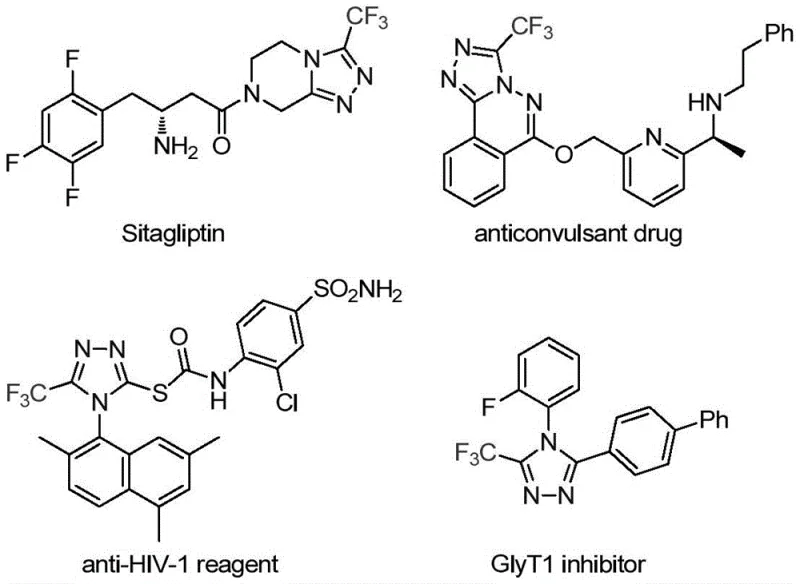
The strategic importance of this synthesis lies in its ability to access diverse substitution patterns that are often difficult to achieve through traditional condensation reactions. By leveraging functionalized isonitriles and trifluoroethylimidoyl chloride, the method bypasses the need for hazardous diazonium salts or unstable hydrazine precursors often found in legacy literature. This shift not only improves the safety profile of the manufacturing process but also opens new avenues for medicinal chemists to explore structure-activity relationships (SAR) with greater flexibility. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic advancements is crucial for maintaining a competitive edge in the global API market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has relied heavily on the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of oxazolinone derivatives. These classical approaches frequently suffer from harsh reaction conditions, requiring strong acids or bases that can degrade sensitive functional groups on the substrate. Furthermore, alternative multi-component reactions involving diazonium salts and trifluorodiazoethane pose significant safety risks due to the explosive nature of diazo compounds, making them less desirable for large-scale commercial production. The limited substrate scope of these older methods often restricts the ability to introduce diverse aryl or alkyl groups at the N-1 and C-5 positions, thereby limiting the chemical space available for drug discovery teams.
The Novel Approach
In stark contrast, the methodology disclosed in CN113307778A utilizes a sophisticated Mo/Cu co-catalytic system to drive a [3+2] cycloaddition between trifluoroethylimidoyl chloride and functionalized isonitrile. This innovative route operates under remarkably mild conditions, typically between 70°C and 90°C, eliminating the thermal stress associated with traditional cyclizations. The use of commercially available starting materials, such as molybdenum hexacarbonyl and cuprous acetate, ensures that the process is both economically viable and easily implementable in standard chemical reactors. This novel approach effectively resolves the regioselectivity issues common in triazole synthesis, delivering the desired 3-trifluoromethyl-1,2,4-triazole core with high precision and minimal byproduct formation.
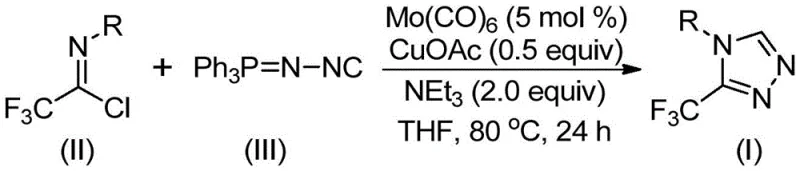
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
The success of this transformation hinges on the synergistic interaction between the molybdenum and copper catalysts. Mechanistically, molybdenum hexacarbonyl acts as a potent activator for the functionalized isonitrile, forming a transient metal-isocyanide complex that enhances the nucleophilicity of the carbon center. Simultaneously, the copper species, likely generated in situ from cuprous acetate, facilitates the activation of the imidoyl chloride electrophile. This dual activation lowers the energy barrier for the initial [3+2] cycloaddition step, allowing the formation of a five-membered ring intermediate under relatively low thermal input. The subsequent elimination of triphenylphosphine oxide, driven by the presence of water or base in the system, aromatizes the ring to yield the final stable triazole product.
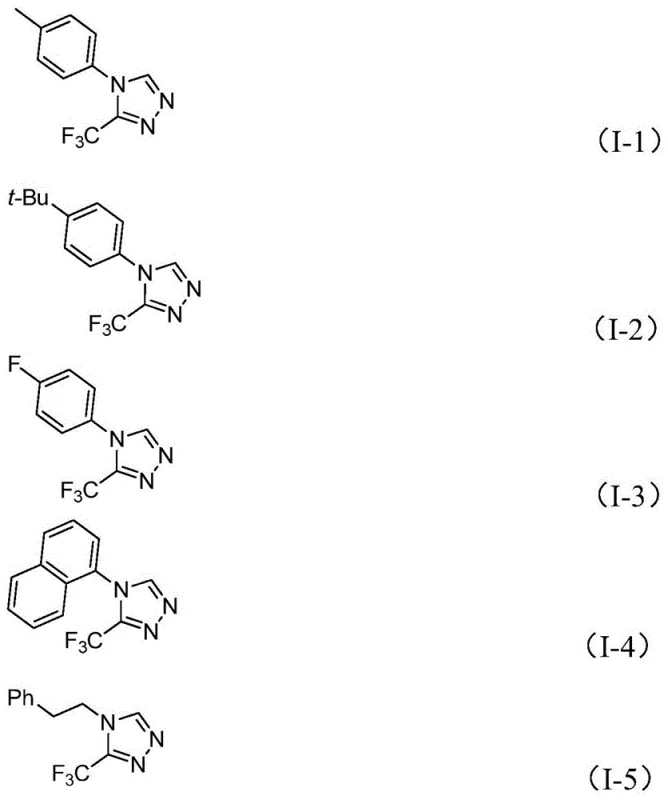
From an impurity control perspective, this mechanism offers distinct advantages over acid-catalyzed condensations. The specificity of the metal-ligand interactions minimizes side reactions such as polymerization of the isonitrile or hydrolysis of the imidoyl chloride. The patent data indicates that the reaction tolerates a wide range of electronic environments on the aromatic ring, including electron-withdrawing groups like nitro and halogens, as well as electron-donating groups like methoxy and alkyl chains. This broad functional group tolerance suggests that the catalytic cycle is robust against steric hindrance and electronic deactivation, ensuring consistent purity profiles across different substrate batches.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis requires careful attention to the stoichiometric balance of the catalysts and the choice of solvent to maximize conversion rates. The protocol involves dissolving the catalysts and base in an aprotic solvent like THF, followed by the sequential addition of the imidoyl chloride and isonitrile components. Maintaining an inert atmosphere is recommended to prevent oxidation of the copper catalyst, although the system demonstrates reasonable stability. The reaction progress is monitored until the starting materials are fully consumed, typically within a 24-hour window, after which standard filtration and chromatographic purification yield the high-purity target compound.
- Combine molybdenum hexacarbonyl (5 mol%), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), and molecular sieves in an organic solvent such as THF.
- Add trifluoroethylimidoyl chloride and the functionalized isonitrile (NIITP) to the reaction mixture under inert atmosphere.
- Heat the reaction mixture to 70-90°C for 18-30 hours, then filter and purify via column chromatography to isolate the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this Mo/Cu co-catalyzed method offers substantial strategic benefits beyond mere chemical efficiency. The reliance on readily available, commodity-grade reagents such as triethylamine and cuprous acetate significantly reduces the raw material cost burden compared to specialized organometallic reagents required by other methods. Furthermore, the operational simplicity of the workup procedure, which involves basic filtration and silica gel treatment, translates to reduced processing time and lower labor costs in a manufacturing setting. This streamlined workflow enhances the overall throughput of the production line, allowing for faster response times to market demands for key pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like diazonium salts directly contributes to a safer and more cost-effective production environment. By utilizing cheap and easily obtainable starting materials, the overall cost of goods sold (COGS) for the triazole intermediate is significantly optimized. Additionally, the high reaction efficiency reported in the patent implies that less raw material is wasted on side products, further driving down the effective cost per kilogram of the final API intermediate.
- Enhanced Supply Chain Reliability: Since all key reagents, including molybdenum hexacarbonyl and the functionalized isonitriles, are commercially available from multiple global suppliers, the risk of supply chain disruption is minimized. This diversification of the supplier base ensures continuity of supply even during periods of market volatility. The robustness of the reaction conditions also means that the process can be transferred between different manufacturing sites with minimal re-validation, providing greater flexibility in logistics and inventory management.
- Scalability and Environmental Compliance: The mild reaction temperatures and the absence of highly toxic byproducts make this process inherently safer for scale-up from gram to multi-kilogram quantities. The use of THF as a primary solvent aligns with standard waste management protocols in modern chemical plants, facilitating easier compliance with environmental regulations. The ability to expand this method to gram-level and beyond without loss of efficiency demonstrates its readiness for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating this technology for adoption.
Q: What are the optimal reaction conditions for this triazole synthesis?
A: The patent specifies reacting at 70-90°C for 18-30 hours in THF solvent, using Mo(CO)6 and CuOAc as co-catalysts with triethylamine as the base.
Q: Can this method tolerate diverse functional groups on the aromatic ring?
A: Yes, the method demonstrates broad substrate tolerance, successfully synthesizing derivatives with methyl, methoxy, fluoro, chloro, and nitro substituents at ortho, meta, or para positions.
Q: Is this process suitable for large-scale manufacturing?
A: The patent explicitly states the method can be expanded to gram-level reactions and potentially larger scales due to mild conditions and simple post-treatment procedures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-trifluoromethyl-1,2,4-triazole meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific drug development programs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your journey from benchtop discovery to commercial manufacturing.
