Advanced Mo/Cu Co-Catalyzed Route for Scalable 3-Trifluoromethyl-1,2,4-Triazole Production
Advanced Mo/Cu Co-Catalyzed Route for Scalable 3-Trifluoromethyl-1,2,4-Triazole Production
The rapid evolution of modern medicinal chemistry demands robust and efficient synthetic methodologies for constructing complex heterocyclic scaffolds, particularly those incorporating fluorine atoms which are pivotal for enhancing metabolic stability and bioavailability. A groundbreaking development in this domain is detailed in patent CN113307778A, which discloses a highly efficient preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. This technology represents a significant leap forward for the pharmaceutical industry, offering a streamlined pathway to access molecular frameworks found in critical therapeutics such as Sitagliptin and various anticonvulsant agents. By leveraging a unique dual-catalyst system involving molybdenum and copper, this process overcomes many of the thermodynamic and kinetic barriers associated with traditional triazole synthesis, providing a reliable foundation for the development of next-generation active pharmaceutical ingredients (APIs).
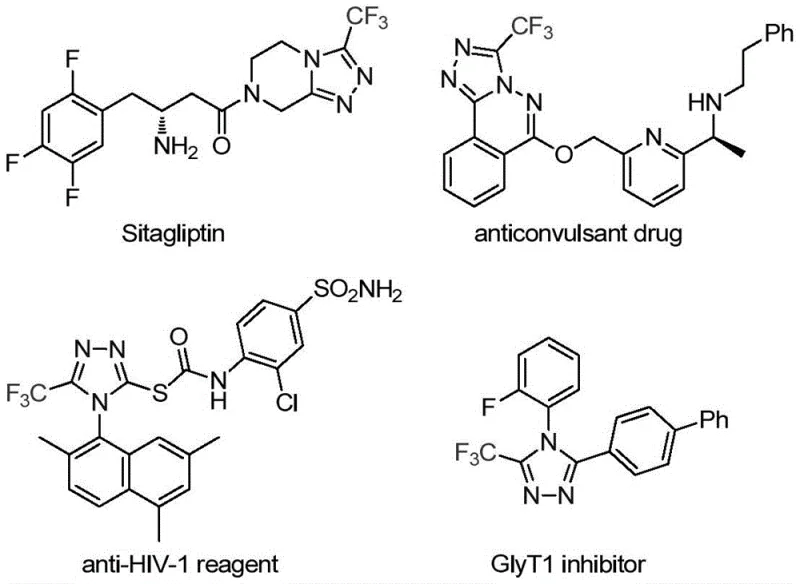
The strategic incorporation of the trifluoromethyl group into the 1,2,4-triazole core is not merely a structural modification but a critical design element that profoundly influences the physicochemical properties of the resulting molecule. As illustrated in the diverse array of bioactive compounds shown above, the presence of the CF3 moiety can drastically improve lipophilicity and electronegativity, thereby optimizing the drug-receptor interaction profiles. For R&D directors and process chemists, the ability to introduce this group efficiently and selectively is paramount. The method described in the patent utilizes a cycloaddition strategy that is both atom-economical and operationally simple, making it an attractive candidate for integration into existing synthetic pipelines where purity and yield are non-negotiable metrics for success.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has relied on methodologies that are often plagued by severe operational drawbacks and safety concerns. Traditional routes frequently involve the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones, processes that typically require harsh reaction conditions and generate significant amounts of hazardous waste. Furthermore, alternative copper-catalyzed multi-component reactions utilizing diazonium salts and trifluorodiazoethane present substantial safety risks due to the explosive nature of diazo compounds, complicating their adoption in large-scale manufacturing environments. These legacy methods often suffer from poor functional group tolerance, limiting the structural diversity accessible to medicinal chemists and necessitating lengthy protection-deprotection sequences that erode overall process efficiency and increase production costs.
The Novel Approach
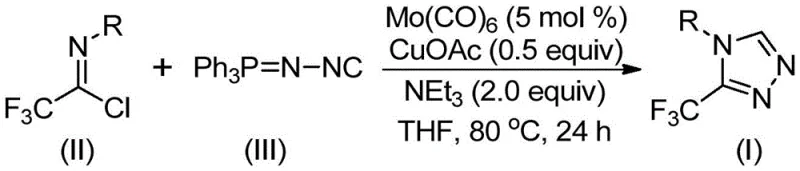
In stark contrast to these cumbersome traditional pathways, the innovative method disclosed in patent CN113307778A utilizes a mild and highly selective cycloaddition reaction between trifluoroethylimidoyl chloride and functionalized isonitrile (NIITP). This approach operates under significantly gentler conditions, typically ranging from 70°C to 90°C, which minimizes thermal degradation of sensitive substrates and reduces energy consumption. The use of commercially available and inexpensive starting materials, combined with a robust catalytic system, ensures that the reaction proceeds with high efficiency and minimal byproduct formation. This novel route not only simplifies the synthetic workflow but also expands the chemical space available for exploration, allowing for the facile introduction of various aryl and alkyl substituents without compromising the integrity of the triazole ring system.
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
The success of this transformation hinges on the synergistic interplay between molybdenum hexacarbonyl and cuprous acetate, which orchestrate a sophisticated catalytic cycle to drive the formation of the five-membered heterocyclic ring. The mechanism likely initiates with the activation of the functionalized isonitrile by the molybdenum species, forming a reactive metal-carbene or metal-imido complex that facilitates the subsequent nucleophilic attack. Concurrently, the copper catalyst promotes the [3+2] cycloaddition step, effectively bridging the trifluoroethylimidoyl chloride and the activated isonitrile intermediate. This dual-catalyst strategy lowers the activation energy barrier for the cyclization, enabling the reaction to proceed smoothly at moderate temperatures while maintaining high regioselectivity for the 3-trifluoromethyl isomer.
From an impurity control perspective, the mildness of this catalytic system is a distinct advantage, as it suppresses common side reactions such as polymerization of the isonitrile or hydrolysis of the imidoyl chloride. The reaction conditions are carefully optimized to ensure that the triphenylphosphine oxide byproduct, generated from the isonitrile precursor, is easily removed during the workup phase, typically via standard silica gel chromatography. This clean reaction profile translates directly into higher crude purity, reducing the burden on downstream purification units and ensuring that the final product meets the stringent quality specifications required for pharmaceutical applications. The mechanistic robustness of this method provides R&D teams with a predictable and reliable tool for synthesizing complex triazole derivatives with confidence.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific procedural parameters to maximize yield and reproducibility. The process begins with the precise charging of the catalytic system, where molybdenum hexacarbonyl is used at a loading of approximately 5 mol %, complemented by 0.5 equivalents of cuprous acetate and 2.0 equivalents of triethylamine as the base. The reaction is conducted in an aprotic solvent, with tetrahydrofuran (THF) identified as the optimal medium due to its superior ability to dissolve the reactants and facilitate the catalytic cycle. Maintaining an inert atmosphere is crucial to prevent oxidation of the catalysts, and the inclusion of molecular sieves helps to scavenge trace moisture that could otherwise deactivate the sensitive imidoyl chloride intermediate.
- Charge a reaction vessel with molybdenum hexacarbonyl (5 mol %), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), and molecular sieves in THF solvent.
- Add trifluoroethylimidoyl chloride and functionalized isonitrile (NIITP) to the mixture under inert atmosphere.
- Heat the reaction mixture to 80°C for 24 hours, then filter and purify via column chromatography to isolate the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition to this novel synthetic route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The reliance on commodity chemicals such as cuprous acetate and triethylamine, rather than exotic or proprietary reagents, significantly de-risks the supply chain by ensuring multiple sources of raw materials are available globally. This diversification of the supply base mitigates the risk of production stoppages due to single-source bottlenecks, a critical consideration for maintaining continuous manufacturing operations in the volatile pharmaceutical market. Furthermore, the operational simplicity of the process reduces the requirement for specialized reactor configurations, allowing for flexible deployment across existing multipurpose manufacturing suites.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like diazonium salts or complex hydrazine derivatives leads to a substantial reduction in raw material costs. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, while the high selectivity minimizes solvent usage for purification, collectively driving down the cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: Since the key starting materials, including the functionalized isonitrile and trifluoroethylimidoyl chloride, are derived from widely available aromatic amines and phosphines, the lead times for raw material acquisition are significantly shortened. This agility allows manufacturers to respond more rapidly to fluctuating market demands and ensures a steady flow of intermediates to downstream API production facilities without prolonged delays.
- Scalability and Environmental Compliance: The process has been validated at the gram scale with high efficiency, indicating a clear path toward kilogram and tonne-scale production. The absence of heavy metal contaminants that are difficult to remove, combined with the use of standard organic solvents, simplifies waste treatment protocols and ensures compliance with increasingly stringent environmental regulations regarding effluent discharge and residual metals in pharmaceutical products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Mo/Cu co-catalyzed technology. These insights are derived directly from the experimental data and scope analysis presented in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential adopters.
Q: What are the key advantages of this Mo/Cu catalytic system over traditional hydrazine cyclization?
A: This novel method utilizes mild reaction temperatures (70-90°C) and avoids the harsh conditions often required for hydrazinolysis. It employs readily available functionalized isonitriles and achieves high conversion rates with excellent functional group tolerance, significantly simplifying the purification process compared to traditional multi-step routes.
Q: Is this synthesis method suitable for large-scale commercial manufacturing?
A: Yes, the patent explicitly demonstrates scalability to gram-level reactions with high efficiency. The use of inexpensive catalysts like cuprous acetate and common solvents like THF suggests a favorable cost profile for scaling up to industrial production volumes without requiring exotic equipment.
Q: What types of substituents are tolerated on the aromatic ring of the starting material?
A: The method exhibits broad substrate scope, successfully accommodating various substituents including alkyl groups (methyl, t-butyl), halogens (fluoro, chloro), alkoxy groups, and even electron-withdrawing groups like nitro. This versatility allows for the synthesis of diverse derivatives crucial for SAR studies in drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs which utilize state-of-the-art analytical instrumentation to verify every batch against exacting standards. Our expertise in fluorine chemistry and transition metal catalysis positions us as an ideal partner for executing complex synthetic routes like the one described in CN113307778A.
We invite pharmaceutical companies and contract research organizations to collaborate with us to leverage this advanced synthetic technology for their pipeline projects. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to obtain specific COA data for our triazole inventory and to discuss route feasibility assessments that can accelerate your drug development programs while optimizing your overall manufacturing budget.
