Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial API Manufacturing
Introduction to Next-Generation Imidazole Synthesis
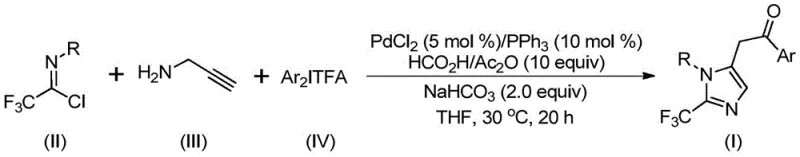
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient and sustainable synthetic routes. A pivotal advancement in this field is detailed in patent CN111423381B, which discloses a novel preparation method for 2-trifluoromethyl substituted imidazole compounds. These heterocyclic structures are critical scaffolds in modern medicinal chemistry, renowned for their ability to enhance metabolic stability and lipophilicity in drug candidates. The disclosed technology leverages a sophisticated palladium-catalyzed carbonylation strategy that operates under exceptionally mild conditions, specifically at 30°C. This represents a significant departure from traditional high-energy synthesis methods, offering a pathway that is not only chemically elegant but also commercially viable for large-scale production. By integrating readily available starting materials such as trifluoroethylimidoyl chloride and propargylamine, this process addresses key pain points in supply chain reliability and cost efficiency.
For R&D directors and procurement specialists, the implications of this patent are profound. It suggests a robust platform for generating diverse libraries of fluorinated imidazoles, which are increasingly prevalent in oncology and anti-infective therapies. The method's compatibility with various functional groups ensures that complex molecular architectures can be accessed without extensive protecting group strategies. Furthermore, the use of formic acid and acetic anhydride as a carbon monoxide surrogate eliminates the need for handling hazardous CO gas directly, thereby enhancing operational safety. As we delve deeper into the technical specifics, it becomes clear that this methodology offers a compelling value proposition for any organization seeking a reliable pharmaceutical intermediate supplier capable of delivering high-quality fluorinated building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted nitrogen-containing heterocycles has been fraught with challenges that hinder efficient commercialization. Traditional approaches often rely on the direct reaction of substrates with specialized trifluoromethyl synthons, such as trifluorodiazoethane or specific trifluoroethylimidoyl halides, which can be expensive and difficult to source in bulk quantities. Moreover, many existing protocols require harsh reaction conditions, including elevated temperatures and strong bases, which can lead to poor functional group tolerance and significant formation of by-products. These limitations not only increase the complexity of downstream purification but also pose safety risks during scale-up. The reliance on unstable intermediates often results in inconsistent yields, making it difficult for supply chain managers to forecast production timelines accurately. Consequently, the industry has long sought a more robust and versatile synthetic strategy that can overcome these inherent inefficiencies while maintaining high standards of product quality.
The Novel Approach
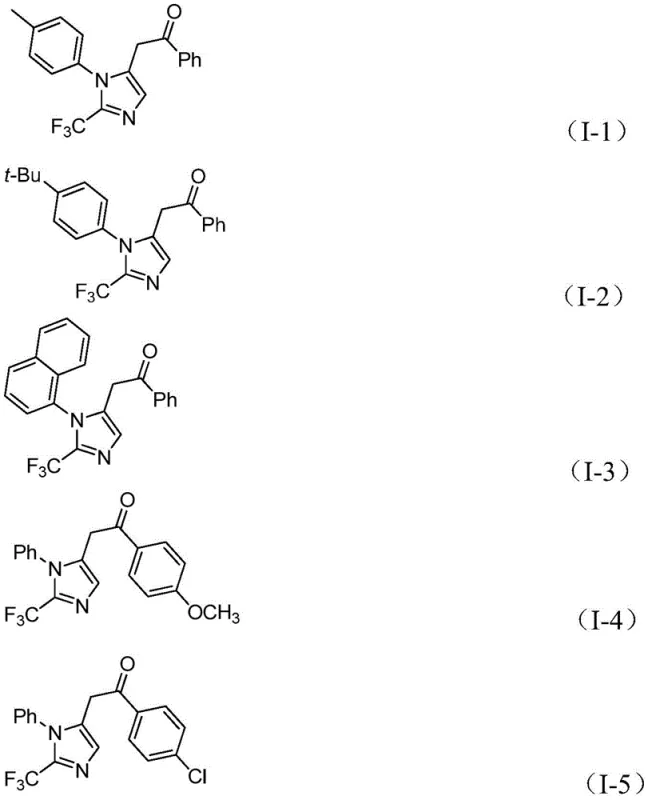
The methodology described in patent CN111423381B introduces a transformative solution by utilizing a transition metal palladium-catalyzed carbonylation series reaction. This innovative approach employs cheap and easily obtained trifluoroethyliminato chloride, propargylamine, and diaryl iodide as the primary starting materials. The reaction proceeds smoothly at a mild temperature of 30°C, drastically reducing energy consumption and thermal stress on the molecular framework. By using formic acid and acetic anhydride to generate carbon monoxide in situ, the process avoids the logistical nightmares associated with high-pressure CO gas cylinders. This multi-component coupling strategy exhibits excellent substrate compatibility, allowing for the introduction of diverse substituents at the 1 and 5 positions of the imidazole ring. The result is a highly efficient synthesis that delivers target compounds with impressive yields, often exceeding 90% in optimized examples, thereby setting a new benchmark for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Palladium-Catalyzed Carbonylation and Cyclization
Understanding the mechanistic underpinnings of this reaction is crucial for R&D teams aiming to optimize the process for specific analogs. The catalytic cycle begins with the formation of an intermolecular carbon-nitrogen bond promoted by the alkaline additive, likely sodium bicarbonate, which generates a trifluoroacetamidine intermediate. This species then undergoes isomerization to align for the subsequent metal-mediated steps. The palladium catalyst, typically introduced as PdCl2 with a triphenylphosphine ligand, facilitates the activation of the alkyne moiety in propargylamine through palladation, forming a key alkenyl palladium intermediate. This intermediate further isomerizes to a more stable alkyl palladium species, positioning the molecule for the critical carbonylation step. The carbon monoxide, released slowly from the decomposition of the formic acid/acetic anhydride mixture, inserts into the palladium-carbon bond to create an acyl palladium intermediate. Finally, oxidative addition of the diaryl iodonium salt generates a transient tetravalent palladium species, which undergoes reductive elimination to forge the final carbon-carbon bond, releasing the 2-trifluoromethyl-substituted imidazole product and regenerating the active catalyst.
From an impurity control perspective, the mild thermal conditions play a pivotal role in maintaining product integrity. High-temperature reactions often promote polymerization of alkynes or decomposition of sensitive trifluoromethyl groups, leading to complex impurity profiles that are difficult to separate. By operating at 30°C, this process minimizes such thermal degradation pathways. Furthermore, the choice of tetrahydrofuran (THF) as the preferred solvent ensures excellent solubility of all reactants, promoting homogeneous reaction kinetics and preventing localized hot spots that could trigger side reactions. The stoichiometry is carefully balanced, with a molar ratio of palladium chloride to triphenylphosphine to sodium bicarbonate optimized at 0.05:0.1:2, ensuring that the catalytic cycle turns over efficiently without excessive accumulation of metal residues. This precise control over reaction parameters translates directly into a cleaner crude product, simplifying the purification workload and enhancing the overall yield of high-purity pharmaceutical intermediates.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
Implementing this synthesis route requires careful attention to reagent quality and reaction monitoring to ensure reproducibility. The patent outlines a straightforward protocol where the catalyst system and additives are mixed with the substrates in an organic solvent, followed by a controlled reaction period. The simplicity of the workup procedure, involving filtration and standard silica gel chromatography, makes this method particularly attractive for process chemistry teams looking to rapidly scale up production. While the general principles are robust, specific optimization of reaction time and solvent volume may be necessary depending on the steric and electronic nature of the aryl substituents used. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below.
- Combine palladium chloride, triphenylphosphine, sodium bicarbonate, acetic anhydride, and formic acid in an organic solvent such as THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salt to the reaction mixture under stirring.
- Maintain the reaction at 30°C for 16 to 24 hours, then filter and purify via column chromatography to isolate the target imidazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic and logistical benefits of this patented process are substantial. The shift towards using commodity chemicals like propargylamine and simple aromatic amines (precursors to the imidoyl chloride) decouples production from the volatility of specialized reagent markets. This strategic sourcing advantage ensures a more stable supply chain, reducing the risk of production stoppages due to raw material shortages. Additionally, the elimination of hazardous gas handling infrastructure for carbon monoxide lowers the capital expenditure required for facility upgrades, making it easier for contract manufacturing organizations to adopt this technology. The mild reaction conditions also imply lower utility costs, as there is no need for extensive heating or cooling systems to maintain extreme temperatures. These factors collectively contribute to a leaner manufacturing model that can respond more agilely to market demands.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive catalysts and abundant starting materials. Unlike methods requiring exotic fluorinating agents or precious metal catalysts in high loadings, this protocol utilizes palladium chloride, which is relatively affordable among palladium sources, and operates at low catalyst loading (5 mol%). The high atom economy of the multi-component reaction means less waste generation, which directly correlates to lower disposal costs. Furthermore, the high conversion rates observed across various substrates minimize the loss of valuable starting materials, ensuring that every kilogram of input translates effectively into saleable product. This efficiency is critical for maintaining competitive pricing in the global pharmaceutical intermediate market.
- Enhanced Supply Chain Reliability: The reliance on commercially available building blocks significantly mitigates supply chain risks. Trifluoroethylimidoyl chloride can be rapidly synthesized from widely sourced aromatic amines, while diaryl iodonium salts are accessible through established supply channels. This diversity in sourcing options prevents bottlenecks that often occur when relying on single-source specialty reagents. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, providing a buffer against supply fluctuations. For supply chain planners, this translates to more predictable lead times and the ability to secure long-term contracts with greater confidence, ensuring continuous availability of critical intermediates for downstream API synthesis.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but the design of this reaction inherently supports expansion. The use of THF, a common industrial solvent, simplifies solvent recovery and recycling operations. The absence of high-pressure gas requirements removes a major barrier to scaling in standard glass-lined or stainless steel reactors. From an environmental standpoint, the generation of carbon monoxide in situ reduces the carbon footprint associated with gas transport and storage. The simplified post-treatment process, which avoids complex extraction sequences, reduces the volume of aqueous waste generated. These attributes align well with modern green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations while achieving commercial scale-up of complex pharmaceutical intermediates with minimal ecological impact.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific experimental data and beneficial effects reported in the patent documentation, providing clarity for stakeholders evaluating this route for their own production needs. Understanding these nuances is essential for making informed decisions about process adoption and integration into existing manufacturing workflows.
Q: What are the key advantages of this palladium-catalyzed method over traditional imidazole synthesis?
A: This method operates at a remarkably mild temperature of 30°C, avoiding the harsh thermal conditions often required for heterocycle formation. It utilizes cheap and readily available starting materials like propargylamine and diaryl iodonium salts, significantly reducing raw material costs compared to specialized trifluoromethylating reagents.
Q: How does this process ensure high purity for pharmaceutical applications?
A: The mild reaction conditions minimize thermal degradation and side reactions, leading to a cleaner crude profile. The use of standard column chromatography for post-processing allows for the effective removal of palladium residues and unreacted starting materials, ensuring the final product meets stringent purity specifications required for API intermediates.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the patent explicitly demonstrates scalability to the gram level with potential for industrial expansion. The use of common solvents like THF and stable catalysts like PdCl2/PPh3 simplifies the engineering requirements for large-scale reactors, facilitating a smoother transition from lab bench to commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient fluorination technologies in modern drug development. Our team of expert chemists has extensively analyzed the potential of the palladium-catalyzed route described in CN111423381B and is fully prepared to leverage this methodology for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl imidazole meets the highest industry standards. We are committed to being a partner that not only supplies chemicals but also provides technical solutions that enhance your overall R&D productivity.
We invite you to discuss how our capabilities can support your specific requirements for fluorinated heterocycles. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your project's volume and timeline. By collaborating with us, you gain access to a supply chain that prioritizes reliability and quality. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you accelerate your drug development pipeline with our superior manufacturing expertise and commitment to excellence.
