Advanced Palladium-Catalyzed Dehydrogenation for High-Purity Steroidal API Intermediates
Advanced Palladium-Catalyzed Dehydrogenation for High-Purity Steroidal API Intermediates
The pharmaceutical industry constantly seeks more efficient and environmentally benign pathways for synthesizing critical active pharmaceutical ingredients (APIs), particularly complex steroidal compounds. Patent CN1319985C introduces a groundbreaking method for introducing a 1,2-double bond into 3-oxo-4-azasteroid compounds, a pivotal transformation in the manufacture of 5-alpha-reductase inhibitors like Finasteride and Dutasteride. This technology represents a significant departure from traditional oxidation methods, utilizing a sophisticated palladium-catalyzed dehydrogenation strategy that enhances both safety and product purity. By leveraging transition metal catalysis, specifically Group VIII metals like Palladium, the process achieves high selectivity for the Δ1-double bond while minimizing the formation of hazardous byproducts. For R&D directors and procurement specialists, this patent outlines a robust framework for producing high-purity pharmaceutical intermediates that meet stringent regulatory standards while offering substantial operational advantages over legacy synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of a 1,2-double bond into 17β-substituted 4-aza-5α-androstanes has relied heavily on oxidation using selenium dioxide or similar harsh oxidizing agents. These conventional methods, while effective in laboratory settings, present severe challenges for commercial scale-up due to the inherent toxicity of selenium compounds and the difficulty in removing trace metal residues from the final API. The use of stoichiometric amounts of toxic oxidants generates significant hazardous waste, complicating disposal and increasing the environmental footprint of the manufacturing process. Furthermore, selenium-based oxidations often suffer from issues with over-oxidation or lack of regioselectivity, leading to complex impurity profiles that require extensive and costly purification steps. These factors collectively drive up production costs and extend lead times, creating bottlenecks in the supply chain for essential urological and dermatological medications.
The Novel Approach
The novel approach detailed in the patent data circumvents these issues by employing a catalytic dehydrogenation mechanism mediated by palladium complexes in the presence of specific hydrogen acceptors. Instead of stoichiometric toxic oxidants, the process utilizes catalytic amounts of palladium species, such as tris(dibenzylideneacetone)dipalladium-chloroform complexes, coupled with benign hydrogen acceptors like substituted benzoquinones or allyl carbonates. This shift from stoichiometric to catalytic chemistry drastically reduces the chemical load and waste generation associated with the reaction. The method involves a strategic protection-deprotection sequence where the 3-oxo-4-aza group is temporarily masked, allowing the catalyst to selectively target the 1,2-position without interfering with the sensitive lactam functionality. This results in a cleaner reaction profile, higher yields, and a final product that is easier to purify to pharmaceutical grades, directly addressing the cost and quality concerns of modern API manufacturing.
Mechanistic Insights into Palladium-Catalyzed Dehydrogenation
The core of this technological advancement lies in the intricate catalytic cycle facilitated by the palladium complex, which operates through a mechanism distinct from traditional radical oxidations. The reaction initiates with the coordination of the palladium catalyst to the protected steroid substrate, where the specific electronic environment created by the protecting groups plays a crucial role in directing the metal center. As the catalytic cycle progresses, the palladium species facilitates the abstraction of hydrogen atoms from the 1 and 2 positions of the steroid backbone. This is achieved through a sequence involving oxidative addition and subsequent beta-hydride elimination, effectively desaturating the A-ring of the steroid nucleus. The presence of hydrogen acceptors, such as allyl methyl carbonate or benzoquinone, is critical as they scavenge the eliminated hydrogen, driving the equilibrium towards the formation of the desired Δ1-double bond and regenerating the active palladium catalyst for further turnover. This mechanistic elegance ensures that the reaction proceeds with high atom economy and minimal side reactions.
Crucial to the success of this mechanism is the temporary modification of the 3-oxo-4-aza moiety, which prevents the catalyst from coordinating to the nitrogen or oxygen atoms in a way that would deactivate the cycle or lead to decomposition. The patent specifies the use of protecting groups such as trialkylsilyl groups or alkoxycarbonyl groups (like BOC) to mask these reactive sites.  shows the starting 3-oxo-4-azasteroid structure which serves as the substrate for this transformation. Following protection, the intermediate adopts the structure shown in
shows the starting 3-oxo-4-azasteroid structure which serves as the substrate for this transformation. Following protection, the intermediate adopts the structure shown in 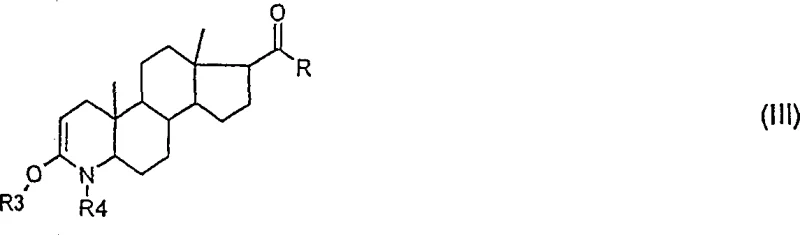 , where R3 and R4 represent the masking groups that stabilize the molecule during the harsh dehydrogenation conditions. This structural stabilization is key to maintaining the integrity of the steroid skeleton while allowing the rigorous conditions required for double bond formation to proceed efficiently without degrading the sensitive lactam ring.
, where R3 and R4 represent the masking groups that stabilize the molecule during the harsh dehydrogenation conditions. This structural stabilization is key to maintaining the integrity of the steroid skeleton while allowing the rigorous conditions required for double bond formation to proceed efficiently without degrading the sensitive lactam ring.
How to Synthesize 17β-substituted 4-aza-androst-1-ene-3-one Efficiently
The synthesis of these valuable intermediates follows a logical three-step sequence designed to maximize yield and purity while minimizing operational complexity. The process begins with the protection of the starting dihydro compound, followed by the critical palladium-catalyzed dehydrogenation step, and concludes with the removal of protecting groups to reveal the final active structure. Each step is optimized for scalability, utilizing common industrial solvents like toluene, acetonitrile, and tetrahydrofuran. The detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating this high-efficiency route.
- Introduce protective groups (such as trialkylsilyl or alkoxycarbonyl) into the 3-keto-4-aza group of the corresponding 1,2-dihydro compound to generate the protected intermediate.
- React the protected compound in the presence of a dehydration catalyst (preferably Palladium-based) and an oxidant like substituted benzoquinone or allyl carbonate to introduce the Δ1-double bond.
- Remove the protective groups using acid treatment and optionally convert the resulting compound to a pharmaceutically acceptable salt through recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this palladium-catalyzed route offers transformative benefits that extend far beyond simple chemical yield improvements. The elimination of selenium dioxide from the process flow removes a major regulatory and safety hurdle, significantly reducing the costs associated with hazardous waste treatment and worker safety protocols. By switching to a catalytic system, the consumption of expensive reagents is minimized, and the reliance on scarce or highly regulated raw materials is reduced, thereby enhancing the resilience of the supply chain against market fluctuations. Furthermore, the simplified purification profile means that fewer processing units and less solvent are required to achieve the necessary purity levels, leading to a drastic simplification of the manufacturing workflow and a reduction in overall production time.
- Cost Reduction in Manufacturing: The transition from stoichiometric toxic oxidants to a catalytic palladium system fundamentally alters the cost structure of API intermediate production. By eliminating the need for expensive selenium reagents and the complex downstream processing required to remove heavy metal residues, manufacturers can achieve substantial cost savings. The catalytic nature of the reaction means that small amounts of palladium can process large quantities of substrate, improving the overall material efficiency. Additionally, the use of readily available hydrogen acceptors like allyl carbonates avoids the price volatility associated with specialized oxidizing agents, providing a more predictable and stable cost base for long-term procurement planning.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes directly to supply chain stability by reducing the risk of production stoppages due to safety incidents or regulatory compliance issues. The reagents used, such as palladium catalysts and common organic solvents, are widely available from multiple global suppliers, reducing the risk of single-source dependency. The process tolerance allows for consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply to downstream formulation partners. This reliability ensures that lead times for high-purity pharmaceutical intermediates can be consistently met, even in the face of fluctuating demand for treatments like benign prostatic hyperplasia and androgenetic alopecia.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this method is ideally suited for commercial expansion. The reaction conditions are compatible with standard stainless steel reactors and do not require specialized corrosion-resistant equipment often needed for harsh acidic or oxidative environments. The reduction in hazardous waste generation aligns with increasingly strict global environmental regulations, facilitating easier permitting and operation in diverse geographic locations. The ability to control polymorphic forms through specific recrystallization protocols, such as cooling rates in toluene, ensures that the physical properties of the final solid are consistent, a critical factor for successful technology transfer from pilot plant to multi-ton commercial production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dehydrogenation technology. These answers are derived directly from the patent specifications and are intended to clarify the operational parameters and benefits for potential manufacturing partners. Understanding these details is essential for evaluating the feasibility of integrating this route into existing production lines.
Q: What are the advantages of using Palladium catalysts over Selenium Dioxide for this synthesis?
A: The use of Palladium catalysts eliminates the toxicity and environmental hazards associated with Selenium Dioxide oxidation. It allows for milder reaction conditions, easier removal of metal residues, and improved safety profiles for large-scale manufacturing.
Q: How does the protection strategy improve the selectivity of the dehydrogenation?
A: Protecting the 3-oxo-4-aza group (e.g., with BOC or silyl groups) stabilizes the enol/imine tautomer and prevents side reactions at the nitrogen or oxygen sites. This ensures the catalyst specifically targets the 1,2-position for double bond introduction.
Q: Can this process be scaled for commercial production of Finasteride intermediates?
A: Yes, the process utilizes common organic solvents like toluene and acetonitrile and robust catalysts suitable for industrial reactors. The recrystallization steps described allow for precise control over polymorphic forms, ensuring consistent quality at scale.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Finasteride Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering high-purity steroidal intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in palladium-catalyzed transformations allows us to offer superior quality products with optimized impurity profiles, giving our partners a distinct advantage in their regulatory filings and market entry strategies.
We invite you to collaborate with us to leverage this cutting-edge technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can drive value and efficiency for your organization.
