Revolutionizing Aryl Boronate Synthesis with Novel Thiosilane Ligand Technology
The landscape of organic synthesis, particularly within the realm of pharmaceutical and agrochemical intermediate manufacturing, is constantly evolving to meet the demands for higher efficiency and sustainability. A pivotal advancement in this field is documented in Chinese Patent CN111217847B, which introduces a novel class of thiosilane ligands designed specifically for iridium-catalyzed aryl boronation reactions. This technology addresses long-standing challenges associated with traditional catalytic systems, offering a robust pathway for the synthesis of arylboronic acid esters, which are critical building blocks in modern drug discovery. The patent details a comprehensive methodology that not only enhances reaction yields but also simplifies downstream processing, making it an attractive option for industrial scale-up. By leveraging the unique electronic and steric properties of sulfur and silicon atoms within the ligand framework, this invention provides a stable and efficient alternative to existing technologies, promising significant improvements in process chemistry for complex molecule synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of arylboronic acid esters has relied heavily on carbon-hydrogen bond boronation catalysis methods utilizing bipyridine ligands in conjunction with iridium catalysts. While effective in certain contexts, these conventional systems suffer from notable drawbacks that hinder their utility in large-scale industrial applications. A primary concern is the relatively low yield observed, particularly when dealing with electron-rich substrates which are common in pharmaceutical scaffolds. Furthermore, the separation and purification processes are often cumbersome and costly; the polarity of bipyridine ligands is frequently too similar to that of the desired boronate products, leading to difficult chromatographic separations and increased material loss. Additionally, alternative systems using borane pyridine ligands involve complex synthetic routes that are not conducive to cost-effective mass production. These limitations create bottlenecks in supply chains, increasing the overall cost of goods and extending lead times for critical intermediates needed in the development of new therapeutic agents.
The Novel Approach
In stark contrast to these legacy methods, the thiosilane ligands described in the patent represent a paradigm shift in catalytic design. These ligands, characterized by structures L1, L2, and L3, are engineered to provide superior performance through a combination of stability and reactivity. 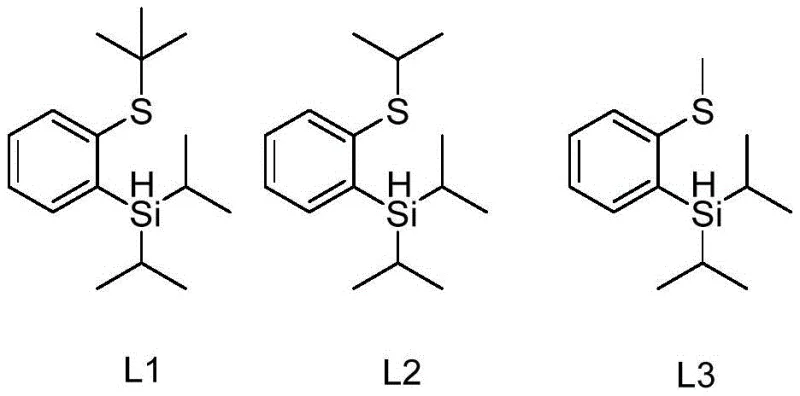 The novel approach utilizes a thiosilane framework that can be synthesized efficiently in just one or two steps from readily available starting materials such as 2-bromobenzene thioether compounds. This simplicity in ligand preparation translates directly to lower raw material costs and reduced waste generation. Crucially, the resulting catalytic system exhibits high atom economy and produces arylboronic acid compounds with significantly improved yields. The distinct chemical nature of the thiosilane ligand ensures that it possesses different polarity characteristics compared to the product, facilitating much easier purification via standard column chromatography. This ease of separation is a major operational advantage, allowing for cleaner product streams and higher overall process efficiency in a manufacturing setting.
The novel approach utilizes a thiosilane framework that can be synthesized efficiently in just one or two steps from readily available starting materials such as 2-bromobenzene thioether compounds. This simplicity in ligand preparation translates directly to lower raw material costs and reduced waste generation. Crucially, the resulting catalytic system exhibits high atom economy and produces arylboronic acid compounds with significantly improved yields. The distinct chemical nature of the thiosilane ligand ensures that it possesses different polarity characteristics compared to the product, facilitating much easier purification via standard column chromatography. This ease of separation is a major operational advantage, allowing for cleaner product streams and higher overall process efficiency in a manufacturing setting.
Mechanistic Insights into Thiosilane-Iridium Catalyzed C-H Activation
The efficacy of this technology lies in the intricate mechanistic interplay between the thiosilane ligand and the iridium catalyst. The proposed mechanism suggests that the thiosilane ligand first coordinates with the iridium center to form a highly active intermediate species. This coordination is stabilized by the soft sulfur donor and the specific steric environment provided by the silyl groups, such as the diisopropylsilyl moiety found in the ligand structures. Once formed, this catalytic complex interacts with the substrate, specifically coordinating with the directing group, typically an ester functionality present on the aryl ring. This coordination activates the ortho-position carbon-hydrogen bond, lowering the energy barrier for insertion. Subsequently, the pinacol ester diboron reagent reacts with this activated complex to effect the boronation of the carbon-hydrogen bond. This directed C-H activation strategy ensures high regioselectivity, minimizing the formation of unwanted isomers and maximizing the yield of the desired ortho-boronated product.
Furthermore, the structural versatility of the ligands allows for fine-tuning the electronic properties to suit different substrate classes. For instance, ligands L1, L2, and L3 feature different thioether groups (tert-butyl, isopropyl, and methyl, respectively), which modulate the steric bulk around the metal center. This modulation is critical for accommodating substrates with varying degrees of steric hindrance or electronic demand. The patent highlights that this system is particularly effective for substrates containing strong electron-withdrawing groups, which traditionally exhibit low reactivity in C-H activation scenarios. The ability to maintain high catalytic turnover numbers across a broad range of electronic environments underscores the robustness of the thiosilane ligand design. This mechanistic flexibility ensures that the process remains viable for a diverse library of chemical building blocks, enhancing its value as a platform technology for medicinal chemistry.
How to Synthesize Thiosilane Ligands Efficiently
The synthesis of these high-performance ligands is designed for operational simplicity and scalability, making it accessible for both laboratory research and pilot plant operations. The general procedure involves a bromine-lithium exchange reaction followed by a silanization step, both of which are standard transformations in organometallic chemistry. The process begins with the treatment of a 2-bromobenzene thioether precursor with n-butyl lithium in tetrahydrofuran at cryogenic temperatures ranging from -30 to -80°C. This step generates a reactive lithiated intermediate which is then trapped with diisopropylchlorosilane. The reaction conditions are carefully controlled to ensure complete conversion while minimizing side reactions. Following the silanization, the mixture is quenched and subjected to a straightforward workup involving extraction and concentration. The final purification is achieved using silica gel chromatography, yielding the target thiosilane ligand as a light yellow oily liquid with high purity. Detailed standardized synthesis steps are provided below to ensure reproducibility.
- Perform bromine-lithium exchange on 2-bromobenzene thioether compounds using n-butyl lithium in tetrahydrofuran at -30 to -80°C under protective atmosphere.
- Add diisopropylchlorosilane to the lithiated intermediate to initiate the silanization reaction at 0-25°C.
- Quench the reaction, extract with ether, and purify the crude product via silica gel column chromatography to obtain the final thiosilane ligand.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this thiosilane ligand technology offers compelling advantages for procurement managers and supply chain directors focused on cost optimization and reliability. The primary driver for cost reduction is the elimination of complex and expensive ligand synthesis pathways associated with traditional bipyridine systems. Since the thiosilane ligands are prepared from low-cost commodity chemicals in high yields, the overall cost of the catalytic system is drastically reduced. Moreover, the high efficiency of the catalyst means that lower loadings may be achievable in optimized processes, further decreasing the consumption of precious iridium metal. The simplified purification process also contributes to cost savings by reducing solvent usage and shortening processing time, which are significant factors in the total cost of manufacturing for fine chemicals.
- Cost Reduction in Manufacturing: The economic benefits extend beyond just the price of the ligand itself. By improving the yield of the aryl boronation reaction, the process minimizes the loss of valuable starting materials, which is crucial when working with complex or multi-step intermediates. The high atom economy of the reaction ensures that a greater proportion of the input mass is converted into the desired product, reducing waste disposal costs. Additionally, the stability of the ligand at room temperature eliminates the need for specialized cold storage infrastructure, lowering overhead expenses associated with inventory management. These cumulative effects result in a leaner, more cost-effective manufacturing process that enhances the competitiveness of the final pharmaceutical or agrochemical product.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available raw materials for ligand synthesis. Precursors such as 2-bromothioanisole and diisopropylchlorosilane are commercially accessible, reducing the risk of supply disruptions that can occur with proprietary or niche reagents. The robustness of the catalytic system also implies a wider operating window for reaction parameters, making the process less sensitive to minor fluctuations in temperature or reagent quality. This tolerance translates to fewer batch failures and more consistent production schedules. For supply chain heads, this reliability is paramount in ensuring continuous availability of key intermediates, thereby preventing delays in downstream drug substance manufacturing and protecting revenue streams.
- Scalability and Environmental Compliance: The technology is inherently designed for scalability, with the patent explicitly noting its suitability for industrial mass synthesis. The reaction conditions, utilizing solvents like 2-methyltetrahydrofuran, are compatible with large-scale reactor setups and standard safety protocols. From an environmental standpoint, the high selectivity and yield reduce the generation of hazardous byproducts, aligning with green chemistry principles. The ease of purification reduces the volume of silica gel and solvents required for chromatography, lowering the environmental footprint of the process. This alignment with sustainability goals is increasingly important for meeting regulatory standards and corporate social responsibility targets in the global chemical industry.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology, we have compiled answers to common questions regarding the implementation and performance of the thiosilane ligand system. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation. Understanding these details is essential for assessing the feasibility of integrating this method into existing production workflows. The following responses address key concerns regarding substrate scope, operational parameters, and product quality.
Q: What are the advantages of thiosilane ligands over traditional bipyridine ligands?
A: Thiosilane ligands offer higher yields, especially for electron-rich substrates, and are significantly easier to separate from products due to polarity differences, unlike bipyridine ligands which often co-elute.
Q: Can this catalytic system be scaled for industrial production?
A: Yes, the ligand synthesis involves only 1-2 steps with low-cost raw materials, and the catalytic reaction demonstrates high atom economy suitable for large-scale manufacturing.
Q: What types of substrates are compatible with this boronation method?
A: The method is highly versatile, accommodating substrates with various substituents including halogens, alkyl groups, methoxy groups, and ester groups, even those with strong electron-withdrawing properties.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thiosilane Ligand Supplier
As the demand for efficient and scalable synthesis methods grows, partnering with an experienced CDMO becomes essential for translating patented innovations into commercial reality. NINGBO INNO PHARMCHEM stands at the forefront of this transition, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is well-versed in the nuances of organometallic catalysis and possesses the infrastructure to handle sensitive reagents like thiosilane ligands and iridium complexes with precision. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch of intermediate meets the highest standards required by the pharmaceutical industry. Our commitment to quality assurance guarantees that the complex chemical architectures enabled by this technology are delivered with consistency and reliability.
We invite potential partners to engage with our technical procurement team to discuss how this advanced boronation technology can be tailored to your specific project needs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that evaluates the economic impact of switching to this novel ligand system for your specific molecule. We encourage you to request specific COA data and route feasibility assessments to validate the performance of our materials in your own processes. Together, we can accelerate the development of next-generation therapeutics by leveraging cutting-edge catalytic solutions that drive efficiency and reduce time-to-market.
