Advanced Metal-Free Synthesis of Gem-Difluoroalkenes for Pharma and Agrochemical Manufacturing
The global demand for fluorinated organic molecules continues to surge, driven by their unique ability to enhance metabolic stability and lipophilicity in drug candidates and agrochemical agents. A significant breakthrough in this domain is detailed in Chinese Patent CN114409515B, which discloses a novel preparation method for gem-difluoroalkene compounds. This technology represents a paradigm shift from traditional carbonyl-based syntheses, offering a robust pathway to convert trifluoromethyl-alkene compounds into valuable gem-difluoroalkenes through a hydrodefluorination reaction. The core innovation lies in the utilization of diphenylphosphine oxide as a reducing agent in conjunction with cesium carbonate and a specific amount of water as a promoter. This combination allows the reaction to proceed under remarkably mild conditions, specifically at 70°C in ethyl acetate, avoiding the harsh environments typically associated with fluorine chemistry. For R&D directors and process chemists, this patent offers a compelling solution for accessing complex fluorinated scaffolds with high regioselectivity and excellent functional group tolerance.
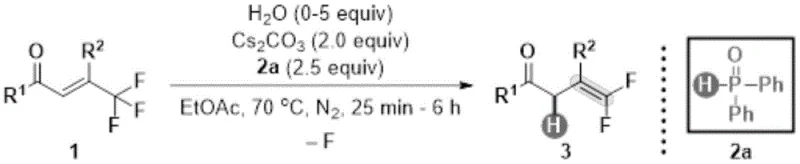
The significance of this methodology extends beyond academic interest; it addresses critical bottlenecks in the supply chain of high-value fine chemicals. By eliminating the need for transition metal catalysts, the process inherently reduces the risk of heavy metal contamination, a persistent challenge in pharmaceutical manufacturing that often necessitates costly purification steps. Furthermore, the reaction demonstrates broad substrate scope, accommodating various substituents including halogenated phenyls, methoxy groups, and heterocycles like thiophene. This versatility makes it an attractive candidate for the late-stage functionalization of complex molecules, allowing medicinal chemists to introduce difluoroalkenyl motifs without compromising sensitive functional groups elsewhere in the molecular architecture. As we delve deeper into the technical specifics, it becomes clear that this patent provides a foundational technology for the next generation of fluorinated intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
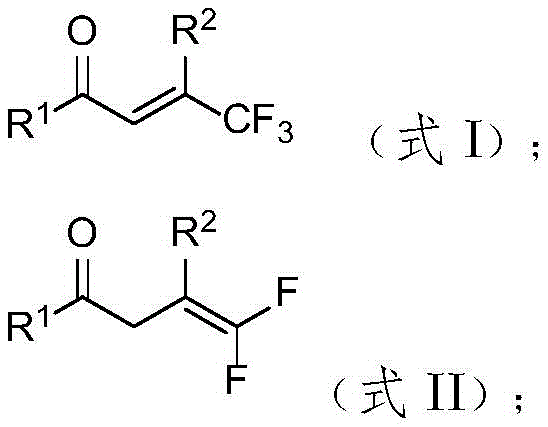
Historically, the synthesis of gem-difluoroalkenes has relied heavily on classical carbon-carbon bond-forming reactions such as the Wittig reaction or the Horner-Wadsworth-Emmons (HWE) reaction. While these methods are well-established in organic synthesis textbooks, they suffer from significant practical limitations when applied to complex industrial settings. Primarily, these traditional routes often necessitate the use of strong bases to generate the requisite ylides or carbanions, conditions that are incompatible with a wide range of electrophilic functional groups commonly found in advanced intermediates. This lack of chemoselectivity frequently leads to side reactions, decomposition of sensitive substrates, and ultimately, lower overall yields. Additionally, the preparation of the phosphorus ylides themselves can be cumbersome, requiring stoichiometric amounts of reagents and generating substantial phosphorus-containing waste. From a supply chain perspective, the reliance on specific aldehyde or ketone precursors can also introduce volatility in raw material sourcing, especially if those precursors are themselves difficult to synthesize or stabilize.
The Novel Approach
In stark contrast, the method disclosed in patent CN114409515B utilizes a hydrodefluorination strategy that transforms readily available trifluoromethyl-alkene compounds directly into the desired gem-difluoroalkenes. This approach bypasses the need for strong bases entirely, operating instead under neutral to mildly basic conditions facilitated by cesium carbonate. The use of diphenylphosphine oxide as a hydrogen source is particularly ingenious, as it serves as a mild reducing agent that selectively removes a fluorine atom while installing a hydrogen atom at the same position. This transformation is not only atom-economical regarding the fluorine retention but also operationally simple. The reaction proceeds efficiently in ethyl acetate, a green solvent preferred in modern manufacturing for its low toxicity and ease of recovery. By shifting the synthetic logic from building the double bond from scratch to modifying an existing trifluoromethyl group, this novel approach drastically simplifies the synthetic route, reduces the number of unit operations, and enhances the overall safety profile of the manufacturing process.
Mechanistic Insights into Diphenylphosphine Oxide Promoted Hydrodefluorination
The mechanistic underpinning of this reaction involves a sophisticated interplay between the phosphine oxide reductant, the base, and the water promoter. Diphenylphosphine oxide (2a), acting as the hydrogen donor, likely undergoes activation by the cesium carbonate base to form a reactive phosphinate species. This species then interacts with the electron-deficient double bond of the trifluoromethyl-alkene substrate. The presence of water is critical; experimental optimization revealed that 5.0 equivalents of water maximize the yield, suggesting that water participates in the proton transfer steps or stabilizes key intermediates in the catalytic cycle. Without water, or with excessive amounts (e.g., 20 equivalents), the reaction efficiency drops precipitously, indicating a delicate balance in the reaction medium's polarity and proton availability. The regioselectivity observed, where the fluorine atom adjacent to the R2 group is retained while the terminal fluorines are removed, points to a mechanism governed by electronic stabilization of the developing negative charge during the defluorination event. This level of control is essential for producing single-isomer products required in regulatory-compliant pharmaceutical synthesis.
Furthermore, the tolerance of diverse functional groups underscores the mildness of the reactive intermediates involved. Unlike radical defluorination methods that might indiscriminately attack other parts of the molecule, this ionic or polar mechanism respects the integrity of esters, nitriles, and halogens. For instance, substrates containing bromo-phenyl groups remain intact, allowing for subsequent cross-coupling reactions downstream. This orthogonality is a massive advantage for process chemists designing convergent synthesis routes. The ability to perform this transformation at 70°C also implies a manageable activation energy barrier, reducing the thermal stress on the equipment and minimizing the formation of thermal degradation byproducts. Understanding these mechanistic nuances allows manufacturers to fine-tune reaction parameters for specific substrates, ensuring consistent quality and minimizing batch-to-batch variability in large-scale production runs.
How to Synthesize Gem-Difluoroalkene Compounds Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized conditions identified in the patent data to achieve the reported high yields. The protocol involves charging a reactor with the trifluoromethyl-alkene starting material, followed by the addition of ethyl acetate as the solvent. Crucially, the stoichiometry must be controlled precisely: 2.5 equivalents of diphenylphosphine oxide and 2.0 equivalents of cesium carbonate are required to drive the reaction to completion. The addition of 5.0 equivalents of water is the key variable that distinguishes this high-yielding process from less efficient variations. The mixture is then heated to 70°C under an inert nitrogen atmosphere to prevent oxidation of the phosphine species. Reaction times can vary from 25 minutes to 6 hours depending on the specific steric and electronic nature of the R1 and R2 substituents. Following the reaction, a standard aqueous workup involving ammonium chloride quenching and extraction isolates the crude product, which is then purified via silica gel chromatography. For detailed step-by-step instructions and safety precautions, please refer to the standardized guide below.
- Combine trifluoromethyl-alkene compound, diphenylphosphine oxide (2.5 equiv), cesium carbonate (2.0 equiv), and water (5.0 equiv) in ethyl acetate solvent.
- Stir the reaction mixture under a nitrogen atmosphere at 70°C for 25 minutes to 6 hours to facilitate hydrodefluorination.
- Quench with saturated ammonium chloride, extract with ethyl acetate, dry over sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible strategic benefits that extend far beyond the chemistry itself. The most immediate impact is seen in the cost structure of the manufacturing process. By eliminating the need for expensive transition metal catalysts such as palladium or rhodium, which are subject to volatile market pricing and geopolitical supply risks, the overall cost of goods sold (COGS) is significantly reduced. Moreover, the removal of heavy metals from the process flow negates the need for specialized scavenging resins or complex filtration systems designed to meet stringent residual metal specifications (e.g., ICH Q3D guidelines). This simplification of the downstream processing train translates directly into reduced capital expenditure (CAPEX) for equipment and lower operational expenditure (OPEX) for consumables and waste disposal. The use of ethyl acetate, a commodity solvent with a stable global supply chain, further insulates the production process from the disruptions often seen with chlorinated or aromatic solvents.
- Cost Reduction in Manufacturing: The economic model of this synthesis is bolstered by the use of commodity chemicals. Diphenylphosphine oxide and cesium carbonate are widely available bulk chemicals with established supply chains, ensuring price stability and availability. Unlike proprietary catalysts that may require long lead times or exclusive licensing agreements, these reagents can be sourced from multiple vendors globally, fostering competitive pricing. Additionally, the high atom economy of the hydrodefluorination reaction means less raw material is wasted as byproduct, maximizing the yield per kilogram of input. The simplified purification process, which avoids complex metal removal steps, reduces the consumption of silica gel and solvents during chromatography, further driving down the variable costs associated with each production batch. These factors combine to create a highly cost-competitive manufacturing route suitable for both clinical trial materials and commercial API production.
- Enhanced Supply Chain Reliability: Supply chain resilience is a top priority for multinational corporations, and this technology supports that goal through原料 accessibility. The starting materials, trifluoromethyl-alkene compounds, are increasingly accessible due to advancements in trifluoromethylation chemistry, creating a robust upstream supply base. The reaction conditions are mild enough to be performed in standard glass-lined or stainless steel reactors without requiring exotic materials of construction that resist highly corrosive acids or bases. This compatibility with standard manufacturing infrastructure means that production can be easily transferred between different contract manufacturing organizations (CMOs) or internal sites without significant retrofitting. The robustness of the reaction against moisture (given the intentional addition of water) also relaxes the stringent anhydrous requirements typical of organometallic chemistry, reducing the risk of batch failures due to environmental humidity fluctuations.
- Scalability and Environmental Compliance: Scaling chemical processes from grams to tons often reveals hidden pitfalls, but this hydrodefluorination method appears inherently scalable. The exotherm is manageable at 70°C, and the use of ethyl acetate facilitates easy solvent recovery and recycling, aligning with green chemistry principles. From an environmental, health, and safety (EHS) perspective, the absence of toxic heavy metals simplifies wastewater treatment and solid waste disposal, helping manufacturers meet increasingly rigorous environmental regulations. The high selectivity of the reaction minimizes the formation of difficult-to-separate impurities, reducing the solvent load required for purification. This efficiency not only lowers the environmental footprint but also accelerates the production cycle time, allowing for faster response to market demands and shorter lead times for delivering high-purity intermediates to customers.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common questions regarding the reaction scope, optimization, and scalability. These insights are derived directly from the experimental data and technical disclosures within the patent literature. Understanding these details is crucial for assessing the feasibility of integrating this method into existing production workflows or new drug development pipelines. The following responses address key concerns regarding substrate compatibility, reagent roles, and process robustness.
Q: What are the advantages of this metal-free hydrodefluorination method over traditional Wittig reactions?
A: Unlike traditional Wittig or Horner-Wadsworth-Emmons reactions that require strong alkaline conditions and have limited substrate tolerance, this patented method operates under mild conditions (70°C) using diphenylphosphine oxide as a reducing agent. It offers superior functional group tolerance and regioselectivity without the need for transition metal catalysts.
Q: What is the role of water in this synthesis reaction?
A: Water acts as a critical reaction promoter in this system. Experimental data indicates that adding 5.0 equivalents of water significantly increases the reaction yield compared to anhydrous conditions, facilitating the hydrodefluorination process while maintaining mild reaction parameters.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes common industrial solvents like ethyl acetate and inexpensive reagents such as cesium carbonate and diphenylphosphine oxide. The absence of precious metal catalysts simplifies purification and waste treatment, making it highly scalable for commercial manufacturing of pharmaceutical and agrochemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gem-Difluoroalkene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free hydrodefluorination technologies in the synthesis of high-value fluorinated intermediates. Our team of expert process chemists has extensively evaluated the methodology described in patent CN114409515B and possesses the capability to adapt and optimize this route for diverse substrate classes. We understand that moving from a patent example to a commercial process requires more than just following a recipe; it demands a deep understanding of reaction engineering, impurity profiling, and crystallization dynamics. Our facility is equipped with state-of-the-art analytical instrumentation and pilot-scale reactors capable of handling fluorine chemistry safely and efficiently. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which guarantee that every batch of gem-difluoroalkene intermediate meets the exacting standards required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Whether you require custom synthesis of novel fluorinated building blocks or scale-up of existing intermediates, our technical procurement team is ready to support your goals. Contact us today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We encourage you to reach out to our technical procurement team to obtain specific COA data and route feasibility assessments that demonstrate how we can add value to your supply chain. Let NINGBO INNO PHARMCHEM be your partner in navigating the complexities of fluorine chemistry and delivering superior chemical solutions.
