Revolutionizing Atorvastatin Intermediate Production via Green Multicomponent One-Pot Catalysis
Introduction to Advanced Atorvastatin Intermediate Synthesis
The global demand for lipid-lowering agents continues to drive innovation in the synthesis of key pharmaceutical building blocks. Patent CN111909048B introduces a transformative approach to producing 4-(4-fluorophenyl)-2-(2-methylpropanoyl)-3-phenyl-4-oxo-N-phenylbutanamide, a critical precursor in the manufacture of Atorvastatin Calcium. This advanced intermediate serves as the foundational scaffold for one of the world's best-selling statins, widely used to manage hypercholesterolemia and reduce cardiovascular risk. The patented technology shifts the paradigm from traditional, linear multi-step syntheses to a highly efficient, green multicomponent one-pot reaction. By leveraging a sophisticated synergistic catalytic system involving Copper and Palladium complexes, this method achieves exceptional atom economy and significantly reduces the environmental footprint associated with legacy manufacturing processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the Atorvastatin core has relied heavily on the Paal-Knorr pyrrole synthesis, which necessitates the prior preparation of complex intermediates like B-4. Conventional routes to synthesize this specific intermediate have been plagued by significant technical and environmental hurdles. For instance, early methodologies reported by Butler et al. utilized a Stetter reaction catalyzed by thiazole carbenes, which suffered from poor catalyst availability and unpleasant odors. Later iterations attempted to optimize this using sodium cyanide (NaCN) as a catalyst promoter, as illustrated in older synthetic schemes. The reliance on such virulent reagents poses severe EHS (Environment, Health, and Safety) risks, generating toxic wastewater that requires costly treatment protocols. Furthermore, alternative pathways involving Friedel-Crafts acylation and bromination steps introduce corrosive reagents like thionyl chloride and liquid bromine. These processes not only generate substantial amounts of acidic waste gas and salt-containing wastewater but also suffer from poor atom economy due to the introduction and subsequent removal of halogen atoms. The cumulative effect of these multi-step sequences is a lower overall yield, typically hovering around 63-66%, coupled with high operational complexity.
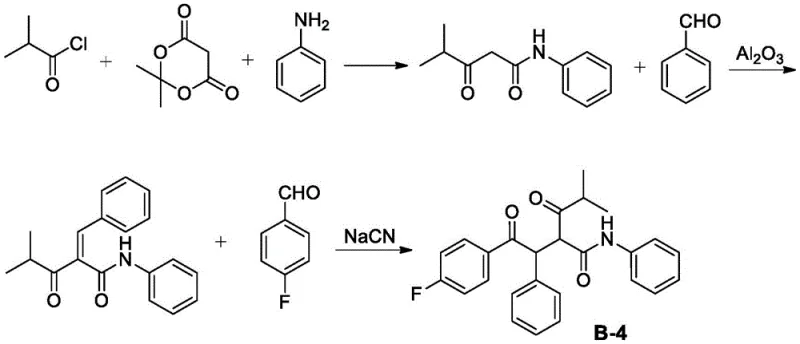
The Novel Approach
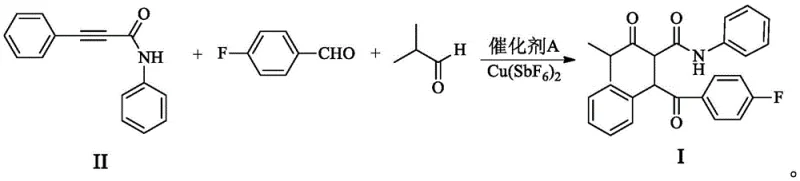
In stark contrast, the methodology disclosed in patent CN111909048B offers a streamlined, one-pot solution that directly assembles the target molecule from three simple starting materials: N-Phenylpropinylamides, 4-fluorobenzaldehyde, and isobutyraldehyde. This multicomponent reaction (MCR) bypasses the need for isolating unstable intermediates or employing hazardous cyanide catalysts. Instead, it utilizes a dual-catalyst system comprising Cu(SbF6)2 and a novel Palladium-ligand complex. This approach aligns perfectly with the principles of green chemistry by maximizing atom utilization and minimizing waste discharge. The reaction proceeds under mild heating conditions (50-60°C) in common aprotic solvents like tetrahydrofuran, delivering the target intermediate with a remarkable molar yield of 80-87%. This substantial improvement in efficiency, combined with the elimination of toxic reagents, represents a significant leap forward in sustainable pharmaceutical manufacturing, offering a robust alternative for reliable pharmaceutical intermediate suppliers seeking to modernize their production capabilities.
Mechanistic Insights into Pd-Cu Synergistic Catalysis
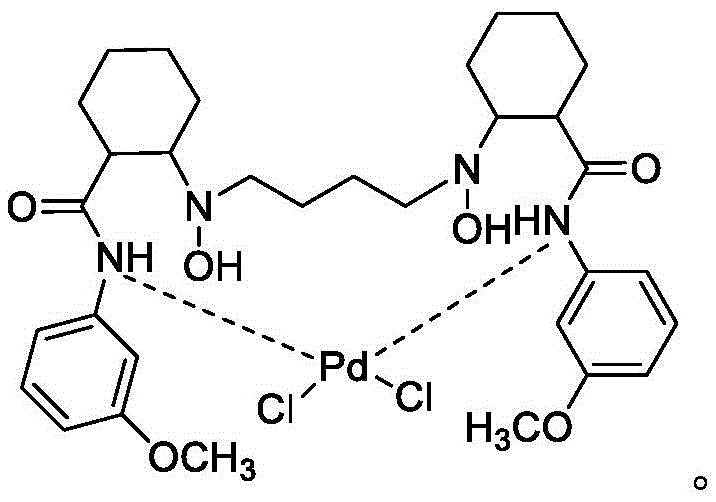
The success of this multicomponent transformation hinges on the intricate interplay between the copper salt and the specialized palladium catalyst. The palladium component is coordinated with a unique N-hydroxylamine cyclohexyl formamide derivative ligand (Formula III), which plays a pivotal role in dictating the reaction's outcome. Unlike simple metal salts, this ligand-metal complex creates a specific steric environment around the catalytic center. Research indicates that the ligand features electron-donating groups (such as methoxy or dimethylamino) at the meta-position, which enhances the electronic density and stability of the complex. When combined with the copper co-catalyst, the system facilitates a highly ordered addition of the aldehyde components across the carbon-carbon triple bond of the propinylamide. This synergy is essential; experiments show that removing either the copper salt or the specific palladium ligand results in a failure to produce the target compound, yielding only simple addition byproducts instead. The reaction requires strictly anhydrous conditions, with water content controlled below 200 ppm, to prevent hydrolysis of the sensitive copper catalyst and ensure smooth progression.
A critical aspect of this mechanism is the exceptional regioselectivity achieved during the bond-forming events. The bulky nature of the 4-fluorobenzaldehyde prevents it from approaching the sterically hindered end of the triple bond near the aniline group. Instead, the smaller isobutyraldehyde reacts preferentially at that position, while the fluorobenzaldehyde adds to the distal carbon. This selectivity is visually conceptualized as a "pocket-like" cavity formed by the transition metal-ligand complex, which effectively wraps the three reactants in a precise orientation. This steric control prevents the formation of regioisomers, such as the compound of Formula VII, which are common impurities in less selective processes. By ensuring that the reaction follows a single, dominant pathway, the catalyst system minimizes the generation of difficult-to-remove impurities, thereby simplifying downstream purification and enhancing the overall purity profile of the final intermediate.
How to Synthesize Atorvastatin Intermediate B-4 Efficiently
The practical implementation of this synthesis involves two primary stages: the preparation of the specialized ligand and the execution of the multicomponent coupling. First, the N-hydroxylamine cyclohexyl formamide ligand is synthesized through a sequence involving the condensation of N-Boc-amino cyclohexyl carboxylic acid with a substituted aniline, followed by Boc-deprotection, alkylation with a dibromoalkane, and final oxidation of the amine to the hydroxylamine. Once the ligand is secured, it is complexed with PdCl2 to form the active Catalyst A. The core synthesis then proceeds by charging the alkyne substrate, both aldehydes, the copper salt, and Catalyst A into a reactor under an inert nitrogen atmosphere. The detailed standardized operating procedures, including specific molar ratios, solvent choices, and workup protocols required to achieve the reported 80-87% yields, are outlined below.
- Prepare the N-hydroxylamine cyclohexyl formamide derivative ligand (Formula III) through condensation, deprotection, alkylation, and oxidation steps.
- Form the active Catalyst A by coordinating the ligand with PdCl2 in anhydrous methanol under nitrogen atmosphere.
- Execute the multicomponent one-pot reaction by combining N-Phenylpropinylamides, 4-fluorobenzaldehyde, and isobutyraldehyde with Cu(SbF6)2 and Catalyst A in THF at 50-60°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented one-pot methodology offers compelling strategic advantages beyond mere technical novelty. The shift from a four-to-five-step linear synthesis to a single-pot operation fundamentally alters the cost structure and risk profile of manufacturing this high-volume intermediate. By consolidating multiple reaction steps into one vessel, manufacturers can drastically reduce the consumption of solvents, energy, and labor hours associated with intermediate isolation and purification. This process intensification leads to substantial cost savings in API manufacturing without compromising on quality. Furthermore, the elimination of hazardous reagents like sodium cyanide and liquid bromine removes significant regulatory burdens and liability risks. Facilities no longer need to invest in specialized containment systems for highly toxic gases or manage complex wastewater streams laden with heavy metals and cyanide residues. This simplification of the EHS landscape ensures greater supply chain reliability and continuity, as production is less susceptible to shutdowns caused by environmental compliance issues or safety incidents.
- Cost Reduction in Manufacturing: The one-pot nature of this reaction eliminates the need for multiple isolation and purification steps that are characteristic of traditional multi-step syntheses. By avoiding the use of expensive and difficult-to-handle reagents like thionyl chloride and specialized thiazole carbene catalysts, the raw material costs are significantly optimized. Additionally, the higher overall yield (80-87% vs. 63-66%) means that less starting material is wasted per kilogram of finished product, directly improving the cost of goods sold (COGS) and enhancing profit margins for large-scale production campaigns.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as isobutyraldehyde and 4-fluorobenzaldehyde, which are readily available in the global market, mitigates the risk of raw material shortages. Unlike proprietary catalysts that may have long lead times or single-source dependencies, the components of this catalytic system are based on standard transition metals and easily synthesized organic ligands. This accessibility ensures that production schedules can be maintained consistently, reducing lead time for high-purity pharmaceutical intermediates and allowing for more responsive inventory management in the face of fluctuating market demand.
- Scalability and Environmental Compliance: The process operates at moderate temperatures (50-60°C) and atmospheric pressure, making it inherently safer and easier to scale from pilot plant to commercial tonnage than exothermic Friedel-Crafts reactions that require strict temperature control and acid gas scrubbing. The significant reduction in three-waste discharge (wastewater, waste gas, and solid waste) aligns with increasingly stringent global environmental regulations. This green chemistry profile facilitates smoother permitting processes and long-term operational sustainability, positioning manufacturers as responsible partners in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this multicomponent synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy routes in terms of safety, efficiency, and product quality. Understanding these nuances is crucial for technical teams evaluating the feasibility of technology transfer.
Q: How does the new one-pot method improve safety compared to traditional Stetter reactions?
A: Traditional Stetter reactions often utilize virulent reagents like NaCN and generate significant hazardous waste. The novel multicomponent one-pot method described in patent CN111909048B eliminates the need for cyanide and corrosive brominating agents, significantly reducing EHS risks and wastewater pollution while maintaining high atom economy.
Q: What represents the key advantage of the Pd-Cu synergistic catalyst system?
A: The synergistic system utilizes a specialized N-hydroxylamine cyclohexyl formamide ligand coordinated with Palladium, working alongside a Copper salt. This creates a unique 'pocket-like' steric environment that ensures extremely high regioselectivity, preventing the formation of unwanted addition byproducts and achieving yields of 80-87%, superior to the 63-66% of conventional multi-step routes.
Q: Is this synthesis method scalable for industrial commercial production?
A: Yes, the process is designed for industrial feasibility. It uses readily available raw materials, operates at moderate temperatures (50-60°C), and simplifies the workflow into a single pot. This reduction in unit operations translates to lower operational complexity and enhanced scalability for large-scale API intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Atorvastatin Intermediate Supplier
As the pharmaceutical industry continues to prioritize sustainability and efficiency, the ability to execute complex transformations like the multicomponent synthesis of Atorvastatin intermediates becomes a key differentiator. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging deep expertise in catalytic process development to bring such innovative routes to life. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of green chemistry are fully realized in practical, large-scale operations. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch meets the exacting standards required for GMP API synthesis.
We invite global partners to collaborate with us to unlock the full potential of this technology. Whether you are looking to optimize an existing supply chain or develop a new source for critical statin intermediates, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can drive value and security for your organization.
