Advanced Catalytic Route for High-Purity 3-Trifluoromethyl-1,2,4-Triazole Pharmaceutical Intermediates
Advanced Catalytic Route for High-Purity 3-Trifluoromethyl-1,2,4-Triazole Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust and efficient synthetic methodologies to construct complex heterocyclic scaffolds that serve as the backbone for next-generation therapeutics. A significant breakthrough in this domain is detailed in patent CN113307778A, which discloses a novel preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. These specific heterocyclic structures are not merely academic curiosities; they are critical pharmacophores found in numerous high-value drugs, including the widely prescribed antidiabetic agent Sitagliptin and various antiviral reagents. The introduction of a trifluoromethyl group into these molecular frameworks is strategically vital, as it dramatically enhances metabolic stability, lipophilicity, and bioavailability, thereby optimizing the pharmacokinetic profile of the final active pharmaceutical ingredient (API). This patent presents a transformative approach that leverages a dual-metal catalytic system to achieve these structures under remarkably mild conditions, offering a compelling alternative to legacy synthetic routes that often suffer from harsh reaction parameters and safety hazards.
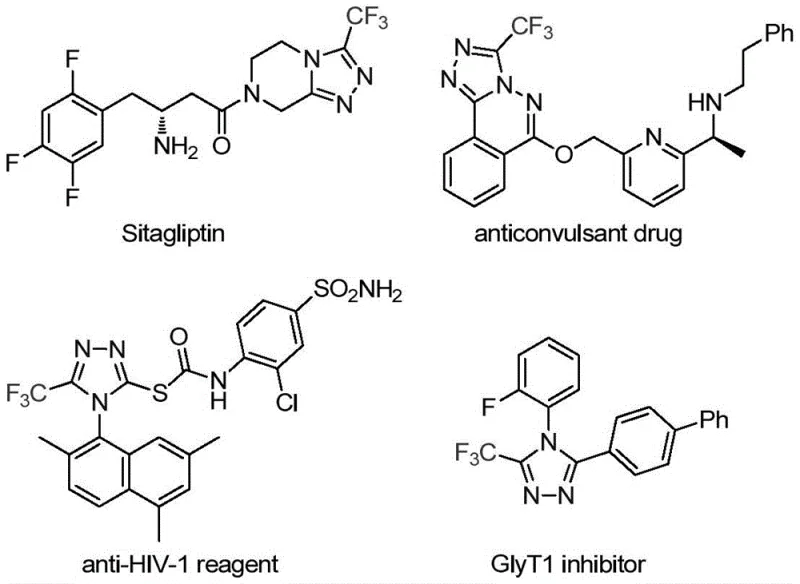
The strategic importance of this technology cannot be overstated for R&D directors and procurement specialists alike. By enabling the efficient construction of these nitrogen-containing heterocycles, the method directly addresses the growing demand for reliable pharmaceutical intermediate suppliers who can deliver high-purity building blocks with consistent quality. The ability to synthesize these compounds with high atom economy and minimal waste generation aligns perfectly with modern green chemistry principles, reducing the environmental footprint of API manufacturing. Furthermore, the versatility of the reaction allows for the facile incorporation of diverse functional groups, providing medicinal chemists with a powerful tool for structure-activity relationship (SAR) studies without being constrained by synthetic limitations. This technological advancement represents a significant leap forward in the cost reduction in pharmaceutical intermediate manufacturing, paving the way for more accessible and affordable healthcare solutions globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted 1,2,4-triazoles has been fraught with significant challenges that hinder efficient large-scale production. Traditional literature methods predominantly rely on the cyclization reaction of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinone. These classical approaches often necessitate the use of hazardous hydrazine derivatives, which pose severe safety risks due to their toxicity and potential explosiveness, requiring specialized handling equipment and rigorous safety protocols that drive up operational costs. Additionally, alternative copper-catalyzed multi-component reactions utilizing diazonium salts and trifluorodiazoethane are plagued by the instability of the diazo precursors, which can decompose violently under certain conditions, creating unacceptable risks for commercial scale-up of complex pharmaceutical intermediates. The reliance on such unstable and dangerous starting materials not only complicates the supply chain but also introduces significant variability in reaction outcomes, leading to inconsistent yields and difficult purification processes that erode profit margins.
The Novel Approach
In stark contrast to these perilous legacy methods, the invention disclosed in CN113307778A introduces a paradigm shift by utilizing cheap and easily obtainable functionalized isonitriles and trifluoroethylimidoyl chloride as the primary starting materials. This innovative strategy employs a sophisticated co-catalytic system comprising molybdenum hexacarbonyl and cuprous acetate, which facilitates a smooth cycloaddition reaction under mild thermal conditions ranging from 70°C to 90°C. The elimination of explosive diazo compounds and toxic hydrazines fundamentally alters the safety profile of the synthesis, making it inherently safer for operators and more compliant with stringent environmental regulations. Moreover, the reaction demonstrates exceptional efficiency and broad substrate tolerance, allowing for the synthesis of various 4-substituted 1,2,4-triazole compounds simply by modifying the starting amine. This flexibility is crucial for rapid drug development cycles, as it enables the quick generation of diverse analog libraries. The simplicity of the post-treatment procedure, involving basic filtration and standard column chromatography, further underscores the practical utility of this method for industrial applications, ensuring that high-purity products can be obtained with minimal downstream processing effort.
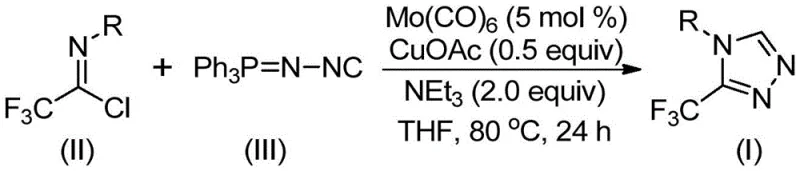
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
The success of this synthetic methodology lies in the intricate interplay between the molybdenum and copper catalysts, which orchestrate a precise sequence of bond-forming events to construct the triazole ring. Mechanistically, the molybdenum hexacarbonyl acts as a potent metal activator for the functionalized isonitrile, forming a reactive metal-isocyanide complex that primes the molecule for subsequent cycloaddition. Simultaneously, the cuprous acetate serves as a Lewis acid promoter, facilitating the nucleophilic attack of the activated isonitrile species onto the electrophilic carbon of the trifluoroethylimidoyl chloride. This cooperative catalysis lowers the activation energy barrier for the [3+2] cycloaddition, allowing the reaction to proceed smoothly at moderate temperatures without the need for extreme heating or pressure. The formation of the five-membered ring intermediate is followed by the elimination of triphenylphosphine oxide, driven by the presence of water in the system, which ultimately yields the stable 3-trifluoromethyl-substituted 1,2,4-triazole product. Understanding this mechanistic pathway is essential for R&D teams aiming to optimize reaction parameters further or adapt the protocol for continuous flow chemistry setups.
From an impurity control perspective, the mildness of the reaction conditions plays a pivotal role in ensuring the high purity of the final product. Harsh acidic or basic conditions often lead to the hydrolysis of sensitive functional groups or the polymerization of reactive intermediates, generating complex impurity profiles that are difficult to separate. However, the neutral to slightly basic environment provided by the triethylamine base in this system preserves the integrity of sensitive substituents such as esters, nitriles, and halogens. The high selectivity of the co-catalytic system minimizes side reactions, resulting in a cleaner crude reaction mixture that requires less aggressive purification techniques. This inherent selectivity is particularly valuable when synthesizing chiral intermediates or molecules with multiple reactive sites, as it reduces the risk of racemization or non-specific functionalization. For quality control laboratories, this translates to simpler analytical methods and faster release times for batches, enhancing the overall agility of the supply chain.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical implementation of this synthesis is straightforward and designed for reproducibility in both laboratory and pilot plant settings. The process begins with the precise weighing of the catalysts and reagents, ensuring the optimal molar ratios identified in the patent are maintained to maximize yield. The reaction is typically conducted in an aprotic solvent such as tetrahydrofuran (THF), which effectively dissolves all components and stabilizes the catalytic species. Maintaining an inert atmosphere is recommended to prevent oxidation of the copper catalyst, although the system shows reasonable robustness. The detailed standardized synthesis steps below outline the exact procedure for achieving optimal results, guiding chemists through the addition sequence, temperature control, and workup protocols necessary to isolate the target compound with high purity.
- Combine molybdenum hexacarbonyl, cuprous acetate, triethylamine, molecular sieves, trifluoroethylimidoyl chloride, and functionalized isonitrile in an organic solvent such as THF.
- Heat the reaction mixture to a temperature between 70°C and 90°C and maintain stirring for a duration of 18 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and purify via column chromatography to isolate the target triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible benefits that extend far beyond the laboratory bench. The primary advantage lies in the substantial cost savings achieved through the use of inexpensive and readily available starting materials. Unlike traditional methods that rely on custom-synthesized or hazardous reagents with volatile pricing, the key inputs for this process—trifluoroethylimidoyl chloride and functionalized isonitriles—are commercially accessible commodities. This stability in raw material sourcing mitigates the risk of supply disruptions and price spikes, ensuring a predictable cost structure for long-term production contracts. Furthermore, the elimination of expensive noble metal catalysts in favor of abundant copper and molybdenum salts drastically reduces the direct material costs associated with the catalytic system, contributing to a more competitive pricing model for the final intermediate.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the simplification of the process workflow. By avoiding the need for specialized safety infrastructure required for handling explosive diazo compounds or toxic hydrazines, manufacturers can significantly lower their capital expenditure and operational overheads. The mild reaction conditions also translate to reduced energy consumption, as the process does not require cryogenic cooling or high-temperature heating, leading to a smaller carbon footprint and lower utility bills. Additionally, the high reaction efficiency minimizes the loss of valuable starting materials, improving the overall mass balance and reducing the cost of goods sold (COGS). These cumulative factors create a leaner manufacturing process that enhances profitability while maintaining high quality standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and scalability of the reaction. The patent data indicates that the method performs consistently across a wide range of substrates, meaning that a single manufacturing line can be utilized to produce a diverse portfolio of triazole derivatives without extensive retooling. This flexibility allows suppliers to respond rapidly to changing market demands or urgent customer requests for specific analogs. The use of common solvents like THF and standard purification techniques like column chromatography ensures that the process can be easily transferred between different manufacturing sites or contract manufacturing organizations (CMOs) without significant technology transfer hurdles. This interoperability reduces the risk of single-source dependency and strengthens the overall security of the supply chain.
- Scalability and Environmental Compliance: Scaling chemical processes from gram to kilogram or ton scale often introduces unforeseen challenges, but this method is inherently designed for expansion. The exothermic profile of the reaction is manageable, and the absence of gas-evolving steps or highly unstable intermediates simplifies reactor design and safety engineering. From an environmental standpoint, the reduction in hazardous waste generation aligns with increasingly strict global regulations on chemical manufacturing. The ability to recycle solvents and the use of less toxic reagents facilitate easier waste treatment and disposal, reducing the environmental compliance burden. This sustainability aspect is not only ethically sound but also commercially advantageous, as it appeals to pharmaceutical clients who are under pressure to meet their own corporate social responsibility (CSR) and environmental, social, and governance (ESG) goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the method's capabilities and limitations. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this route into their existing production pipelines or R&D workflows.
Q: What are the key advantages of this Mo/Cu co-catalyzed method over traditional triazole synthesis?
A: This novel method utilizes mild reaction conditions (70-90°C) and avoids the use of hazardous hydrazine derivatives or unstable diazonium salts often required in conventional routes. The co-catalytic system ensures high reaction efficiency and broad substrate tolerance, significantly simplifying the purification process and reducing safety risks associated with explosive intermediates.
Q: Is this synthetic route scalable for commercial production of API intermediates?
A: Yes, the patent explicitly demonstrates that the method can be expanded to gram-level reactions with high efficiency. The use of commercially available and inexpensive catalysts like cuprous acetate and molybdenum hexacarbonyl, combined with simple post-treatment procedures like filtration and column chromatography, makes the process highly amenable to scale-up for industrial manufacturing.
Q: What types of substituents are tolerated on the aromatic ring of the starting materials?
A: The method exhibits excellent functional group tolerance, accommodating a wide range of substituents including alkyl groups (methyl, ethyl, t-butyl), alkoxy groups, halogens (fluoro, chloro), and electron-withdrawing groups like nitro and methylthio. This versatility allows for the synthesis of diverse 3-trifluoromethyl-1,2,4-triazole derivatives tailored for specific drug discovery programs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of new medicines. Our team of expert chemists has thoroughly analyzed the technology disclosed in CN113307778A and is fully prepared to leverage this advanced Mo/Cu co-catalyzed methodology for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from discovery to clinical supply is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-trifluoromethyl-1,2,4-triazole delivered meets the highest industry standards for potency and impurity profiles.
We invite you to collaborate with us to unlock the full potential of this synthetic route for your specific drug candidates. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements and timeline. We encourage you to reach out today to discuss your specific needs,索取 specific COA data for our reference standards, and review our comprehensive route feasibility assessments. Let us be your trusted partner in navigating the complexities of fine chemical synthesis, delivering value through innovation, reliability, and scientific excellence.
