Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Pharmaceutical Applications
Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Pharmaceutical Applications
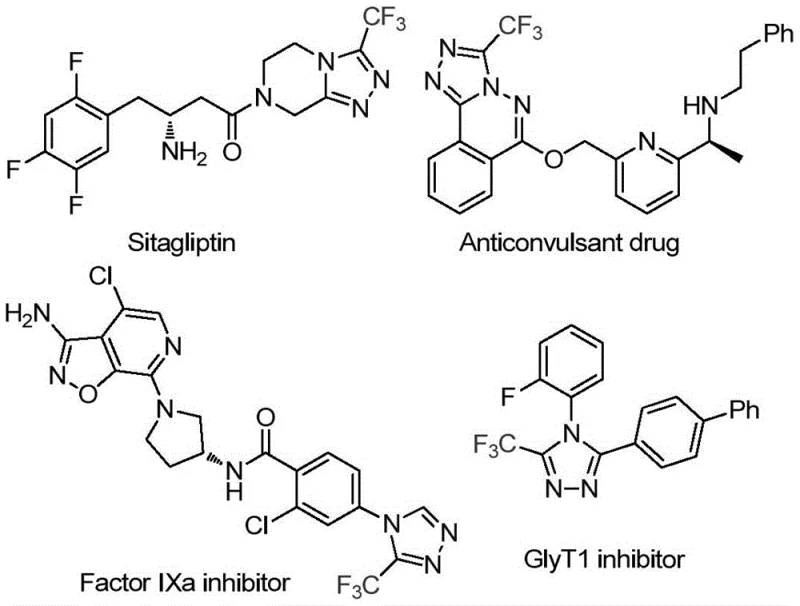
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for heterocyclic scaffolds that serve as core structures in bioactive molecules. Patent CN114920707B discloses a groundbreaking preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds, addressing critical bottlenecks in traditional heterocycle synthesis. This technology leverages the ubiquitous solvent N,N-dimethylformamide (DMF) not merely as a medium, but as an active carbon source, enabling a tandem cyclization reaction promoted by molecular iodine. The significance of this development cannot be overstated for R&D teams focusing on fluorinated drug candidates, as the trifluoromethyl group is a privileged motif known to enhance metabolic stability and lipophilicity in lead compounds. By operating under air atmosphere and utilizing commercially available starting materials, this process represents a paradigm shift towards greener and more economically viable manufacturing of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 1,2,4-triazole rings, particularly those bearing trifluoromethyl groups, has relied on multi-step sequences involving hazardous reagents or苛刻 conditions that complicate scale-up. Traditional protocols often necessitate the use of specialized one-carbon synthons that are expensive, moisture-sensitive, or difficult to handle on a large industrial scale. Furthermore, many existing methodologies require strict anhydrous and anaerobic environments, demanding specialized equipment such as gloveboxes or Schlenk lines, which drastically increases capital expenditure and operational complexity. The reliance on transition metal catalysts in some conventional routes also introduces the risk of heavy metal contamination, necessitating costly purification steps to meet stringent regulatory limits for Active Pharmaceutical Ingredients (APIs). These factors collectively contribute to extended lead times and inflated production costs, creating significant friction in the supply chain for critical drug intermediates.
The Novel Approach
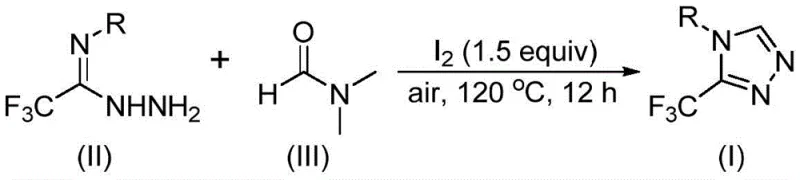
In stark contrast, the method described in patent CN114920707B offers a streamlined, atom-economical solution that fundamentally simplifies the synthetic landscape. The core innovation lies in the dual functionality of DMF, which acts simultaneously as the polar aprotic solvent and the donor of the carbon atom required for ring closure. This eliminates the need for external carbon sources, thereby reducing raw material costs and waste generation. The reaction is promoted by molecular iodine, a benign and inexpensive reagent, and proceeds efficiently under ambient air conditions at temperatures between 110°C and 130°C. This operational simplicity allows for the direct conversion of trifluoroethyliminohydrazides into the desired triazole products with high efficiency. The broad substrate tolerance means that diverse functional groups can be incorporated without protecting group strategies, further shortening the synthetic route and enhancing the overall yield profile for complex molecule synthesis.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
From a mechanistic perspective, this transformation is a fascinating example of solvent-participating organic synthesis where the solvent molecules are actively consumed to build the molecular skeleton. The reaction pathway involves the activation of DMF by molecular iodine, which facilitates the cleavage of specific bonds to generate reactive intermediates. Depending on the specific reaction trajectory, either the formyl group or the N-methyl group of the DMF molecule can serve as the carbon source. When the formyl group participates, it undergoes condensation with the trifluoroethyliminohydrazide to form a hydrazone intermediate, followed by intramolecular cyclization and elimination of dimethylamine. Alternatively, when the N-methyl group is utilized, DMF reacts with iodine to form an amine salt, which then undergoes nucleophilic addition and subsequent elimination steps to generate an azadiene intermediate before final oxidative aromatization. Understanding these dual pathways is crucial for process chemists aiming to optimize reaction parameters and minimize byproduct formation during scale-up.
Impurity control is inherently managed by the simplicity of the reagent system. Since DMF is used in large excess as the solvent, the kinetics favor the desired intermolecular interactions over competing side reactions that might occur with stoichiometric reagents. The use of iodine as a promoter ensures a clean oxidation state management during the aromatization step, preventing the accumulation of partially oxidized species that often plague metal-catalyzed processes. Moreover, the absence of sensitive organometallic reagents reduces the likelihood of decomposition products arising from hydrolysis or oxidation by atmospheric moisture. For R&D directors, this implies a cleaner crude reaction profile, which translates directly to simpler downstream processing and higher final purity of the API intermediate, ensuring compliance with strict pharmacopoeial standards without the need for extensive recrystallization cycles.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical implementation of this synthesis is designed for ease of execution in standard laboratory and pilot plant settings. The protocol requires mixing trifluoroethyliminohydrazide with molecular iodine in DMF, followed by heating under air. The detailed standardized synthesis steps, including precise molar ratios and workup procedures, are outlined below to ensure reproducibility and safety during technology transfer.
- Combine molecular iodine, trifluoroethyliminohydrazide, and excess DMF solvent in a reaction vessel under air atmosphere.
- Heat the reaction mixture to a temperature range of 110-130°C and maintain stirring for 10 to 15 hours to ensure complete conversion.
- Upon completion, perform standard post-treatment including filtration, washing, and column chromatography purification to isolate the target triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic methodology offers tangible strategic benefits that extend beyond mere chemical curiosity. The primary value proposition lies in the drastic simplification of the raw material portfolio. By utilizing DMF, a commodity chemical available in bulk quantities globally, the dependency on niche, high-cost specialty reagents is eliminated. This shift significantly mitigates supply chain risks associated with single-source suppliers or geopolitical disruptions affecting complex reagent availability. Furthermore, the operational robustness of the process—specifically its tolerance to air and moisture—means that manufacturing can be conducted in standard stainless steel reactors without the need for exotic lining or inert gas blanketing systems, leading to substantial capital savings and reduced maintenance overheads.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive catalysts and the reduction of unit operations. Since DMF serves as both solvent and reactant, the material cost per kilogram of product is significantly lowered compared to traditional methods requiring stoichiometric amounts of formylating agents. Additionally, the removal of transition metals from the process flow negates the need for expensive scavenging resins or activated carbon treatments to remove metal residues, which are often required to meet regulatory specifications for pharmaceutical ingredients. This streamlined workflow reduces both direct material costs and indirect processing costs, resulting in a more competitive pricing structure for the final intermediate.
- Enhanced Supply Chain Reliability: The reliance on universally available starting materials ensures a stable and resilient supply chain. Trifluoroethyliminohydrazides can be readily synthesized from common aromatic amines and hydrazine hydrate, commodities that are produced at massive scales globally. This abundance prevents bottlenecks that often occur with proprietary or custom-synthesized building blocks. Moreover, the simplified reaction conditions reduce the technical barrier for contract manufacturing organizations (CMOs) to adopt the process, increasing the number of qualified suppliers in the market and fostering a competitive environment that further secures supply continuity for long-term projects.
- Scalability and Environmental Compliance: Scaling this reaction from gram to ton scale is straightforward due to the absence of exothermic hazards associated with strong bases or pyrophoric reagents. The use of molecular iodine, while requiring careful handling, is manageable with standard scrubbing systems, and the solvent DMF can be recovered and recycled efficiently, aligning with green chemistry principles. The reduction in waste generation, particularly the avoidance of heavy metal sludge, simplifies environmental compliance and waste disposal logistics. This eco-friendly profile not only reduces disposal costs but also enhances the sustainability credentials of the final drug product, a factor increasingly valued by end-users and regulatory bodies alike.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethyl-triazole synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear picture of the method's capabilities and limitations for potential adopters.
Q: What is the primary advantage of using DMF in this triazole synthesis?
A: DMF serves a dual role as both the reaction solvent and the carbon source (providing either the formyl or N-methyl group), which eliminates the need for separate, expensive one-carbon reagents and simplifies the workup process significantly.
Q: Does this reaction require strict anhydrous or anaerobic conditions?
A: No, one of the key operational benefits of this patented method is that it proceeds efficiently under standard air atmosphere without the need for rigorous anhydrous or oxygen-free environments, greatly reducing equipment and operational costs.
Q: What types of substituents are tolerated on the aromatic ring of the substrate?
A: The method demonstrates broad substrate scope, successfully accommodating various substituents including alkyl, alkoxy, alkylthio, halogens (fluorine, chlorine), and trifluoromethyl groups at ortho, meta, or para positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development timelines. Our team of expert process chemists has extensively evaluated the technology disclosed in CN114920707B and is fully equipped to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from clinical trials to market launch. Our state-of-the-art facilities are designed to handle fluorinated chemistry safely and efficiently, supported by stringent purity specifications and rigorous QC labs that guarantee every batch meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this cost-effective synthesis for your next-generation therapeutics. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing expertise can optimize your supply chain and reduce your overall cost of goods sold.
