Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Pharmaceutical Manufacturing
Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust and economically viable synthetic routes for heterocyclic scaffolds that serve as critical building blocks for drug discovery. Patent CN111675662B introduces a transformative preparation method for 2-trifluoromethyl substituted quinazolinone compounds, addressing key limitations in current synthetic methodologies. This innovation leverages an inexpensive iron-based catalytic system to facilitate the cyclization of readily available trifluoroethylimidoyl chloride and isatin derivatives. The introduction of the trifluoromethyl group is strategically significant, as it enhances the electronegativity, metabolic stability, and lipophilicity of the resulting bioactive molecules. By shifting away from expensive reagents and harsh conditions, this technology provides a sustainable pathway for producing high-value pharmaceutical intermediates. The detailed disclosure within the patent outlines a procedure that balances high conversion rates with operational simplicity, making it an attractive candidate for integration into existing supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinone derivatives bearing trifluoromethyl functionalities has relied heavily on cyclization reactions involving synthons such as trifluoroacetic anhydride or ethyl trifluoroacetate. These traditional approaches often suffer from severe reaction conditions that necessitate rigorous temperature control and specialized equipment to manage safety risks. Furthermore, the substrates required for these transformations, such as anthranilamide or isatoic anhydride derivatives, can be prohibitively expensive or difficult to source in bulk quantities. A major drawback observed in literature precedents is the narrow substrate scope, where the presence of sensitive functional groups often leads to decomposition or side reactions, drastically reducing the overall yield. Consequently, the purification processes become complex and costly, involving extensive chromatography to remove impurities generated by non-selective reagents. These cumulative factors create significant bottlenecks for procurement teams aiming to secure reliable supplies of these intermediates at a competitive cost structure.
The Novel Approach
In stark contrast to legacy methods, the novel approach described in the patent utilizes a synergistic combination of ferric chloride and sodium hydride to drive the reaction forward under relatively mild conditions. This methodology employs trifluoroethylimidoyl chloride and isatin as starting materials, both of which are commercially accessible and cost-effective compared to traditional trifluoromethylating agents. The reaction proceeds through a tandem sequence involving alkali-promoted carbon-nitrogen bond formation followed by iron-catalyzed decarbonylation and cyclization. 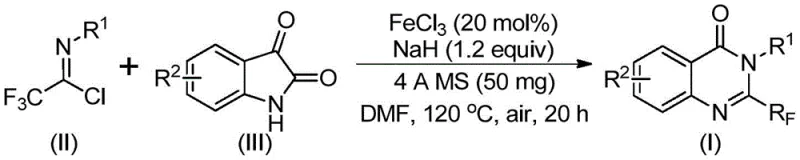 As illustrated in the reaction scheme, the process operates effectively in polar aprotic solvents like DMF and tolerates exposure to air, eliminating the need for stringent inert atmosphere protocols. This simplification of the operational environment significantly reduces the technical barrier for scale-up, allowing for smoother transitions from laboratory benchtop to pilot plant reactors without compromising product integrity or yield.
As illustrated in the reaction scheme, the process operates effectively in polar aprotic solvents like DMF and tolerates exposure to air, eliminating the need for stringent inert atmosphere protocols. This simplification of the operational environment significantly reduces the technical barrier for scale-up, allowing for smoother transitions from laboratory benchtop to pilot plant reactors without compromising product integrity or yield.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The mechanistic pathway of this transformation is driven by the Lewis acidic nature of the ferric chloride catalyst, which activates the carbonyl species towards nucleophilic attack. Initially, the sodium hydride acts as a strong base to deprotonate the isatin nitrogen, generating a nucleophilic anion that attacks the electrophilic carbon of the trifluoroethylimidoyl chloride. This step forms a transient trifluoroacetamidine intermediate, which is crucial for the subsequent ring closure. The iron catalyst then facilitates a decarbonylation event, effectively removing the oxygen atom and enabling the formation of the fused quinazolinone ring system. This specific catalytic cycle is highly efficient because it avoids the formation of stable byproducts that typically poison other catalytic systems. The presence of 4A molecular sieves in the reaction mixture plays a critical role in sequestering moisture, thereby preventing the hydrolysis of the sensitive imidoyl chloride starting material and ensuring high conversion rates throughout the extended reaction period.
From an impurity control perspective, the high selectivity of this iron-catalyzed system ensures that the final product profile is clean, with minimal generation of regioisomers or over-reacted species. The tolerance for various substituents on the aromatic rings, including electron-withdrawing halogens and electron-donating alkyl groups, suggests that the electronic properties of the substrate do not significantly hinder the catalytic turnover. 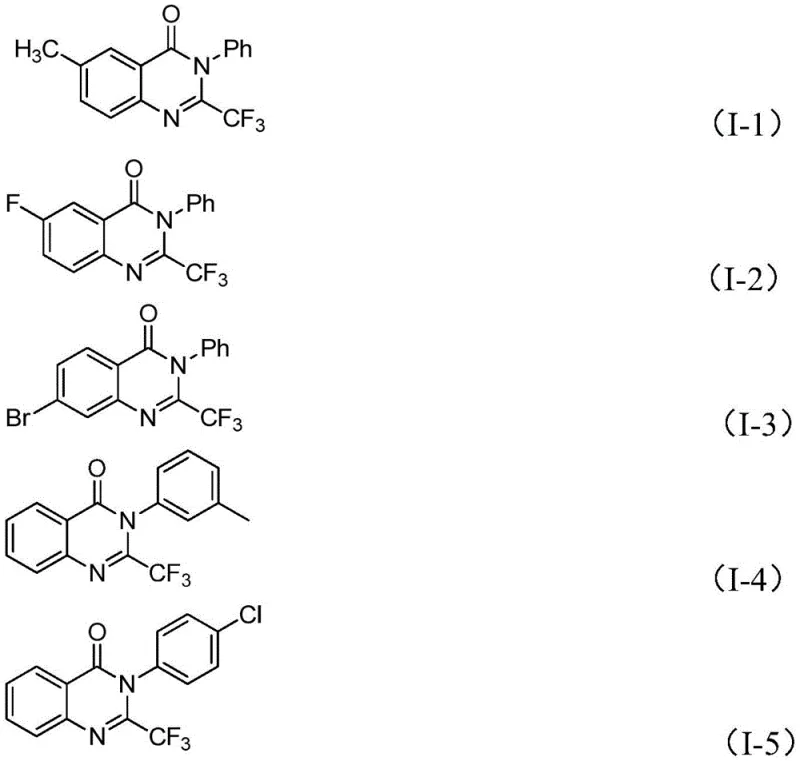 This robustness is evidenced by the successful synthesis of diverse derivatives such as compounds I-1 through I-5, which maintain high purity levels even with structural variations. For R&D directors, this implies that the method can be adapted for library synthesis without requiring extensive re-optimization for each new analog. The ability to predictably manage the impurity profile simplifies the downstream purification strategy, potentially allowing for crystallization-based purification rather than resource-intensive column chromatography in larger batches.
This robustness is evidenced by the successful synthesis of diverse derivatives such as compounds I-1 through I-5, which maintain high purity levels even with structural variations. For R&D directors, this implies that the method can be adapted for library synthesis without requiring extensive re-optimization for each new analog. The ability to predictably manage the impurity profile simplifies the downstream purification strategy, potentially allowing for crystallization-based purification rather than resource-intensive column chromatography in larger batches.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for executing this transformation with high reproducibility and safety. The process begins with the precise weighing of ferric chloride and sodium hydride, which are added to a reaction vessel along with 4A molecular sieves to ensure anhydrous conditions. Trifluoroethylimidoyl chloride and the chosen isatin derivative are then introduced into the organic solvent, typically DMF, and the mixture is stirred to ensure homogeneity. The reaction is initiated at a lower temperature of 40°C for a preliminary period, allowing for the controlled formation of the intermediate before the energy barrier for cyclization is overcome by heating to 120°C. Detailed standardized synthetic steps for optimizing this process are provided in the guide below.
- Mix ferric chloride, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin derivative in an organic solvent like DMF.
- Stir the reaction mixture at 40°C for approximately 10 hours to initiate the bond formation.
- Heat the mixture to 120°C and continue reacting for 20 hours under air atmosphere to complete the cyclization and decarbonylation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron-catalyzed methodology presents a compelling value proposition centered on cost efficiency and supply reliability. The substitution of precious metal catalysts with abundant iron salts results in a drastic reduction in raw material costs, which directly impacts the bottom line of the manufacturing budget. Additionally, the use of commodity chemicals like isatin and simple aromatic amines for the precursor synthesis ensures that the supply chain is not vulnerable to the volatility often associated with specialized fluorinating reagents. The operational simplicity of the reaction, which does not require glovebox techniques or high-pressure equipment, further lowers the capital expenditure required for production facilities. These factors combine to create a resilient supply model that can withstand market fluctuations and demand surges.
- Cost Reduction in Manufacturing: The elimination of expensive palladium or rhodium catalysts in favor of ferric chloride represents a fundamental shift in the cost structure of quinazolinone production. Since iron is one of the most abundant and inexpensive metals available, the catalyst loading cost becomes negligible compared to traditional noble metal systems. Furthermore, the high yields reported across multiple examples indicate efficient atom economy, meaning less starting material is wasted as byproduct. This efficiency translates to lower waste disposal costs and reduced consumption of solvents per kilogram of product produced. The overall process economics are further improved by the ability to run the reaction under air, removing the need for costly nitrogen or argon purging systems.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, specifically isatin derivatives and trifluoroethylimidoyl chlorides, are established commodities with multiple global suppliers. This diversification of the supply base mitigates the risk of single-source dependency that often plagues the procurement of exotic heterocyclic building blocks. The robustness of the reaction conditions means that production schedules are less likely to be disrupted by minor variations in environmental parameters or reagent quality. Consequently, manufacturers can offer more consistent lead times and maintain higher safety stock levels without fear of rapid degradation of sensitive intermediates. This reliability is crucial for pharmaceutical clients who require uninterrupted supply to meet their own clinical or commercial timelines.
- Scalability and Environmental Compliance: The transition from milligram to kilogram scale is facilitated by the thermal stability of the reaction mixture and the absence of hazardous high-pressure steps. The use of DMF as a solvent, while requiring careful handling, is well-understood in industrial settings with established recovery and recycling protocols. The iron catalyst leaves minimal heavy metal residue in the final product, simplifying the regulatory compliance process for API manufacturing where metal limits are strictly enforced. This ease of purification reduces the number of processing steps required to meet pharmacopeial standards, accelerating the time to market for new drug candidates. The overall green chemistry profile is enhanced by the high selectivity, which minimizes the generation of toxic organic waste streams.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their current manufacturing portfolios. The responses cover aspects ranging from catalyst selection to substrate compatibility and post-processing requirements.
Q: What are the primary advantages of using FeCl3 over traditional catalysts for this synthesis?
A: The use of ferric chloride offers a significant economic advantage due to its low cost compared to precious metal catalysts, while maintaining high catalytic efficiency and broad functional group tolerance.
Q: Can this method tolerate diverse substituents on the aromatic rings?
A: Yes, the protocol demonstrates excellent substrate scope, successfully accommodating electron-donating and electron-withdrawing groups such as methyl, halogen, methoxy, and nitro groups at various positions.
Q: Is this process suitable for large-scale industrial production?
A: The method utilizes readily available starting materials and simple reaction conditions without requiring inert gas protection, making it highly amenable to scale-up for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes like the one described in CN111675662B for advancing pharmaceutical pipelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 2-trifluoromethyl quinazolinone meets the highest international standards. Our commitment to quality assurance means that you can rely on us for consistent material performance in your downstream drug synthesis applications.
We invite you to contact our technical procurement team to discuss how we can tailor this technology to your specific requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this iron-catalyzed method for your specific volume needs. We are ready to provide specific COA data and route feasibility assessments to support your decision-making process. Let us partner with you to optimize your supply chain and accelerate your path to market with high-quality chemical intermediates.
