Revolutionizing 5-Trifluoromethyl-1,2,3-Triazole Synthesis: A Metal-Free, Scalable Route for Advanced Pharmaceutical Intermediates
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable pathways to access fluorinated heterocycles, particularly 1,2,3-triazoles, due to their profound impact on metabolic stability and bioavailability. Patent CN113121462B introduces a transformative preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds that fundamentally shifts the synthetic paradigm away from hazardous azide chemistry. This innovation leverages a base-promoted cyclization strategy using readily available trifluoroethylimidoyl chlorides and diazo compounds, effectively bypassing the need for toxic organic azides or expensive transition metal catalysts. For R&D directors and process chemists, this represents a significant leap forward in designing safer, more efficient routes for complex molecular scaffolds. The methodology not only addresses critical safety concerns associated with traditional triazole synthesis but also offers a streamlined workflow that is highly amenable to commercial scale-up, ensuring a reliable supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
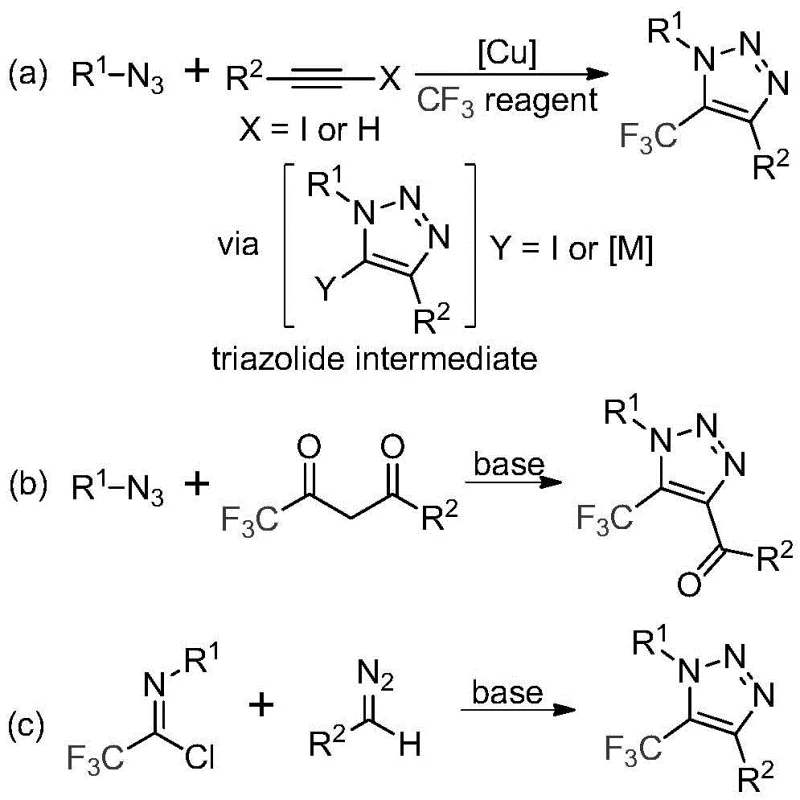
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,3-triazoles has relied heavily on copper-catalyzed [3+2] cycloaddition reactions between alkynes and organic azides, followed by trifluoromethylation, or organocatalytic 1,3-dipolar cycloadditions involving azides and trifluoromethyl ketones. These conventional pathways are fraught with significant drawbacks that hinder efficient manufacturing. Primarily, the reliance on organic azides introduces severe safety hazards due to their potential explosivity and toxicity, necessitating specialized handling protocols and equipment that drive up operational costs. Furthermore, the use of copper catalysts creates a downstream burden; removing trace heavy metals to meet stringent pharmaceutical specifications requires additional purification steps, such as scavenging or recrystallization, which inevitably reduce overall yield and increase waste generation. The complexity of managing these hazardous reagents and metal residues makes traditional methods less attractive for large-scale production of API intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN113121462B utilizes a metal-free, base-promoted strategy that circumvents these historical bottlenecks. By employing trifluoroethylimidoyl chlorides and diazo compounds as starting materials, the process eliminates the need for explosive azides and toxic transition metals entirely. The reaction proceeds under mild thermal conditions, typically around 60°C in acetonitrile, utilizing cesium carbonate as a benign promoter. This shift not only enhances the safety profile of the synthesis but also drastically simplifies the workup procedure, as there are no metal catalysts to remove. The result is a cleaner reaction profile with high atom economy, allowing for the direct isolation of the target 5-trifluoromethyl-1,2,3-triazole derivatives with excellent purity. This approach aligns perfectly with modern green chemistry principles, offering a sustainable alternative for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Base-Promoted 5-endo-dig Cyclization
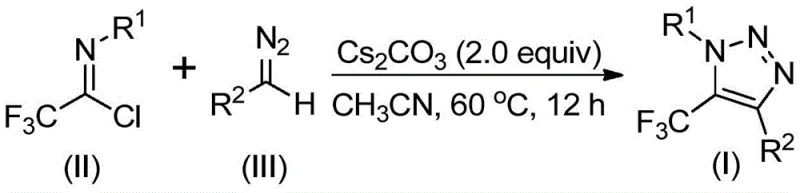
The mechanistic pathway of this transformation is a fascinating example of base-mediated heterocycle formation that avoids the pitfalls of radical or metal-mediated processes. The reaction likely initiates with the deprotonation of the diazo compound by the cesium carbonate base, generating a nucleophilic diazo species. This activated intermediate then undergoes an intermolecular nucleophilic addition to the electrophilic carbon of the trifluoroethylimidoyl chloride, accompanied by the elimination of a chloride ion. This critical step establishes the carbon-carbon bond necessary for ring closure. Subsequently, the intermediate undergoes an intramolecular 5-endo-dig cyclization, where the terminal nitrogen of the diazo moiety attacks the nitrile-like carbon, closing the five-membered triazole ring. This specific cyclization mode is energetically favorable under the promoted conditions and leads directly to the observed 5-trifluoromethyl substitution pattern. Understanding this mechanism is vital for process optimization, as it highlights the importance of base strength and solvent polarity in facilitating the initial nucleophilic attack and subsequent ring closure without side reactions.
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed variants. Since no transition metals are involved, the risk of metal-induced side reactions, such as homocoupling of alkynes or decomposition of sensitive functional groups, is minimized. The primary impurities are likely to be unreacted starting materials or hydrolysis products of the imidoyl chloride, both of which are easily removed during standard aqueous workup or silica gel chromatography. The absence of metal complexes means the final product does not require rigorous testing for heavy metal residuals, a common regulatory hurdle in API synthesis. This clean impurity profile ensures that the resulting high-purity pharmaceutical intermediates meet the strict quality standards required for downstream drug development, reducing the analytical burden on QC laboratories and accelerating the path to clinical trials.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazoles Efficiently
To implement this synthesis effectively, precise control over reaction stoichiometry and conditions is essential to maximize yield and minimize byproduct formation. The patent data indicates that a molar ratio of trifluoroethylimidoyl chloride to diazo compound to cesium carbonate of approximately 1:1.5:2 is optimal, ensuring complete consumption of the limiting imidoyl chloride while maintaining a basic environment conducive to cyclization. The choice of solvent is also critical; while various aprotic solvents can support the reaction, acetonitrile has been identified as the superior medium, providing the best balance of solubility and reaction rate at 60°C. Detailed standardized synthesis steps see the guide below.
- Charge a reaction vessel with cesium carbonate (2.0 equiv), trifluoroethylimidoyl chloride, and the corresponding diazo compound (1.5 equiv) in anhydrous acetonitrile.
- Heat the reaction mixture to 60°C and maintain stirring for 12 hours to ensure complete conversion via 5-endo-dig cyclization.
- Filter the mixture to remove inorganic salts, concentrate the filtrate, and purify the crude residue via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis route offers compelling strategic advantages that extend beyond simple chemical efficiency. The elimination of copper catalysts and organic azides translates directly into a more resilient and cost-effective supply chain. By removing the dependency on specialized, hazard-classified reagents, companies can source raw materials from a broader range of suppliers, mitigating the risk of shortages and price volatility associated with niche chemicals. Furthermore, the simplified purification process reduces the consumption of auxiliary materials like metal scavengers and specialized filtration media, contributing to substantial cost savings in manufacturing overheads.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the complete removal of transition metal catalysts. In traditional processes, the cost of the catalyst itself, combined with the expensive downstream processing required to reduce metal content to ppm levels, constitutes a major portion of the COGS. By utilizing inexpensive cesium carbonate instead, the process eliminates the need for costly metal scavenging resins and the associated validation testing. Additionally, the higher reaction efficiency and simpler workup reduce solvent usage and energy consumption per kilogram of product, leading to a leaner, more profitable manufacturing model for complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, specifically trifluoroethylimidoyl chlorides and diazo compounds, are derived from commodity chemicals and are generally more stable and easier to transport than organic azides. Organic azides often require cold chain logistics and special hazardous material handling, which complicates international shipping and increases lead times. By switching to stable, non-explosive precursors, the supply chain becomes more robust, allowing for larger batch sizes and longer storage periods without degradation. This stability ensures a continuous flow of materials to production sites, reducing the risk of unplanned downtime and enabling better inventory management for high-purity API intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental risks, but this base-promoted method is inherently safer for large-scale operations. The absence of explosive azides removes a major barrier to increasing batch sizes from grams to tons. Moreover, the reduction in heavy metal waste aligns with increasingly strict environmental regulations regarding effluent discharge. Facilities can operate with a smaller environmental footprint, avoiding the costs associated with treating metal-contaminated waste streams. This compliance advantage not only protects the company from regulatory fines but also enhances its reputation as a sustainable manufacturer, which is increasingly important for partnerships with major multinational pharmaceutical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and mechanistic understanding provided in the patent literature, aiming to clarify the practical benefits for industrial application.
Q: Why is this metal-free method superior to traditional copper-catalyzed routes?
A: Traditional methods often rely on copper catalysts and toxic organic azides, requiring expensive heavy metal scavenging steps to meet pharmaceutical purity standards. This base-promoted method eliminates transition metals entirely, simplifying downstream purification and reducing regulatory burden.
Q: What are the key safety advantages of using diazo compounds over azides in this synthesis?
A: Organic azides are notoriously unstable and explosive, posing significant safety risks during scale-up. The disclosed method utilizes stable diazo compounds and trifluoroethylimidoyl chlorides, significantly enhancing operational safety for commercial manufacturing.
Q: Can this process be scaled for industrial production of API intermediates?
A: Yes, the patent explicitly demonstrates scalability to the gram level with mild reaction conditions (60°C) and commercially available reagents. The absence of sensitive catalysts and the use of standard solvents like acetonitrile facilitate straightforward technology transfer to pilot and production plants.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to stay competitive in the global fine chemicals market. Our team of expert process chemists has thoroughly analyzed the technology disclosed in CN113121462B and is fully prepared to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 5-trifluoromethyl-1,2,3-triazole intermediate delivered meets the highest industry standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this cutting-edge chemistry for your next project. Whether you require custom synthesis of specific derivatives or large-scale supply of the core scaffold, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can optimize your supply chain and accelerate your drug development timeline.
