Scalable Base-Promoted Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Pharmaceutical Applications
Scalable Base-Promoted Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Pharmaceutical Applications
The rapid evolution of medicinal chemistry demands increasingly sophisticated heterocyclic scaffolds, particularly those incorporating fluorine motifs to enhance metabolic stability and bioavailability. A significant breakthrough in this domain is detailed in Chinese Patent CN113121462B, which discloses a novel preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds. These structures are pivotal in the development of potent pharmaceutical agents, including selective beta-3 adrenergic receptor agonists, as well as advanced agrochemicals functioning as fungicides and herbicides. The patent introduces a transformative synthetic strategy that bypasses the limitations of traditional transition-metal catalysis, offering a streamlined, metal-free pathway that utilizes cesium carbonate as a benign promoter. For global procurement teams and R&D directors seeking a reliable pharmaceutical intermediate supplier, this technology represents a critical advancement in accessing high-value building blocks with improved safety profiles and reduced environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,3-triazoles has relied heavily on copper-catalyzed azide-alkyne cycloaddition (CuAAC) reactions or organocatalytic variants involving toxic azide precursors. As illustrated in the prior art comparison, these conventional routes typically involve the reaction of alkynes with organic azides in the presence of copper catalysts to form triazole metal complexes, followed by trifluoromethylation. Alternatively, some methods utilize 1,3-dipolar cycloadditions between azides and trifluoromethyl ketones. Both approaches suffer from severe drawbacks: the handling of organic azides poses significant safety hazards due to their potential explosiveness, and the reliance on copper catalysts introduces the risk of heavy metal contamination in the final active pharmaceutical ingredient (API). Furthermore, the removal of residual copper to meet stringent regulatory standards adds complex purification steps, driving up both production costs and lead times for cost reduction in API manufacturing.
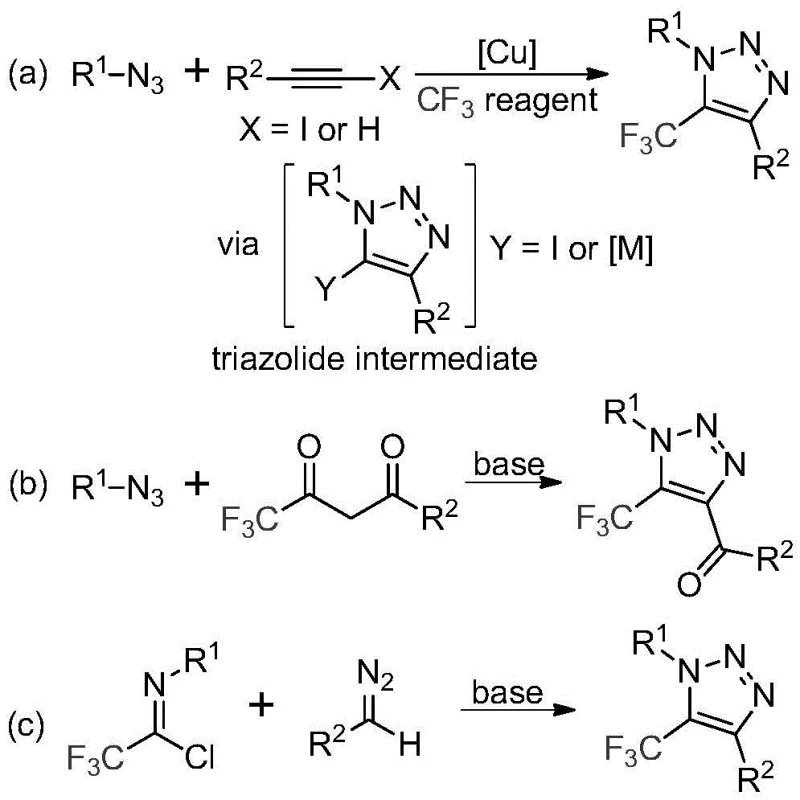
The Novel Approach
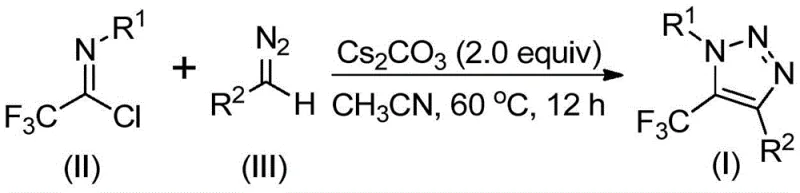
In stark contrast to these hazardous and complex legacy methods, the invention described in CN113121462B employs a simple, efficient, and base-promoted synthesis that completely eliminates the need for metals, external azides, or specialized trifluoromethylating reagents. The core innovation lies in the direct coupling of readily available trifluoroethylimidoyl chlorides with diazo compounds. This reaction proceeds smoothly under mild thermal conditions, typically between 50°C and 70°C, using cesium carbonate as the sole promoter in aprotic solvents like acetonitrile. By shifting the synthetic logic from metal-catalyzed cycloaddition to a base-mediated cyclization, the process not only enhances operational safety by avoiding explosive azides but also simplifies the downstream purification workflow. This methodology provides a robust platform for the commercial scale-up of complex pharmaceutical intermediates, ensuring that high-purity products can be delivered with greater consistency and lower ecological footprint.
Mechanistic Insights into Base-Promoted Cyclization
The mechanistic pathway proposed in the patent suggests a fascinating divergence from standard cycloaddition logic. The reaction is hypothesized to initiate with a base-promoted intermolecular nucleophilic addition-elimination sequence. Specifically, the diazo compound, acting as a nucleophile under the influence of the carbonate base, attacks the electrophilic carbon of the trifluoroethylimidoyl chloride. This initial interaction facilitates the formation of a critical carbon-carbon bond while eliminating the chloride leaving group. Following this addition step, the intermediate undergoes an intramolecular 5-endo-dig cyclization. This ring-closing event effectively constructs the five-membered 1,2,3-triazole core, embedding the trifluoromethyl group at the 5-position with high regioselectivity. The absence of transition metals means the electronic properties of the substrates drive the reactivity, allowing for a broad tolerance of functional groups on both the imidoyl chloride and the diazo components.
From an impurity control perspective, this metal-free mechanism offers distinct advantages for high-purity pharmaceutical intermediate production. Traditional copper-catalyzed routes often generate difficult-to-remove metal-organic complexes or homocoupling byproducts derived from the alkyne starting materials. In this novel base-promoted system, the primary byproducts are inorganic salts (such as cesium chloride) and nitrogen gas, which are easily separated via filtration or evaporation. The use of cesium carbonate, a mild yet effective base, minimizes side reactions such as hydrolysis of the sensitive imidoyl chloride or decomposition of the diazo species, provided the reaction temperature is maintained within the optimal 50-70°C window. This clean reaction profile translates directly to higher crude purity, reducing the burden on chromatographic purification and enhancing the overall yield of the desired triazole scaffold.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazoles Efficiently
The practical execution of this synthesis is designed for operational simplicity, making it highly attractive for process chemistry teams aiming to transfer laboratory protocols to pilot plant operations. The general procedure involves charging a reaction vessel with cesium carbonate, the specific trifluoroethylimidoyl chloride derivative, and the corresponding diazo compound in an anhydrous organic solvent. Acetonitrile is identified as the preferred solvent due to its ability to dissolve all reactants effectively while promoting high conversion rates. The mixture is then heated and stirred for a duration of 8 to 16 hours, allowing the cyclization to reach completion. Detailed standardized synthesis steps for specific derivatives are provided in the guide below.
- Mix cesium carbonate, trifluoroethylimidoyl chloride, and diazo compound in an organic solvent like acetonitrile.
- Heat the reaction mixture to 50-70°C and stir for 8-16 hours to ensure complete conversion.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers substantial strategic benefits beyond mere chemical novelty. The shift away from precious metal catalysts and hazardous azide reagents fundamentally alters the cost structure and risk profile of the supply chain. By utilizing commodity chemicals like cesium carbonate and commercially accessible imidoyl chlorides, manufacturers can mitigate the volatility associated with specialty catalyst pricing and the strict regulatory compliance costs linked to explosive precursors. This results in a more resilient supply chain capable of sustaining long-term production campaigns without the interruptions often caused by sourcing difficulties for exotic reagents.
- Cost Reduction in Manufacturing: The elimination of copper catalysts removes the necessity for expensive and time-consuming heavy metal scavenging steps, which are mandatory for API grade materials. Furthermore, the use of inexpensive inorganic bases like cesium carbonate instead of costly organometallic complexes significantly lowers the raw material cost per kilogram. The simplified workup procedure, involving basic filtration and standard chromatography rather than complex extractive removals of metal residues, reduces solvent consumption and labor hours, leading to substantial overall cost savings in the manufacturing process.
- Enhanced Supply Chain Reliability: The starting materials, specifically the trifluoroethylimidoyl chlorides and diazo compounds, are derived from widely available industrial feedstocks such as aromatic amines and acid chlorides. This ensures a stable and continuous supply of raw materials, reducing the risk of production delays caused by single-source dependencies on specialized catalysts. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further securing the continuity of supply for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: The reaction operates at moderate temperatures (50-70°C) and atmospheric pressure, which are ideal parameters for scaling up from gram-scale laboratory synthesis to multi-ton commercial production without requiring specialized high-pressure reactors. Additionally, the metal-free nature of the process drastically reduces the generation of heavy metal waste, simplifying wastewater treatment and aligning with increasingly stringent global environmental regulations. This eco-friendly profile facilitates faster regulatory approvals and supports sustainable manufacturing goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in patent CN113121462B, providing clarity on the feasibility and advantages of this route for potential partners and licensees.
Q: Does this synthesis require toxic copper catalysts?
A: No, the patented method (CN113121462B) is metal-free, utilizing cesium carbonate as a promoter instead of copper, which eliminates heavy metal contamination risks.
Q: What are the safety advantages over traditional azide methods?
A: Traditional methods often rely on hazardous organic azides. This novel approach uses stable diazo compounds and imidoyl chlorides, significantly reducing explosion risks during manufacturing.
Q: Is this process suitable for large-scale production?
A: Yes, the patent explicitly states the method can be easily extended to the gram level and beyond, offering mild reaction conditions (50-70°C) ideal for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis technology for the next generation of therapeutic agents. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 5-trifluoromethyl-1,2,3-triazole intermediate adheres to the highest quality standards required by global regulatory bodies.
We invite you to collaborate with us to leverage this efficient synthetic route for your specific drug development programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your project's volume requirements. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets, ensuring a secure and cost-effective supply chain for your critical intermediates.
