Scalable Metal-Free Synthesis of 3-Hydroxymethyl-4-Phenyl-3,4-Dihydroquinolinone Intermediates
Introduction to Advanced Quinolinone Synthesis
The development of efficient synthetic routes for functionalized heterocycles remains a cornerstone of modern medicinal chemistry, particularly for scaffolds exhibiting potent biological activity. Patent CN112125843B introduces a groundbreaking methodology for the preparation of 3-hydroxymethyl-4-phenyl-3,4-dihydroquinolinone compounds, a structural motif prevalent in numerous bioactive natural products and synthetic pharmaceuticals. This innovation addresses the critical industry demand for greener, more cost-effective manufacturing processes by eliminating the reliance on expensive transition metal catalysts and harsh acidic additives. The hydroxymethyl functional group incorporated into the quinolinone core is strategically significant, as it is known to enhance the metabolic stability and bioavailability of drug candidates, thereby accelerating the lead optimization phase in drug discovery pipelines. By leveraging a tandem epoxidation-intramolecular Friedel-Crafts alkylation sequence, this technology offers a streamlined pathway that aligns perfectly with the principles of sustainable chemistry while maintaining high operational simplicity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of complex dihydroquinolinone skeletons has often relied upon multi-step sequences involving stoichiometric amounts of hazardous reagents or precious metal catalysts such as palladium, rhodium, or copper. These conventional approaches frequently necessitate rigorous exclusion of moisture and oxygen, requiring specialized equipment and inert atmosphere conditions that drastically inflate capital expenditure and operational complexity. Furthermore, the use of strong Lewis or Brønsted acids to promote cyclization can lead to poor functional group tolerance, resulting in the degradation of sensitive moieties and the formation of difficult-to-remove impurities that compromise the purity profile of the final active pharmaceutical ingredient. The downstream processing associated with removing trace metal residues to meet stringent regulatory limits adds significant time and cost to the manufacturing timeline, creating a bottleneck for rapid scale-up and commercial deployment of new therapeutic agents.
The Novel Approach
In stark contrast to these legacy methods, the disclosed technology utilizes potassium monopersulfate (Oxone) as a dual-function reagent in an acetonitrile medium to drive the transformation under remarkably mild conditions. This approach bypasses the need for external acid additives or transition metals, as the byproduct potassium bisulfate generated in situ serves as the necessary proton source to activate the epoxy intermediate for cyclization. The reaction demonstrates exceptional substrate scope, accommodating various substituents including alkyl, alkoxy, and halogen groups on the aromatic ring without compromising efficiency. As illustrated in the general reaction scheme below, the process converts readily available N-methyl-N-aryl-2-phenylacrylamides directly into the target heterocycles with high atom economy.

This simplification of the reaction manifold not only reduces the environmental footprint by minimizing waste generation but also significantly lowers the barrier to entry for large-scale production, making it an attractive option for industrial applications where cost and safety are paramount concerns.
Mechanistic Insights into Oxone-Mediated Cascade Cyclization
A deep understanding of the reaction mechanism is essential for R&D teams aiming to optimize process parameters and ensure robust quality control during scale-up. The transformation proceeds through a sophisticated cascade sequence initiated by the oxidation of the alkene moiety in the starting acrylamide by the peroxymonosulfate anion. This initial step generates an epoxide intermediate along with potassium bisulfate, which plays a pivotal role in the subsequent steps by providing the acidic environment required for ring opening. The protonation of the epoxide oxygen increases its electrophilicity, facilitating an intramolecular nucleophilic attack by the electron-rich aromatic ring in a Friedel-Crafts type manner. This cyclization event constructs the core six-membered nitrogen-containing ring system while establishing the stereochemical relationships at the newly formed chiral centers.
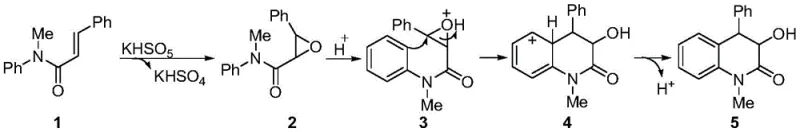
Following the cyclization, the resulting cationic intermediate undergoes a dehydroaromatization step to restore the aromaticity of the benzene ring, ultimately yielding the stable 3-hydroxymethyl-4-phenyl-3,4-dihydroquinolinone product. This mechanistic pathway highlights the elegance of using a single reagent to perform multiple distinct chemical transformations sequentially, thereby minimizing the number of unit operations required. For process chemists, this implies a reduced risk of intermediate isolation losses and a simpler impurity profile, as fewer side reactions associated with multiple reagent additions are possible. The absence of transition metals also eliminates the risk of metal-catalyzed decomposition pathways, ensuring greater batch-to-batch consistency and reliability in the manufacturing process.
How to Synthesize 3-Hydroxymethyl-4-Phenyl-3,4-Dihydroquinolinone Efficiently
Implementing this synthesis protocol requires careful attention to solvent selection and thermal management to achieve the reported high yields. The procedure involves dissolving the acrylamide substrate in acetonitrile, which has been identified as the critical reaction medium, followed by the addition of 1.5 equivalents of potassium monopersulfate. The mixture is then heated to a precise temperature of 90°C and maintained for 24 hours to ensure complete conversion while avoiding the formation of byproducts associated with higher temperatures. Detailed standardized synthesis steps including specific workup procedures and purification protocols are provided in the guide below to assist technical teams in replicating these results effectively.
- Combine N-methyl-N-aryl-2-phenylacrylamide and 1.5 equivalents of potassium monopersulfate in acetonitrile solvent.
- Heat the reaction mixture to 90°C and stir for 24 hours to facilitate the epoxidation-cyclization cascade.
- Purify the crude product via silica gel column chromatography using petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing and supply chain perspective, this metal-free methodology offers substantial advantages that directly impact the bottom line and operational resilience of pharmaceutical manufacturing. The elimination of precious metal catalysts removes a major cost driver and supply chain vulnerability, as the prices of metals like palladium and rhodium are subject to extreme volatility and geopolitical constraints. Furthermore, the use of commodity chemicals like potassium monopersulfate and acetonitrile ensures a stable and abundant supply of raw materials, mitigating the risk of production delays caused by reagent shortages. The simplified workflow also translates to reduced labor costs and shorter cycle times, as the need for complex metal scavenging steps and extensive purification to remove trace contaminants is obviated.
- Cost Reduction in Manufacturing: The replacement of expensive transition metal catalysts with inexpensive Oxone results in a drastic reduction in raw material costs per kilogram of product. Additionally, the removal of metal scavenging resins and the associated filtration steps simplifies the downstream processing train, leading to significant savings in consumables and waste disposal fees. The high selectivity of the reaction minimizes the formation of difficult-to-separate impurities, which further enhances the overall process efficiency and yield, maximizing the return on investment for every batch produced.
- Enhanced Supply Chain Reliability: By relying on globally available bulk chemicals rather than specialized catalytic systems, manufacturers can secure long-term supply contracts with multiple vendors, ensuring continuity of supply even during market disruptions. The robustness of the reaction conditions, which tolerate a wide range of functional groups, allows for the use of diverse starting materials that can be sourced flexibly based on availability and price. This flexibility empowers procurement teams to negotiate better terms and avoid single-source dependencies that often plague complex synthetic routes reliant on proprietary ligands or catalysts.
- Scalability and Environmental Compliance: The absence of heavy metals simplifies the regulatory approval process for new drug applications, as the burden of proving low residual metal levels is removed. This green chemistry approach aligns with increasingly stringent environmental regulations regarding heavy metal discharge, reducing the compliance burden and potential fines associated with wastewater treatment. The straightforward nature of the reaction makes it highly amenable to scale-up from laboratory to pilot and commercial scales without the need for specialized reactor configurations, facilitating rapid technology transfer and faster time-to-market for new products.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic route, derived directly from the experimental data and mechanistic studies presented in the patent literature. Understanding these nuances is crucial for process engineers and quality assurance personnel tasked with validating the method for GMP production. The answers provided reflect the specific findings regarding solvent effects, stoichiometry, and thermal parameters that define the operational window for this transformation.
Q: Why is acetonitrile critical for this specific cyclization reaction?
A: Experimental data indicates that acetonitrile is the only effective solvent; alternatives like DCM, toluene, and DMF result in no reaction, likely due to specific solvation effects required for the Oxone activation.
Q: Does this method require expensive transition metal catalysts?
A: No, the process is entirely metal-free, utilizing inexpensive potassium monopersulfate which acts as both the oxidant and the acid promoter, significantly reducing raw material costs.
Q: What is the optimal temperature range for maximizing yield?
A: The optimal temperature is 90°C; lowering to 80°C reduces yield to approximately 56% due to incomplete conversion, while 100°C decreases yield to 69% due to side reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Hydroxymethyl-4-Phenyl-3,4-Dihydroquinolinone Supplier
As a leading CDMO partner, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure to translate this innovative patent technology into commercial reality for your organization. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency and quality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-hydroxymethyl-4-phenyl-3,4-dihydroquinolinone delivered meets the highest industry standards for pharmaceutical intermediates.
We invite you to engage with our technical procurement team to discuss how this metal-free synthesis can be integrated into your supply chain to drive efficiency and value. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the economic benefits of switching to this greener protocol. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your specific project requirements, ensuring a seamless partnership from process development to full-scale manufacturing.
