Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)-one Derivatives for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing privileged heterocyclic scaffolds, particularly those found in bioactive natural products and drug candidates. Patent CN113045489A discloses a groundbreaking preparation method for 3-arylquinoline-2(1H)-ketone derivatives, a structural motif prevalent in compounds exhibiting antibiotic, antiplatelet, antitumor, and receptor antagonist activities. This novel approach leverages a palladium-catalyzed aminocarbonylation reaction that uniquely utilizes benzisoxazole as a dual nitrogen and formyl source, effectively bypassing the limitations of traditional synthetic routes. By integrating this advanced catalytic system, manufacturers can access a versatile library of quinolin-2(1H)-one derivatives with high efficiency and operational simplicity. The significance of this technology extends beyond academic interest, offering a tangible pathway for the commercial scale-up of complex pharmaceutical intermediates with improved safety and cost profiles.
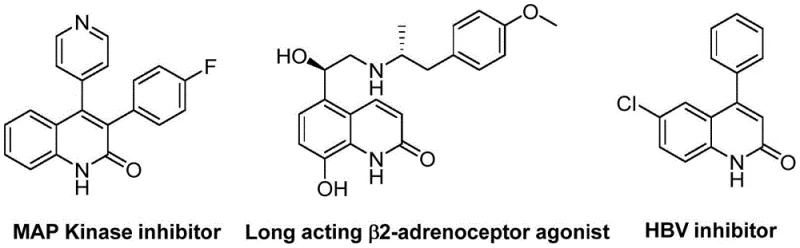
Historically, the construction of the quinolin-2(1H)-one core has relied heavily on classical condensation reactions such as the Vilsmeier-Haack, Knorr, and Friedlander reactions. While these methods are well-established, they often suffer from significant drawbacks including harsh reaction conditions, poor functional group tolerance, and the generation of substantial chemical waste. Furthermore, transition metal-catalyzed modifications to these processes have frequently required the use of hazardous carbon monoxide gas under high pressure, posing severe safety challenges for industrial implementation. These conventional limitations restrict the diversity of substituents that can be introduced, often necessitating lengthy protection-deprotection sequences that drive up costs and extend lead times. For procurement managers and supply chain heads, these inefficiencies translate into higher raw material costs and unpredictable delivery schedules for critical high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The reliance on traditional cyclization strategies creates bottlenecks in the development of new drug candidates, particularly when late-stage functionalization is required. The use of gaseous CO in carbonylation reactions demands specialized high-pressure equipment and rigorous safety protocols, which are not universally available in standard pilot plants. Additionally, the substrate scope in older methods is often narrow, failing to accommodate sensitive functional groups like nitriles or trifluoromethyl moieties without degradation. This lack of versatility forces R&D teams to explore alternative, often more expensive, synthetic pathways, delaying project timelines. The environmental footprint of these legacy processes is also a growing concern, as they frequently generate stoichiometric amounts of acidic or toxic byproducts that require complex waste treatment procedures, further impacting the overall cost of goods sold.
The Novel Approach
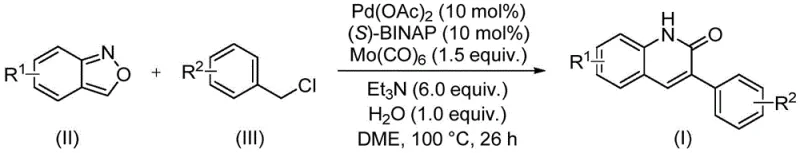
In stark contrast, the method described in CN113045489A introduces a paradigm shift by employing benzisoxazole as a safe, solid surrogate for both nitrogen and carbon monoxide equivalents. This innovative strategy operates under relatively mild conditions (100°C) using a palladium catalyst system composed of Pd(OAc)2 and (S)-BINAP, with molybdenum hexacarbonyl serving as the carbonyl source. The reaction exhibits remarkable generality, tolerating a wide array of electron-withdrawing and electron-donating groups on both the benzisoxazole and benzyl chloride coupling partners. As illustrated in the reaction scheme below, this single-step transformation directly constructs the quinolinone core with high atom economy. This streamlined process not only simplifies the operational workflow but also significantly enhances the safety profile by eliminating the need for handling toxic CO gas, thereby facilitating easier cost reduction in API manufacturing.
Mechanistic Insights into Palladium-Catalyzed Aminocarbonylation
The success of this transformation hinges on a sophisticated catalytic cycle initiated by the oxidative addition of the benzyl chloride substrate to the active Pd(0) species generated in situ from Pd(OAc)2 and the chiral ligand (S)-BINAP. Following oxidative addition, the coordination and subsequent insertion of carbon monoxide—released from the decomposition of Mo(CO)6 under the reaction conditions—into the palladium-carbon bond forms an acyl-palladium intermediate. Concurrently, the benzisoxazole undergoes ring-opening, likely facilitated by the basic conditions provided by triethylamine and water, releasing the necessary nitrogen nucleophile. This nucleophile then attacks the acyl-palladium species, followed by reductive elimination to forge the C-N bond and close the quinolinone ring, regenerating the Pd(0) catalyst. The presence of water is critical, acting as a proton source to facilitate the ring-opening of the benzisoxazole and stabilize intermediates, ensuring high turnover numbers and minimal catalyst deactivation.
From an impurity control perspective, the use of the bulky, chiral (S)-BINAP ligand plays a pivotal role in suppressing side reactions such as homocoupling of the benzyl chloride or premature hydrolysis of the acyl intermediate. The specific electronic and steric properties of this ligand optimize the rate of reductive elimination relative to competing pathways, resulting in the high yields (ranging from 68% to 97%) observed across diverse substrates. Furthermore, the mild basicity of triethylamine prevents the degradation of sensitive functional groups like esters or nitriles, which might otherwise decompose under the strongly acidic or basic conditions of traditional methods. This precise control over the reaction environment ensures that the final high-purity heterocyclic compounds meet stringent quality specifications with minimal downstream purification requirements, a critical factor for regulatory compliance in pharmaceutical production.
How to Synthesize 3-Arylquinolin-2(1H)-one Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and reaction parameters to maximize yield and purity. The process involves charging a sealed vessel with the catalyst system, substrates, and solvent, followed by heating to the optimal temperature for a defined period. Detailed standard operating procedures regarding reagent grades, solvent drying, and workup techniques are essential for reproducibility. For a comprehensive guide on the specific molar ratios, solvent volumes, and purification steps validated in the patent examples, please refer to the standardized protocol below.
- Combine palladium acetate, (S)-BINAP, molybdenum carbonyl, triethylamine, water, benzisoxazole, and benzyl chloride in a sealed tube with DME solvent.
- Heat the reaction mixture to 100°C and stir continuously for approximately 26 hours to ensure complete conversion.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final 3-arylquinolin-2(1H)-one derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain directors, the adoption of this benzisoxazole-based carbonylation route offers distinct strategic advantages over legacy technologies. The primary benefit lies in the drastic simplification of the supply chain for raw materials; benzisoxazoles and substituted benzyl chlorides are commodity chemicals available from multiple global vendors, reducing dependency on single-source suppliers. Moreover, the replacement of gaseous carbon monoxide with solid molybdenum hexacarbonyl eliminates the need for specialized high-pressure reactors and the associated regulatory burdens, allowing production to be shifted to standard multipurpose facilities. This flexibility significantly reduces capital expenditure and operational overhead, directly contributing to cost reduction in pharmaceutical intermediate manufacturing without compromising on quality or safety standards.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive, commercially available starting materials and the elimination of costly protecting group manipulations. By utilizing benzisoxazole as a dual-purpose reagent, the step count is minimized, which inherently reduces labor, energy, and solvent consumption per kilogram of product. The high catalytic efficiency ensures that precious palladium usage is optimized, and the straightforward workup involving filtration and chromatography minimizes waste disposal costs. These factors collectively lower the total cost of ownership for the manufacturing process, providing a competitive edge in pricing for bulk orders.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch quality, which is paramount for maintaining uninterrupted supply lines to downstream API manufacturers. The broad substrate scope means that a single platform technology can be adapted to produce a wide variety of derivatives, allowing for agile responses to changing market demands or pipeline shifts. By avoiding hazardous gases and extreme conditions, the risk of unplanned shutdowns due to safety incidents is markedly reduced, ensuring reducing lead time for high-purity intermediates and guaranteeing on-time delivery for critical projects.
- Scalability and Environmental Compliance: Scaling this reaction from gram to tonnage levels is facilitated by the homogeneous nature of the catalytic system and the absence of gas-liquid mass transfer limitations typical of CO carbonylations. The process generates fewer hazardous byproducts compared to traditional methods, aligning with modern green chemistry principles and simplifying environmental permitting. The ability to run the reaction in common solvents like DME further enhances its adaptability to existing infrastructure, making it an ideal candidate for rapid technology transfer and commercial scale-up in regulated environments.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing clarity on reaction scope, safety, and scalability. Understanding these details is crucial for R&D teams evaluating this route for their specific pipeline candidates and for procurement teams assessing supplier capabilities.
Q: What are the key advantages of using benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual role as both the nitrogen source and the formyl (C1) source, eliminating the need for separate toxic carbon monoxide gas or complex formylating agents. This simplifies the reaction setup, improves safety profiles, and enhances atom economy compared to traditional Vilsmeier-Haack or Knorr reactions.
Q: How does this method improve functional group tolerance?
A: The palladium-catalyzed system utilizing (S)-BINAP and mild conditions (100°C) demonstrates exceptional tolerance for diverse substituents including halogens, cyano groups, alkoxy groups, and trifluoromethyl groups on both the benzisoxazole and benzyl chloride substrates, allowing for the synthesis of a wide library of derivatives without protecting group strategies.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes commercially available and inexpensive starting materials like benzyl chlorides and benzisoxazoles. The use of solid Mo(CO)6 instead of gaseous CO significantly reduces safety risks associated with high-pressure carbonylation, making the scale-up to multi-kilogram or tonnage production feasible and cost-effective.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists has extensively evaluated the palladium-catalyzed aminocarbonylation route described in CN113045489A and confirmed its potential for delivering high-quality intermediates. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 3-arylquinolin-2(1H)-one derivative meets the highest industry standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in complex heterocycle synthesis can drive value and efficiency in your supply chain.
