Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)-one Derivatives for Commercial Scale-up
Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)-one Derivatives for Commercial Scale-up
The pharmaceutical industry continuously seeks robust synthetic routes for heterocyclic scaffolds that possess significant biological activity. A recent breakthrough detailed in patent CN113045489B introduces a highly efficient preparation method for 3-arylquinolin-2(1H) ketone derivatives, utilizing a novel palladium-catalyzed aminocarbonylation strategy. This technology addresses critical bottlenecks in traditional heterocycle synthesis by employing benzisoxazole as a dual-purpose reagent, acting simultaneously as a nitrogen source and a formyl source. The significance of this chemical class cannot be overstated, as quinolin-2(1H)one derivatives are pervasive in medicinal chemistry, serving as core structures for antibiotics, antiplatelet agents, and antitumor drugs. As illustrated in the structural diversity of bioactive molecules below, the ability to rapidly access these scaffolds with high functional group tolerance is a major asset for drug discovery teams.
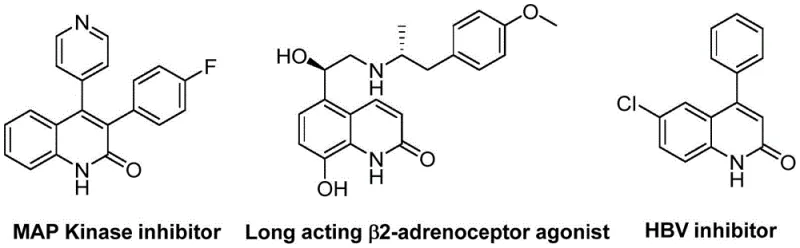
For R&D directors evaluating new process technologies, the versatility of this method is paramount. The patent discloses a catalytic system comprising palladium acetate and (S)-1,1'-binaphthyl-2,2'-bisdiphenylphosphine, which facilitates the coupling of readily available benzisoxazoles with benzyl chloride compounds. This approach circumvents the harsh conditions often associated with classical cyclization methods like the Vilsmeier-Haack or Friedlander reactions. By operating at a moderate temperature of 100°C in ethylene glycol dimethyl ether (DME), the process ensures compatibility with sensitive functional groups such as cyano, trifluoromethyl, and various halogens. This broad substrate scope allows for the rapid generation of diverse libraries, accelerating the structure-activity relationship (SAR) studies essential for modern drug development programs targeting complex diseases.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolin-2(1H)one derivatives has relied on multi-step sequences involving strong acids, high temperatures, or toxic reagents. Traditional pathways such as the Knorr quinoline synthesis or modifications thereof often suffer from poor atom economy and limited functional group tolerance. For instance, the use of gaseous carbon monoxide in carbonylation reactions poses significant safety hazards and requires specialized high-pressure equipment, which complicates scale-up and increases capital expenditure. Furthermore, conventional methods frequently struggle with regioselectivity issues, leading to difficult-to-separate impurities that compromise the purity profile of the final active pharmaceutical ingredient (API). These inefficiencies result in prolonged development timelines and inflated production costs, creating a pressing need for more streamlined and safer synthetic alternatives in the fine chemical sector.
The Novel Approach
The methodology described in CN113045489B represents a paradigm shift by integrating the nitrogen and carbonyl components into a single, stable heterocyclic precursor. By leveraging benzisoxazole, the reaction eliminates the need for handling hazardous gases or unstable intermediates. The catalytic cycle, driven by a palladium complex and molybdenum hexacarbonyl as a solid CO source, proceeds under relatively mild conditions. As shown in the general reaction scheme below, the transformation efficiently constructs the quinolinone core with excellent yields, often surpassing 90% for optimized substrates. This one-pot strategy not only simplifies the operational workflow but also drastically reduces the environmental footprint by minimizing waste generation and energy consumption, aligning perfectly with the principles of green chemistry.
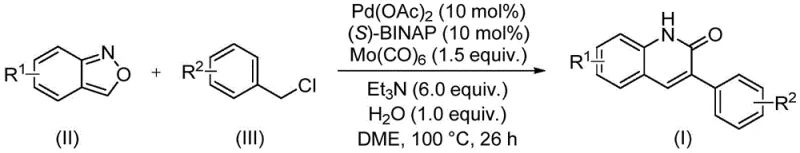
Mechanistic Insights into Palladium-Catalyzed Aminocarbonylation
The success of this transformation hinges on the precise interplay between the palladium catalyst and the unique reactivity of the benzisoxazole ring. Mechanistically, the reaction initiates with the oxidative addition of the benzyl chloride to the palladium(0) species generated in situ from palladium acetate and the chiral ligand (S)-BINAP. Subsequently, the benzisoxazole undergoes ring-opening, facilitated by the transition metal center, to release the necessary nitrogen and formyl equivalents. The presence of molybdenum hexacarbonyl serves as a safe, solid surrogate for carbon monoxide, slowly releasing CO into the reaction medium to participate in the carbonylation step. This controlled release mechanism prevents the accumulation of toxic gas and ensures a steady concentration of the carbonylating agent, which is critical for driving the reaction to completion without side reactions.
From an impurity control perspective, the use of (S)-BINAP as a ligand enhances the selectivity of the catalytic cycle, minimizing the formation of homocoupling byproducts or dehalogenated species. The reaction conditions, specifically the use of triethylamine as a base and water as an additive, are optimized to promote the reductive elimination step that forms the final C-C and C-N bonds of the quinolinone ring. This mechanistic understanding allows process chemists to fine-tune parameters such as temperature and stoichiometry to maximize yield. The robustness of this catalytic system is evidenced by its tolerance to electron-withdrawing and electron-donating groups on both the benzisoxazole and benzyl chloride partners, ensuring consistent quality across a wide range of derivatives.
How to Synthesize 3-Arylquinolin-2(1H)-one Efficiently
The practical implementation of this synthesis is designed for ease of execution in both laboratory and pilot plant settings. The protocol involves charging a sealed vessel with the catalyst system, substrates, and solvent, followed by heating to a specific temperature window. The simplicity of the workup procedure, which typically involves filtration and standard column chromatography, makes it highly attractive for rapid prototyping. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during operation.
- Charge a sealed tube with palladium acetate, (S)-BINAP, molybdenum hexacarbonyl, triethylamine, water, benzisoxazole, and benzyl chloride compound.
- Add ethylene glycol dimethyl ether (DME) as solvent and stir the mixture thoroughly to ensure homogeneity.
- Heat the reaction mixture to 100°C for 26 hours, then filter, mix with silica gel, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of adopting this technology are substantial. The primary cost drivers in pharmaceutical manufacturing—raw material availability, safety compliance, and processing time—are all favorably impacted by this novel route. By replacing hazardous gaseous reagents with stable solids and utilizing commodity chemicals like benzyl chlorides, the process mitigates supply chain risks associated with specialized reagent sourcing. Furthermore, the high conversion rates and clean reaction profiles reduce the burden on downstream purification units, leading to significant operational efficiencies.
- Cost Reduction in Manufacturing: The elimination of high-pressure carbon monoxide infrastructure removes a major capital expense and ongoing maintenance cost. Additionally, the use of benzisoxazole as a dual-source reagent reduces the total number of raw materials required per batch, simplifying inventory management. The high yields reported in the patent data imply less raw material waste per kilogram of product, directly lowering the cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: The starting materials, including benzisoxazoles and benzyl chlorides, are widely available from global chemical suppliers, ensuring a stable and continuous supply stream. Unlike processes dependent on niche catalysts or cryogenic reagents, this method relies on robust, shelf-stable components that can be stockpiled effectively. This reliability is crucial for maintaining production schedules and meeting tight delivery deadlines for downstream API manufacturers.
- Scalability and Environmental Compliance: The reaction operates at atmospheric pressure (in a sealed tube context) and moderate temperatures, making it inherently safer and easier to scale from grams to tons. The reduced generation of hazardous waste and the avoidance of toxic gas emissions simplify regulatory compliance and waste disposal procedures. This alignment with environmental, social, and governance (ESG) goals adds long-term value to the manufacturing process by future-proofing it against tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis. These insights are derived directly from the experimental data and specifications outlined in the patent documentation, providing clarity on process capabilities and limitations.
Q: What is the role of benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual function as both the nitrogen source and the formyl source, eliminating the need for separate amine and carbonyl reagents.
Q: Is gaseous carbon monoxide required for this reaction?
A: No, the process utilizes molybdenum hexacarbonyl (Mo(CO)6) as a solid CO surrogate, significantly enhancing operational safety compared to high-pressure CO gas.
Q: What is the typical yield range for these derivatives?
A: The patented method demonstrates high reaction efficiency, with isolated yields frequently exceeding 90% for various substituted substrates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes for high-value pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Our capability to implement advanced catalytic technologies like the one described in CN113045489B positions us as a preferred partner for complex molecule synthesis.
We invite potential partners to engage with our technical procurement team to discuss how this innovative chemistry can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your project volume. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and profitability in your drug development pipeline.
