Advanced Synthesis of N-N Axis Chiral Bisindoles for High-Value Anticancer Intermediates
The pharmaceutical industry is constantly seeking novel scaffolds that can unlock new biological activities, particularly in the challenging field of oncology. Patent CN116082217B introduces a groundbreaking methodology for the synthesis of N-N axis chiral bisindole compounds, a class of molecules that has remained largely unexplored compared to their N-aryl or C-aryl counterparts. This innovation addresses a critical gap in medicinal chemistry by providing a robust, enantioselective route to access these complex structures, which have demonstrated potent cytotoxic activity against human prostate cancer PC-3 cells. The core of this technology lies in the utilization of asymmetric organocatalysis, specifically employing chiral phosphoric acids to induce axial chirality during the bond-forming event. For research directors and procurement specialists alike, this represents a significant opportunity to diversify pipeline candidates with high-value intermediates that possess both structural novelty and proven biological relevance.
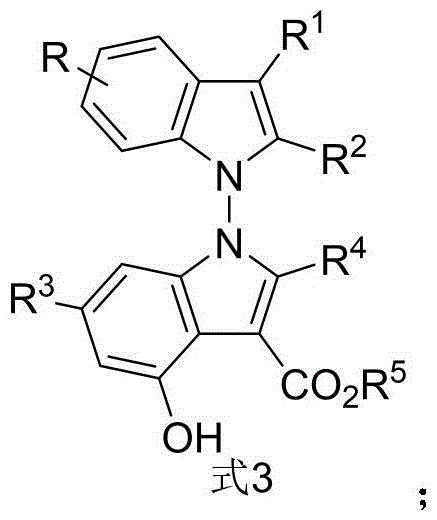
Traditionally, the synthesis of axially chiral indole derivatives has focused heavily on N-aryl, 3-aryl, or 2-aryl configurations, leaving the N-N axis connectivity underdeveloped due to the synthetic challenges associated with controlling rotation around the nitrogen-nitrogen bond. Conventional methods often suffer from harsh reaction conditions, poor stereocontrol, or limited substrate scope, which hinders their application in the rapid generation of diverse libraries for drug screening. Furthermore, many existing protocols rely on transition metal catalysts that introduce concerns regarding residual metal contamination, a critical quality attribute for active pharmaceutical ingredients (APIs). The lack of efficient methods to construct the N-N axis has previously restricted the exploration of this chemical space, limiting the ability of medicinal chemists to optimize lead compounds for improved potency and selectivity in cancer therapy.
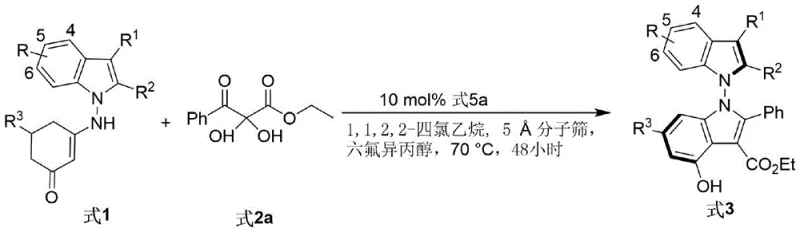
The novel approach disclosed in the patent overcomes these limitations through a highly efficient, one-pot condensation reaction between indole-derived enamines and 2,3-diketone ester derivatives. By leveraging the dual activation capability of chiral phosphoric acids, the reaction proceeds under remarkably mild thermal conditions, typically between 60°C and 90°C, eliminating the need for cryogenic temperatures or hazardous reagents. This method not only achieves excellent yields but also delivers exceptional enantioselectivity, with some examples reaching up to 96% ee, ensuring the production of high-purity chiral intermediates essential for regulatory compliance. The versatility of this protocol is further evidenced by its tolerance to a wide range of functional groups, allowing for the synthesis of structurally diverse bisindole derivatives from readily available starting materials.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Cyclization
The success of this synthesis hinges on the precise molecular recognition and activation provided by the chiral phosphoric acid catalyst, specifically derivatives based on the binaphthyl skeleton such as Formula 5a. Mechanistically, the catalyst acts as a bifunctional promoter, simultaneously activating the electrophilic 2,3-diketone ester through hydrogen bonding while organizing the nucleophilic indole enamine within a well-defined chiral pocket. This supramolecular assembly lowers the activation energy for the C-C bond formation and rigidifies the transition state, effectively locking the conformation to favor the formation of one atropisomer over the other. The presence of hexafluoroisopropanol (HFIP) as an additive plays a crucial role in enhancing the acidity of the catalyst and stabilizing the charged intermediates through strong hydrogen bond donor interactions, which is vital for achieving high turnover and stereoselectivity.
Impurity control is inherently built into this catalytic system due to the high specificity of the organocatalyst. Unlike radical-based or metal-catalyzed processes that often generate complex mixtures of regioisomers and side products, this Brønsted acid-catalyzed pathway follows a concerted mechanism that minimizes off-cycle reactions. The use of molecular sieves as a dehydrating agent drives the equilibrium forward by removing water generated during the condensation, preventing hydrolysis of the sensitive ester or enamine functionalities. This results in a cleaner crude reaction profile, significantly reducing the burden on downstream purification processes and ensuring that the final product meets stringent purity specifications required for preclinical and clinical evaluation.
How to Synthesize N-N Axis Chiral Bisindoles Efficiently
To implement this synthesis in a laboratory or pilot plant setting, operators should follow a standardized protocol that ensures reproducibility and safety. The process begins with the careful selection of high-quality starting materials, specifically the indole enamine and the diketone ester, which should be dried thoroughly to prevent catalyst deactivation. The reaction is typically conducted in 1,1,2,2-tetrachloroethane, a solvent chosen for its ability to dissolve both polar and non-polar components while maintaining stability at elevated temperatures. The addition of 5Å molecular sieves is critical for water scavenging, and the mixture must be stirred vigorously to ensure effective mass transfer between the solid desiccant and the liquid phase.
- Combine indole-derived enamine and 2,3-diketone ester derivatives in an organic solvent such as 1,1,2,2-tetrachloroethane.
- Add chiral phosphoric acid catalyst (e.g., Formula 5a), a dehydrating agent like 5Å molecular sieves, and hexafluoroisopropanol as an additive.
- Stir the reaction mixture at 70°C for 48 hours, then filter, concentrate, and purify via silica gel column chromatography to obtain the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers substantial advantages for supply chain stability and cost management in the production of complex pharmaceutical intermediates. The reliance on organocatalysis rather than precious metal catalysis eliminates the need for expensive palladium, rhodium, or iridium complexes, which are subject to volatile market pricing and supply chain disruptions. Furthermore, the absence of heavy metals simplifies the purification workflow, removing the need for specialized scavenging resins or extensive washing steps to meet residual metal limits, thereby reducing both material costs and processing time. The use of commodity chemicals as starting materials ensures a reliable supply base, mitigating the risk of raw material shortages that can plague more exotic synthetic routes.
- Cost Reduction in Manufacturing: The process operates at moderate temperatures (70°C) using standard glass-lined reactors, avoiding the capital expenditure associated with cryogenic cooling systems or high-pressure equipment. The high atom economy of the condensation reaction means that a significant proportion of the reactant mass is incorporated into the final product, minimizing waste disposal costs. Additionally, the simplified workup procedure involving filtration and concentration allows for faster batch turnover, increasing overall plant throughput and reducing labor costs per kilogram of produced intermediate.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions allows for flexibility in sourcing solvents and reagents, as the protocol tolerates minor variations in purity without compromising yield or selectivity. This resilience is crucial for maintaining continuous production schedules in the face of global logistics challenges. The scalability of the method from milligram to multi-kilogram scales has been demonstrated through the broad substrate scope, indicating that the kinetics and thermodynamics remain favorable even as reaction volumes increase, ensuring consistent quality across different batch sizes.
- Scalability and Environmental Compliance: The elimination of toxic heavy metals aligns with increasingly strict environmental regulations and green chemistry principles, reducing the environmental footprint of the manufacturing process. The solvents used, such as tetrachloroethane, can be efficiently recovered and recycled through distillation, further lowering the environmental impact and operational expenses. The high selectivity of the reaction reduces the generation of hazardous byproducts, simplifying waste treatment protocols and ensuring compliance with local discharge standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this N-N axis chiral bisindole synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a factual basis for decision-making.
Q: What distinguishes N-N axis chiral bisindoles from traditional axial chiral indoles?
A: Unlike traditional N-aryl or 3-aryl indoles, N-N axis chiral bisindoles feature a unique nitrogen-nitrogen axial chirality, offering novel structural diversity for drug discovery, particularly in oncology applications targeting PC-3 prostate cancer cells.
Q: What are the optimal reaction conditions for this synthesis?
A: The optimal conditions involve using a binaphthyl-derived chiral phosphoric acid (Formula 5a) in 1,1,2,2-tetrachloroethane at 70°C for 48 hours, with 5Å molecular sieves and hexafluoroisopropanol to maximize enantioselectivity up to 96% ee.
Q: Does this method support large-scale industrial production?
A: Yes, the process utilizes conventional solvents, mild temperatures (60-90°C), and commercially available starting materials, making it highly suitable for scalable manufacturing without requiring complex cryogenic conditions or expensive transition metals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-N Axis Chiral Bisindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of accessing novel chiral scaffolds for the development of next-generation anticancer therapeutics. Our team of expert process chemists has extensively evaluated the methodology described in CN116082217B and is fully prepared to support your R&D initiatives with high-quality intermediates. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee enantiomeric excess levels that match or exceed patent benchmarks.
We invite you to collaborate with us to accelerate your drug discovery programs. By leveraging our expertise in asymmetric synthesis and process optimization, we can help you navigate the complexities of scaling this innovative chemistry. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain efficiency and reduce your overall cost of goods sold.
