Advanced Organocatalytic Synthesis of N-N Axis Chiral Bisindoles for Oncology Applications
Advanced Organocatalytic Synthesis of N-N Axis Chiral Bisindoles for Oncology Applications
The pharmaceutical industry is constantly seeking novel scaffolds that offer unique biological activities, particularly in the realm of oncology. Patent CN116082217B introduces a groundbreaking methodology for the synthesis of N-N axis chiral bisindole compounds, a class of molecules that has previously been underexplored despite their significant potential. These compounds, characterized by their distinct axial chirality around the nitrogen-nitrogen bond, have demonstrated potent cytotoxic activity against human prostate cancer PC-3 cells. For R&D directors and procurement specialists alike, this patent represents a pivotal shift towards more efficient, metal-free synthetic routes that can deliver high-value intermediates with exceptional stereochemical control. The disclosed method utilizes a sophisticated organocatalytic system that not only simplifies the manufacturing process but also aligns with the stringent purity requirements necessary for modern drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of axially chiral indole compounds has relied heavily on transition metal catalysis or harsh reaction conditions that often compromise atom economy and environmental safety. Conventional routes to N-aryl or C-aryl axially chiral indoles frequently require expensive palladium or rhodium catalysts, which introduce the risk of heavy metal contamination—a critical failure point for API intermediates intended for human consumption. Furthermore, existing methods for generating N-N axis chirality are scarce and often suffer from low enantioselectivity, necessitating costly and time-consuming resolution steps to isolate the desired enantiomer. The reliance on stoichiometric chiral auxiliaries or difficult-to-remove metal residues creates a significant bottleneck in the supply chain, inflating costs and extending lead times for preclinical candidates. These legacy processes are ill-suited for the rapid iteration required in modern medicinal chemistry, where speed and purity are paramount.
The Novel Approach
In stark contrast, the methodology described in CN116082217B leverages a highly efficient organocatalytic asymmetric coupling reaction that operates under mild, metal-free conditions. By employing chiral phosphoric acids derived from binaphthyl skeletons, the process achieves remarkable levels of stereocontrol without the baggage of transition metals. The reaction proceeds smoothly in common organic solvents like 1,1,2,2-tetrachloroethane at moderate temperatures, typically around 70°C, which significantly reduces energy consumption compared to high-temperature thermal cyclizations. This novel approach not only streamlines the synthetic workflow by eliminating metal scavenging steps but also broadens the scope of accessible chemical space, allowing for the incorporation of diverse functional groups that might be incompatible with traditional metal-catalyzed systems. The result is a robust, scalable platform technology that delivers complex chiral architectures with high fidelity.
Mechanistic Insights into Chiral Phosphoric Acid Catalysis
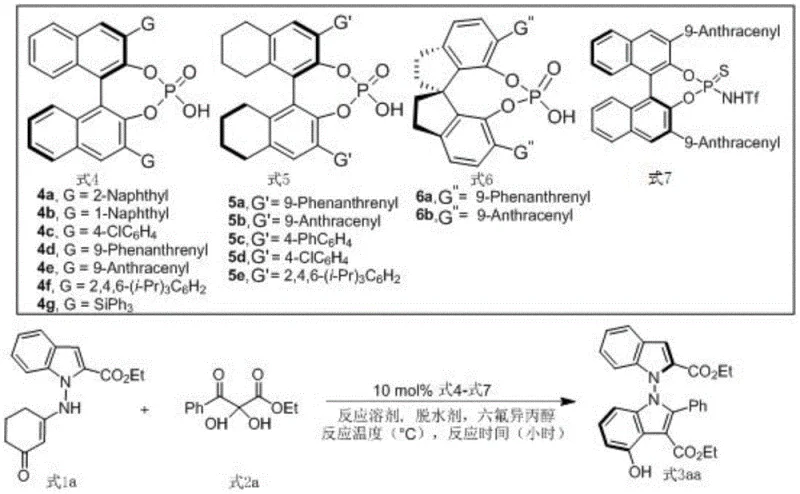
The core of this synthetic breakthrough lies in the precise activation of substrates through hydrogen bonding networks facilitated by the chiral phosphoric acid catalyst. Specifically, the binaphthyl skeleton derivative designated as Formula 5a acts as a dual-function catalyst, simultaneously activating the electrophilic 2,3-diketone ester and organizing the nucleophilic indole-derived enamine within a well-defined chiral pocket. This bifunctional activation ensures that the bond formation occurs with high regio- and enantioselectivity, effectively locking the N-N axis into the desired configuration. The presence of hexafluoroisopropanol (HFIP) as an additive further enhances this interaction by stabilizing the transition state through additional hydrogen bonding, thereby boosting both reaction rate and stereoselectivity. Understanding this mechanistic nuance is crucial for process chemists aiming to optimize the reaction for kilogram-scale production, as slight deviations in catalyst loading or solvent choice can impact the delicate balance of non-covalent interactions.
Furthermore, the impurity profile of the resulting bisindole compounds is exceptionally clean due to the high specificity of the organocatalyst. Unlike radical-based or metal-mediated processes that often generate a plethora of side products through uncontrolled pathways, this Brønsted acid-catalyzed reaction follows a concerted mechanism that minimizes byproduct formation. The use of 5A molecular sieves as a dehydrating agent effectively drives the equilibrium towards product formation by sequestering water generated during the condensation, preventing hydrolysis of the sensitive ester functionalities. This inherent selectivity translates directly to simplified downstream processing, as the crude reaction mixtures require less rigorous purification to meet pharmaceutical grade specifications. For quality control teams, this means fewer batches are rejected due to out-of-specification impurities, ensuring a more reliable supply of high-purity intermediates for subsequent drug synthesis steps.
How to Synthesize N-N Axis Chiral Bisindole Efficiently
The synthesis of these valuable chiral building blocks is designed to be operationally simple, making it accessible for both laboratory-scale discovery and pilot-plant manufacturing. The general procedure involves combining the indole-derived enamine and the 2,3-diketone ester in a suitable organic solvent, followed by the addition of the chiral catalyst and additives. The reaction is then heated to a moderate temperature and monitored until completion, after which standard workup procedures yield the target compound. This straightforward protocol eliminates the need for specialized equipment or inert atmosphere techniques often required for sensitive organometallic reactions. Detailed standardized synthesis steps for implementing this protocol in your facility are provided in the guide below.
- Prepare the reaction mixture by adding indole-derived enamine (Formula 1) and 2,3-diketone ester derivative (Formula 2) into 1,1,2,2-tetrachloroethane solvent.
- Add the chiral phosphoric acid catalyst (preferably Formula 5a), 5A molecular sieves as a dehydrating agent, and hexafluoroisopropanol as an additive.
- Stir the reaction at 70°C for 48 hours, then filter, concentrate, and purify via silica gel column chromatography to obtain the target bisindole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this organocatalytic route offers substantial strategic advantages over traditional metal-dependent syntheses. The elimination of precious metal catalysts such as palladium or rhodium immediately removes a major cost driver and supply risk associated with fluctuating commodity prices and geopolitical instability. Moreover, the absence of heavy metals simplifies the regulatory compliance landscape, reducing the burden of extensive testing for residual metals in the final API. This streamlined compliance pathway accelerates the time-to-market for new drug candidates, providing a competitive edge in the fast-paced pharmaceutical sector. The robustness of the reaction conditions also implies a lower risk of batch failures, ensuring a more consistent and predictable supply of critical intermediates.
- Cost Reduction in Manufacturing: The transition to an organocatalytic system fundamentally alters the cost structure of producing these complex chiral intermediates. By removing the necessity for expensive transition metal catalysts and the associated ligands, the direct material costs are significantly lowered. Additionally, the simplified purification process, which avoids the need for specialized metal scavengers or complex chromatographic separations to remove metal traces, reduces operational expenditures related to consumables and waste disposal. The high atom economy of the reaction further contributes to cost efficiency by maximizing the conversion of raw materials into the desired product, minimizing waste generation. These cumulative savings create a more economically viable pathway for the large-scale production of N-N axis chiral bisindoles.
- Enhanced Supply Chain Reliability: The reliance on readily available organic starting materials and stable organocatalysts enhances the resilience of the supply chain. Unlike transition metals, which are subject to supply constraints and mining limitations, the reagents used in this process are synthesized from abundant feedstocks and can be sourced from multiple vendors globally. This diversification of supply sources mitigates the risk of production stoppages due to raw material shortages. Furthermore, the mild reaction conditions reduce the wear and tear on manufacturing equipment, leading to lower maintenance costs and higher equipment availability. The overall stability and predictability of the process ensure that delivery schedules can be met consistently, supporting just-in-time manufacturing models.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram quantities is facilitated by the benign nature of the reagents and the simplicity of the operational parameters. The reaction does not require extreme pressures or temperatures, making it compatible with standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. From an environmental perspective, the metal-free nature of the synthesis aligns with green chemistry principles, reducing the toxicity of the waste stream and simplifying effluent treatment. The use of molecular sieves as a recyclable dehydrating agent further minimizes chemical waste. These factors collectively lower the environmental footprint of the manufacturing process, aiding in meeting increasingly stringent global environmental regulations and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these N-N axis chiral bisindole compounds. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a reliable basis for decision-making. Whether you are evaluating the feasibility of this route for a specific project or assessing the quality attributes of the final product, these insights offer clarity on the capabilities and limitations of the technology. We encourage technical teams to review these details closely to understand how this methodology can be integrated into their existing workflows.
Q: What is the primary advantage of using chiral phosphoric acid in this synthesis?
A: The use of chiral phosphoric acid, specifically binaphthyl skeleton derivatives like Formula 5a, enables high enantioselectivity (up to 96% ee) without the need for toxic transition metals, ensuring a cleaner product profile suitable for pharmaceutical applications.
Q: What are the optimal reaction conditions for maximizing yield and selectivity?
A: Optimal conditions involve using 1,1,2,2-tetrachloroethane as the solvent at a concentration of 0.5 mL per 1 mmol of substrate, with 5A molecular sieves and hexafluoroisopropanol at 70°C for 48 hours, achieving yields up to 93%.
Q: Does this method support diverse substrate scopes for drug discovery?
A: Yes, the method demonstrates excellent substrate tolerance, accommodating various substituents on both the indole enamine and the 2,3-diketone ester, allowing for the generation of a diverse library of chiral bisindole derivatives for biological screening.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-N Axis Chiral Bisindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the N-N axis chiral bisindole scaffold in next-generation anticancer therapies. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate this innovative patent technology into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of intermediate meets the highest industry standards. Our commitment to quality assurance ensures that the complex stereochemistry of these bisindoles is preserved throughout the manufacturing process.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact us to request specific COA data for our reference standards and comprehensive route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a secure, scalable, and cost-effective supply chain for high-purity pharmaceutical intermediates, empowering you to accelerate your journey from discovery to clinic.
