Reversible Amino Protection via Imidazole Hydrochloride Catalysis for Industrial Scale-Up
Reversible Amino Protection via Imidazole Hydrochloride Catalysis for Industrial Scale-Up
The landscape of pharmaceutical intermediate synthesis is constantly evolving, driven by the need for greener, more efficient, and cost-effective methodologies. A significant breakthrough in this domain is documented in Chinese Patent CN110028422B, which introduces a novel method for amino protection utilizing imidazole hydrochloride as a promoter. This technology leverages a reversible Michael addition strategy, allowing for the protection of primary and secondary amines at moderate temperatures and their subsequent deprotection under elevated thermal conditions. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this patent represents a paradigm shift away from traditional, hazardous protecting group chemistries towards a more sustainable and operationally simple catalytic cycle. The core innovation lies in the dual functionality of imidazole hydrochloride, which acts not merely as a passive additive but as an active accelerator that drives the equilibrium forward for protection and backward for deprotection simply by adjusting the thermal energy input.
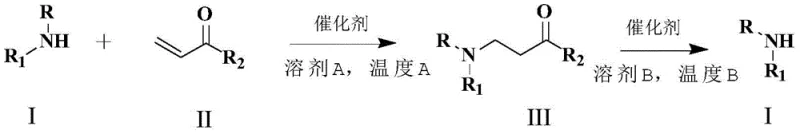
This specific catalytic approach addresses long-standing pain points in the synthesis of complex amine derivatives, particularly regarding the removal of protecting groups which often requires harsh reagents. By establishing a temperature-dependent equilibrium, the process ensures that the protected amine can be generated with high selectivity and subsequently reverted to the free amine with excellent recovery rates. This level of control is critical for the manufacturing of high-purity pharmaceutical intermediates where impurity profiles must be tightly managed. The methodology eliminates the need for transition metals, thereby removing the risk of heavy metal contamination in the final active pharmaceutical ingredient (API), a crucial consideration for regulatory compliance and cost reduction in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the protection of amino groups in organic synthesis has relied heavily on reagents such as benzyl chloroformate (Cbz), di-tert-butyl dicarbonate (Boc), or fluorenylmethoxycarbonyl chloride (Fmoc). While effective, these classical methods suffer from significant drawbacks that impact both the economic and environmental viability of large-scale production. The use of chloroformates, for instance, introduces toxicity concerns and requires stringent safety protocols due to their lachrymatory and corrosive nature. Furthermore, the deprotection steps often necessitate strong acids like trifluoroacetic acid or hydrogenolysis conditions, which can be incompatible with other sensitive functional groups present in complex drug molecules. These harsh conditions frequently lead to side reactions, lowering the overall yield and complicating the purification process, which in turn drives up the cost of goods sold. Additionally, the generation of stoichiometric waste from these protecting groups creates a substantial burden on waste treatment facilities, conflicting with modern green chemistry principles.
The Novel Approach
In stark contrast, the method disclosed in CN110028422B utilizes a catalytic amount of imidazole hydrochloride to facilitate a Michael addition between amines and acrylamide derivatives. This approach fundamentally changes the operational parameters of amino protection. Instead of relying on stoichiometric amounts of expensive and toxic reagents, the process uses a cheap, commercially available salt to drive the reaction. The novelty lies in the reversibility; the same catalyst that promotes the formation of the protected amine at 120°C facilitates the cleavage of the carbon-nitrogen bond at 150°C. This thermal switchability eliminates the need for separate deprotection reagents, streamlining the workflow significantly. For supply chain managers, this translates to a simplified inventory requirement and reduced dependency on specialized, hazardous chemicals. The method's ability to proceed under solvent-free conditions or in benign solvents like N,N-dimethylacrylamide further enhances its profile as a sustainable alternative for the commercial scale-up of complex amine derivatives.
Mechanistic Insights into Imidazole Hydrochloride-Catalyzed Michael Addition
The mechanistic pathway of this transformation is rooted in the activation of the Michael acceptor by the imidazole hydrochloride catalyst. The proton from the hydrochloride salt likely activates the carbonyl oxygen of the acrylamide derivative, increasing the electrophilicity of the beta-carbon. This activation lowers the energy barrier for the nucleophilic attack by the amine nitrogen, facilitating the formation of the carbon-nitrogen bond under relatively mild thermal conditions (90-120°C). The imidazole ring itself may also participate in hydrogen bonding networks that stabilize the transition state, ensuring high regioselectivity for the 1,4-addition product. This precise control over the reaction trajectory is vital for minimizing the formation of oligomeric byproducts, which are common pitfalls in uncatalyzed Michael additions of amines to acrylates.
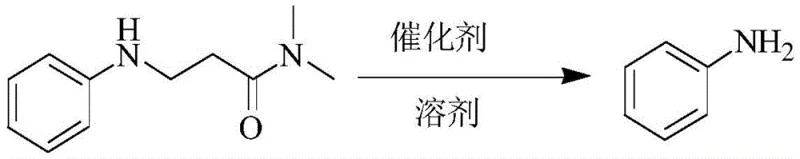
Furthermore, the deprotection mechanism is equally elegant, relying on thermal energy to reverse the addition. At elevated temperatures (130-150°C), the equilibrium shifts back towards the starting materials. The catalyst continues to play a pivotal role by stabilizing the leaving amine group, effectively promoting the retro-Michael reaction. This thermal lability is a distinct advantage over covalent protecting groups that require chemical cleavage. From an impurity control perspective, this mechanism ensures that the deprotection step does not introduce new chemical contaminants, as the only byproducts are the volatile acrylamide derivatives which can be easily removed. This results in a cleaner crude product profile, reducing the load on downstream purification units and enhancing the overall efficiency of the synthesis of high-purity pharmaceutical intermediates.
How to Synthesize Amino Protected Intermediates Efficiently
The practical implementation of this technology involves a straightforward protocol that is easily adaptable to existing reactor setups. The process begins with the mixing of the amine substrate and the acrylamide derivative in the presence of the imidazole hydrochloride catalyst. The reaction is then heated to the optimal temperature range to drive the protection to completion. Following the protection step, the same reaction mixture can potentially be subjected to higher temperatures to effect deprotection if the protected intermediate is used as a transient species, or the protected amine can be isolated for use in subsequent steps. Detailed standard operating procedures regarding specific stoichiometry, solvent choices, and workup techniques are critical for maximizing yield and purity.
- Mix the primary or secondary amine with an acrylamide derivative and 0.3 equivalents of imidazole hydrochloride catalyst in a suitable solvent like DMA.
- Heat the reaction mixture to 120°C to facilitate the Michael addition, monitoring progress via TLC until the starting material is consumed.
- For deprotection, heat the protected intermediate to 150°C with 0.7 equivalents of catalyst to cleave the carbon-nitrogen bond and recover the free amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this imidazole hydrochloride-catalyzed method offers tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By replacing specialized, often imported protecting reagents with a ubiquitous chemical salt, companies can mitigate risks associated with supplier shortages and price volatility. This shift supports a more resilient supply chain, ensuring continuous production even during market disruptions. Moreover, the elimination of toxic reagents reduces the regulatory burden and costs associated with hazardous material handling, storage, and disposal, contributing to significant operational expenditure savings.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the replacement of expensive stoichiometric reagents with a low-cost catalyst. Traditional methods often require more than one equivalent of protecting group reagent, whereas this method uses sub-stoichiometric amounts of imidazole hydrochloride. Additionally, the simplified workup procedure, which often avoids complex aqueous washes required to remove acidic or basic byproducts, reduces solvent consumption and processing time. The absence of heavy metal catalysts also removes the need for expensive scavenging resins or specialized filtration steps, further driving down the cost per kilogram of the final intermediate.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical intermediates is paramount for pharmaceutical manufacturing. This method utilizes raw materials that are commodity chemicals with robust global supply chains, unlike specialized protecting groups that may have limited suppliers. The robustness of the reaction conditions, which tolerate a wide range of functional groups and do not require inert atmospheres or anhydrous conditions, means that production is less susceptible to minor variations in raw material quality or environmental factors. This inherent stability ensures consistent batch-to-batch quality and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but this methodology is inherently scalable. The reaction does not require high-pressure autoclaves or cryogenic cooling, making it compatible with standard glass-lined or stainless steel reactors found in most multipurpose plants. From an environmental standpoint, the atom economy is improved by the reversible nature of the reaction, and the lack of halogenated waste streams (common in Cbz/Boc chemistry) simplifies wastewater treatment. This alignment with green chemistry principles facilitates easier regulatory approval and enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
Understanding the nuances of this new catalytic technology is essential for stakeholders evaluating its integration into their production pipelines. The following questions address common inquiries regarding the scope, safety, and operational parameters of the imidazole hydrochloride-catalyzed amino protection method. These answers are derived directly from the experimental data and technical specifications outlined in the patent literature, providing a factual basis for decision-making.
Q: What are the advantages of using imidazole hydrochloride over traditional protecting groups like Boc or Cbz?
A: Unlike traditional methods requiring toxic reagents like chloroformates or harsh acidic/basic conditions, imidazole hydrochloride catalysis operates under metal-free, mild thermal conditions. It offers a reversible mechanism where protection occurs at lower temperatures (120°C) and deprotection at higher temperatures (150°C), simplifying the synthetic route and reducing hazardous waste.
Q: Is this catalytic system compatible with sensitive functional groups?
A: Yes, the method demonstrates excellent functional group tolerance. It successfully protects various primary and secondary amines, including those with electron-donating or withdrawing substituents, without affecting other sensitive moieties, ensuring high purity of the final pharmaceutical intermediates.
Q: Can this process be scaled for industrial production?
A: Absolutely. The process avoids high-pressure autoclaves and uses commercially available, inexpensive catalysts. The simple workup procedure involving extraction and crystallization makes it highly suitable for commercial scale-up, ensuring consistent supply chain reliability for large-volume manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amino Protected Intermediates Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the imidazole hydrochloride-catalyzed amino protection technology described in CN110028422B. As a leading CDMO partner, we possess the technical expertise to translate this laboratory-scale innovation into robust, industrial-scale processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this novel chemistry are fully realized in a manufacturing environment. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of amino protected intermediates meets the highest standards required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cost-effective and sustainable synthesis route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this technology can optimize your budget without compromising quality. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments for your target compounds.
