Revolutionizing Pharmaceutical Intermediate Production with Safe Palladium-Catalyzed Carbonylation Technology
Introduction to Next-Generation Biheterocyclic Synthesis
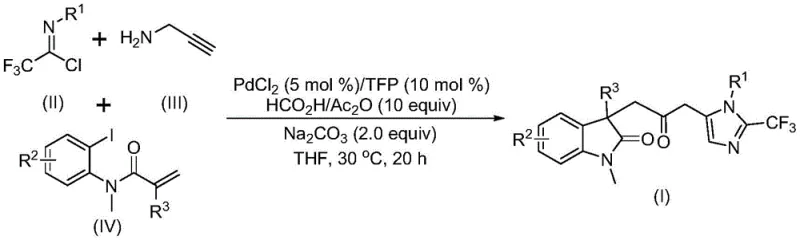
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for safer, more efficient, and cost-effective synthetic routes for complex molecular scaffolds. A significant breakthrough in this domain is documented in Chinese Patent CN115353511A, which discloses a novel multi-component method for synthesizing carbonyl-bridged biheterocyclic compounds. These specific molecular architectures, often comprising indolinone and imidazole moieties linked by a carbonyl bridge, are of immense value in medicinal chemistry due to their presence in numerous bioactive natural products and drug candidates. The patent highlights a transition away from hazardous traditional methods towards a streamlined, palladium-catalyzed cascade reaction that operates under remarkably mild conditions. By leveraging a unique combination of trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives, this technology enables the one-pot construction of multiple chemical bonds simultaneously. For R&D directors and procurement specialists alike, this represents a pivotal shift towards processes that not only enhance molecular diversity but also drastically simplify the supply chain logistics associated with producing high-value fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carbonyl-bridged biheterocyclic compounds has been fraught with significant technical and safety challenges that hinder efficient commercial production. Traditional approaches often rely on the direct coupling of two pre-formed heterocyclic substrates, a strategy that frequently suffers from low atom economy and poor regioselectivity, leading to difficult purification processes and reduced overall yields. Alternatively, oxidative cyclization reactions involving activated methyl-substituted heterocycles often require harsh oxidizing agents and extreme temperatures, which can degrade sensitive functional groups and limit the scope of applicable substrates. Perhaps the most critical bottleneck in conventional carbonylation strategies is the reliance on exogenous carbon monoxide gas. Handling toxic CO gas necessitates specialized high-pressure equipment, rigorous safety protocols, and expensive infrastructure, all of which contribute to inflated operational expenditures and increased regulatory burdens. Furthermore, these legacy methods often struggle with substrate compatibility, failing to tolerate the diverse array of functional groups required for modern drug discovery programs, thereby restricting the chemical space accessible to researchers.
The Novel Approach
In stark contrast to these legacy limitations, the methodology described in patent CN115353511A introduces a transformative palladium-catalyzed carbonylation cascade that elegantly circumvents the need for external carbon monoxide sources. This innovative approach utilizes a mixture of formic acid and acetic anhydride to generate carbon monoxide in-situ, effectively acting as a safe and controllable CO surrogate under atmospheric pressure. The reaction proceeds at a mild temperature of 30°C, significantly reducing energy consumption and thermal stress on the reactants compared to traditional high-temperature protocols. By employing a multi-component strategy, the process constructs the complex biheterocyclic core in a single operational step, merging the formation of the indolinone and imidazole rings with the installation of the bridging carbonyl group. This telescoped synthesis not only accelerates the timeline from raw materials to final product but also minimizes waste generation and solvent usage. The versatility of this system is further evidenced by its ability to accommodate a wide range of substituents, enabling the rapid generation of diverse libraries of trifluoromethyl-containing biheterocycles for biological screening.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific target molecules. The reaction is believed to initiate with the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate, generating a reactive organopalladium intermediate. This species subsequently undergoes an intramolecular Heck-type cyclization to form a five-membered alkyl-palladium(II) complex, establishing the indolinone core structure. Concurrently, the formic acid and acetic anhydride mixture decomposes to release carbon monoxide, which then inserts into the palladium-carbon bond to form an acyl-palladium intermediate. This carbonylation step is the linchpin of the entire transformation, introducing the essential carbonyl bridge that links the two heterocyclic systems. The mechanism further involves the base-promoted intermolecular reaction between trifluoroethylimidoyl chloride and propargylamine to generate a trifluoroacetamidine species in situ. This amidine intermediate then undergoes isomerization and subsequent nucleophilic attack or cyclization facilitated by the activated acyl-palladium complex, ultimately yielding the final carbonyl-bridged biheterocyclic product with high regioselectivity.
From an impurity control perspective, the mild reaction conditions play a pivotal role in maintaining a clean reaction profile. The use of 30°C prevents the thermal degradation of sensitive intermediates that often plagues high-temperature carbonylations, thereby reducing the formation of polymeric byproducts or decomposition artifacts. Furthermore, the specific choice of trifurylphosphine as a ligand enhances the stability of the palladium catalyst and promotes the desired reductive elimination step, minimizing the accumulation of off-cycle palladium black or inactive species. The compatibility with various functional groups such as halogens, nitro groups, and trifluoromethyl moieties suggests that the catalytic cycle is robust against electronic variations in the substrate. For quality assurance teams, this implies a more predictable impurity profile, simplifying the downstream purification via standard silica gel column chromatography. The ability to tune the electronic properties of the substrates without compromising the catalytic turnover allows for the precise engineering of the final molecule's physicochemical properties, a critical factor in optimizing drug candidates for bioavailability and metabolic stability.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
Implementing this advanced synthetic route requires careful attention to reagent stoichiometry and reaction parameters to maximize yield and purity. The protocol outlined in the patent provides a robust framework for executing this multi-component transformation, emphasizing the importance of using anhydrous conditions and high-purity solvents to prevent catalyst deactivation. The reaction is typically conducted in tetrahydrofuran (THF), which offers an optimal balance of solubility for the diverse organic substrates and stability for the palladium catalyst system. Operators should note that while the reaction is tolerant of various substituents, the molar ratios of the three key components—trifluoroethylimidoyl chloride, propargylamine, and acrylamide—are critical for driving the equilibrium towards the desired product. Detailed standardized operating procedures for scaling this reaction from milligram to kilogram quantities are essential for ensuring reproducibility and safety in a manufacturing environment.
- Prepare the reaction mixture by combining palladium chloride catalyst, trifurylphosphine ligand, sodium carbonate base, and the specific substrate ratios in an organic solvent like THF.
- Add the carbon monoxide substitute mixture consisting of formic acid and acetic anhydride along with trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives.
- Maintain the reaction at a mild temperature of 30°C for 12 to 20 hours, followed by filtration and column chromatography purification to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers substantial strategic advantages that extend beyond mere chemical efficiency. The elimination of toxic carbon monoxide gas from the process inventory removes a major safety hazard and the associated regulatory compliance costs, directly contributing to a safer working environment and lower insurance premiums. Moreover, the starting materials utilized in this synthesis, such as propargylamine and various acrylamide derivatives, are commodity chemicals that are readily available from multiple global suppliers, mitigating the risk of supply chain disruptions caused by reliance on exotic or single-source reagents. The mild reaction temperature of 30°C significantly reduces the energy footprint of the manufacturing process, aligning with corporate sustainability goals and lowering utility costs associated with heating and cooling large-scale reactors. Additionally, the high atom economy of the multi-component approach means that less raw material is wasted as byproduct, leading to more efficient utilization of expensive precursors and reducing the volume of chemical waste requiring disposal.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the synthetic sequence and the removal of hazardous reagents. By avoiding the need for high-pressure autoclaves and specialized gas handling infrastructure required for traditional carbonylation, capital expenditure for new production lines is significantly reduced. The use of inexpensive palladium chloride as the catalyst precursor, combined with the ability to recover and recycle the solvent, further drives down the cost of goods sold (COGS). Furthermore, the high yields reported across a broad substrate scope mean that less starting material is required to produce a given amount of API intermediate, directly improving the margin profile for the final pharmaceutical product. The streamlined one-pot nature of the reaction also reduces labor costs and reactor occupancy time, allowing for higher throughput in existing manufacturing facilities without the need for additional hardware investment.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of stable, shelf-stable reagents that do not require cryogenic storage or special transportation permits. Unlike carbon monoxide, which poses logistical nightmares for transport and storage, the formic acid and acetic anhydride mixture used as the CO source is easy to handle and source globally. This stability ensures consistent production schedules and reduces the likelihood of delays caused by regulatory hold-ups on hazardous material shipments. The broad substrate tolerance also allows for flexibility in sourcing; if a specific substituted acrylamide becomes unavailable, alternative analogs can often be substituted without re-optimizing the entire process, providing a buffer against market volatility. This flexibility is crucial for maintaining continuous supply to downstream pharmaceutical customers who depend on just-in-time delivery models for their clinical and commercial programs.
- Scalability and Environmental Compliance: The scalability of this process is evidenced by its successful demonstration at the gram scale in the patent literature, with clear pathways for expansion to multi-kilogram and tonne-level production. The mild conditions minimize the formation of thermal runaway risks, making the scale-up process inherently safer and easier to manage from an engineering perspective. From an environmental standpoint, the reduction in waste generation and the avoidance of toxic gases align with increasingly stringent global environmental regulations, such as REACH in Europe and TSCA in the United States. The simplified workup procedure, involving basic filtration and chromatography, reduces the consumption of auxiliary chemicals and solvents during purification. This 'green chemistry' profile not only enhances the corporate image but also future-proofs the manufacturing process against tightening environmental legislation, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, offering clarity on the practical aspects of adopting this technology. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What is the primary advantage of this synthesis method over traditional carbonylation?
A: The primary advantage is safety and operational simplicity. Unlike traditional methods that require toxic and high-pressure carbon monoxide gas, this patent utilizes a formic acid and acetic anhydride mixture to generate carbon monoxide in-situ under mild atmospheric conditions.
Q: What types of substrates are compatible with this palladium-catalyzed reaction?
A: The reaction demonstrates excellent substrate compatibility, tolerating various functional groups including alkyl, alkoxy, halogen, trifluoromethyl, and nitro groups on the aromatic rings, allowing for the synthesis of diverse derivatives.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly mentions that the method has been expanded to gram-scale reactions with high efficiency, indicating strong potential for commercial scale-up due to the use of cheap, readily available starting materials and mild reaction conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodologies described in patent CN115353511A for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market supply is seamless and efficient. Our state-of-the-art facilities are equipped to handle complex palladium-catalyzed reactions with the utmost precision, adhering to stringent purity specifications and rigorous QC labs to guarantee the quality of every batch. We understand that the integrity of your supply chain depends on the reliability of your chemical partners, and we are committed to delivering consistent, high-quality carbonyl-bridged biheterocyclic compounds that meet the exacting standards of the global pharmaceutical industry.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project needs. By leveraging our expertise, you can access a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this safer, more efficient manufacturing process. We encourage potential partners to contact us to request specific COA data for related compounds and comprehensive route feasibility assessments. Let us collaborate to accelerate your drug development timeline and secure a robust, cost-effective supply of these critical building blocks for your next-generation therapeutics.
