Advanced Pd-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Pharmaceutical Applications
Advanced Pd-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Pharmaceutical Applications
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, safer, and cost-effective synthetic routes. A significant breakthrough in this domain is documented in Chinese Patent CN115353511A, which discloses a novel multi-component method for synthesizing carbonyl-bridged biheterocyclic compounds. These complex molecular scaffolds, particularly those combining indolinone and imidazole motifs, are of immense value in medicinal chemistry due to their broad-spectrum biological activities. The patent outlines a transition metal palladium-catalyzed carbonylation cascade reaction that utilizes cheap and readily available starting materials such as trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives. This technology represents a paradigm shift from traditional methods by operating under mild conditions without the need for toxic carbon monoxide gas, thereby addressing critical safety and environmental concerns in fine chemical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of biheterocyclic systems has relied on three primary strategies, each fraught with significant operational challenges for large-scale manufacturing. The first approach involves the direct coupling of two pre-formed heterocyclic substrates, which often suffers from low atom economy and requires harsh reaction conditions that can degrade sensitive functional groups. The second strategy employs oxidative cyclization of substrates containing dual nucleophiles with activated methyl-substituted heterocycles; however, this route frequently necessitates stoichiometric amounts of oxidants, generating substantial waste and complicating downstream purification. The third conventional method utilizes transition metal-catalyzed tandem cyclization, which, while efficient, often struggles with the specific incorporation of carbonyl bridges in a single pot. For carbonyl-bridged biheterocycles specifically, applying traditional carbonylation reactions has remained a formidable challenge due to the requirement for high-pressure carbon monoxide gas, posing severe safety risks and requiring specialized high-pressure equipment that limits accessibility for many contract manufacturing organizations.
The Novel Approach
In stark contrast to these legacy techniques, the methodology described in CN115353511A introduces a streamlined, one-pot multi-component reaction that elegantly constructs the carbonyl-bridged bis-heterocyclic framework. By leveraging a palladium catalyst system with a trifurylphosphine ligand, the process facilitates a cascade sequence that simultaneously forms multiple chemical bonds. A critical innovation is the use of a formic acid and acetic anhydride mixture as a safe carbon monoxide surrogate, effectively bypassing the hazards associated with handling toxic CO gas cylinders. The reaction proceeds efficiently at a mild temperature of 30°C in common organic solvents like tetrahydrofuran (THF), demonstrating exceptional substrate compatibility. This approach not only simplifies the operational workflow but also significantly enhances the safety profile of the synthesis, making it highly attractive for the production of high-purity pharmaceutical intermediates.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors aiming to optimize process parameters. The reaction likely initiates with the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate. This is followed by an intramolecular Heck reaction, generating a divalent alkyl-palladium intermediate. Subsequently, carbon monoxide, released in situ from the decomposition of the formic acid and acetic anhydride mixture, inserts into the palladium-carbon bond to form an acyl-palladium intermediate. Parallel to this organometallic cycle, the base-promoted reaction between trifluoroethylimidoyl chloride and propargylamine generates a trifluoroacetamidine compound, which undergoes isomerization. The final step involves the activation of this amidine by the acyl-palladium species, triggering an intramolecular cyclization that furnishes the target carbonyl-bridged biheterocyclic compound with high regioselectivity.
From an impurity control perspective, this mechanism offers distinct advantages. The mild reaction temperature of 30°C minimizes thermal degradation pathways that often lead to complex impurity profiles in high-temperature syntheses. Furthermore, the use of specific ligands like trifurylphosphine helps stabilize the palladium center, reducing the formation of palladium black and other metal-related particulates that can contaminate the final API intermediate. The broad functional group tolerance observed in the patent data, accommodating substituents such as halogens, trifluoromethyl groups, and nitro groups, suggests that the catalytic cycle is robust against electronic variations in the substrate. This robustness ensures consistent quality and reduces the burden on purification teams, as fewer side reactions occur compared to more aggressive oxidative methods.
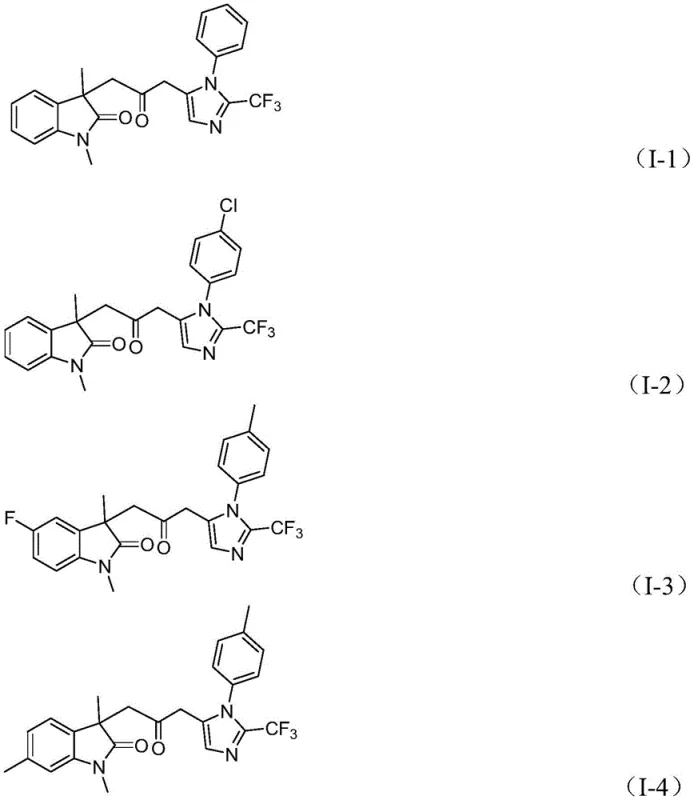
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent ratios and reaction monitoring to maximize yield. The patent specifies a molar ratio where trifluoroethylimidoyl chloride serves as the limiting reagent, typically used in a 1:2:1.5 ratio relative to propargylamine and acrylamide, ensuring complete consumption of the valuable imidoyl chloride. The catalyst loading is optimized at approximately 5 mol% for PdCl2 and 10 mol% for the ligand, balancing cost and activity. While the general procedure is straightforward, precise control over the addition of the CO surrogate mixture is vital to maintain steady carbonylation pressure within the closed system. For detailed standard operating procedures and specific work-up protocols tailored to your facility's capabilities, please refer to the comprehensive guide below.
- Combine palladium chloride catalyst, trifurylphosphine ligand, sodium carbonate base, and a carbon monoxide surrogate mixture of formic acid and acetic anhydride in an organic solvent such as THF.
- Add the substrates including trifluoroethylimidoyl chloride, propargylamine, and the specific acrylamide derivative to the reaction mixture under inert atmosphere.
- Stir the reaction at 30°C for 12 to 20 hours, followed by filtration, silica gel treatment, and column chromatography purification to isolate the final biheterocyclic product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic benefits beyond mere chemical novelty. The primary driver for cost optimization lies in the raw material selection; starting materials like propargylamine and acrylamide derivatives are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency. Furthermore, the elimination of high-pressure carbon monoxide infrastructure removes a significant capital expenditure barrier, allowing production to occur in standard glass-lined reactors rather than specialized autoclaves. This flexibility drastically simplifies the manufacturing footprint and reduces the regulatory burden associated with handling toxic gases, leading to substantial operational cost savings.
- Cost Reduction in Manufacturing: The economic viability of this process is enhanced by the use of inexpensive palladium chloride as the catalyst source compared to more exotic palladium complexes. Additionally, the reaction achieves high conversion rates, often exceeding 80% for optimized substrates, which minimizes raw material waste. The simplified post-treatment process, involving basic filtration and standard column chromatography, reduces solvent consumption and labor hours compared to multi-step sequences requiring cryogenic conditions or extensive aqueous workups. These factors collectively contribute to a lower cost of goods sold (COGS) for the final biheterocyclic intermediates.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the robustness of the reaction conditions. Since the synthesis does not rely on cryogenic temperatures or ultra-dry environments, it is less susceptible to disruptions caused by utility failures or seasonal variations in plant cooling capacity. The compatibility with common solvents like THF and acetonitrile ensures that solvent supply chains remain stable and cost-effective. Moreover, the ability to tolerate various functional groups means that supply chain managers can source alternative substrate grades without compromising the integrity of the final product, providing greater flexibility in vendor management.
- Scalability and Environmental Compliance: The patent explicitly notes the successful expansion of this method to gram-scale reactions, indicating a clear path towards kilogram and ton-scale production. The absence of toxic CO gas emissions aligns with increasingly stringent environmental regulations, facilitating easier permitting and compliance audits. The generation of waste is minimized through high atom efficiency and the use of recyclable solvents, supporting sustainability goals. This scalability ensures that the technology can meet the growing demand for these bioactive scaffolds in the pharmaceutical sector without encountering the bottlenecks typical of batch processes requiring specialized equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed carbonylation technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a reliable basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the key advantages of this Pd-catalyzed method over traditional biheterocycle synthesis?
A: This method eliminates the need for toxic carbon monoxide gas by using a safe formic acid/acetic anhydride surrogate. It operates under mild conditions (30°C) and achieves high yields (up to 92%) with excellent functional group tolerance, unlike harsh oxidative cyclization methods.
Q: Is this synthesis scalable for industrial production of pharmaceutical intermediates?
A: Yes, the patent explicitly demonstrates scalability to gram-scale reactions. The use of commercially available starting materials and simple post-treatment processes like filtration and column chromatography supports robust commercial scale-up.
Q: What types of substituents are compatible with this carbonylation cascade reaction?
A: The reaction exhibits broad substrate compatibility, tolerating various substituents on the aryl rings including alkyl groups, halogens (fluoro, chloro, bromo), alkoxy groups, trifluoromethyl, and nitro groups at ortho, meta, or para positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists has extensively evaluated the protocol described in CN115353511A and possesses the technical expertise to adapt this Pd-catalyzed carbonylation cascade for your specific project needs. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of carbonyl-bridged biheterocyclic compounds meets the highest industry standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this cutting-edge technology for your next generation of therapeutics. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing capabilities can drive efficiency and reliability in your supply chain.
