Advanced Chemical Synthesis of Spermidine: A Scalable Route for Industrial Production
Advanced Chemical Synthesis of Spermidine: A Scalable Route for Industrial Production
The global demand for polyamines, particularly spermidine, has surged due to their critical role in cellular growth, differentiation, and anti-aging research. As a vital physiologically active substance found in microorganisms, plants, and animals, spermidine is increasingly recognized for its potential in counteracting age-related diseases such as cardiovascular disorders and neurodegenerative conditions. However, securing a reliable supply of high-purity spermidine has historically been challenging due to the limitations of extraction and biosynthetic methods. Patent CN113548971A introduces a groundbreaking chemical synthesis process that addresses these bottlenecks by utilizing a concise reductive amination strategy. This innovative approach not only streamlines the production workflow but also establishes a new standard for efficiency in the manufacturing of this valuable pharmaceutical intermediate.
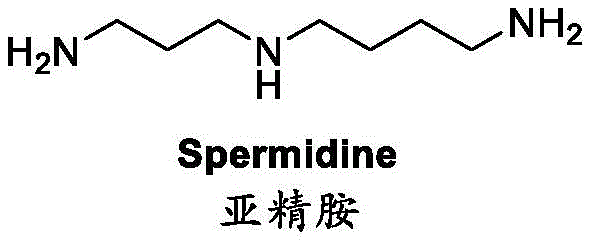
Traditional methods for obtaining spermidine often rely on extraction from natural sources or enzymatic synthesis, both of which suffer from inherently low yields and complex downstream processing. Extraction from biological matrices is inefficient due to the low natural content of spermidine, while biosynthetic routes face hurdles related to enzyme stability and difficult product isolation. Consequently, the industry has long sought a robust chemical synthesis pathway. Existing chemical methods, such as the Michael addition of 1,4-butanediamine with acrylonitrile followed by hydrogenation, are plagued by low conversion rates and the formation of stubborn byproducts that are difficult to separate. Furthermore, alternative routes involving lithium aluminum hydride reductions or Mitsunobu reactions introduce significant safety hazards and environmental burdens, rendering them unsuitable for large-scale commercial operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The historical reliance on multi-step synthetic pathways has created substantial barriers to entry for manufacturers aiming to produce spermidine at a commercial scale. For instance, routes utilizing lithium aluminum hydride require strictly anhydrous conditions and pose severe safety risks due to the pyrophoric nature of the reagent, driving up operational costs and requiring specialized equipment. Similarly, methods employing Mitsunobu reactions generate stoichiometric amounts of triphenylphosphine oxide waste, which is notoriously difficult to remove from the final product, thereby compromising purity and increasing waste disposal costs. These conventional approaches often result in long reaction sequences with cumulative yield losses, making the final product economically unviable for widespread application in the nutraceutical and pharmaceutical sectors.
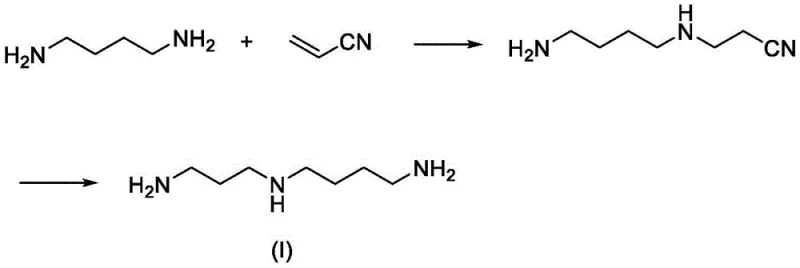
The Novel Approach
In stark contrast, the methodology disclosed in CN113548971A offers a streamlined solution by leveraging a highly efficient reductive amination between 3-phthalimidopropionaldehyde and N-tert-butoxycarbonyl-1,4-butanediamine. This strategy effectively bypasses the need for hazardous reducing agents and complex coupling reactions. By carefully selecting orthogonal protecting groups, the process ensures high regioselectivity, minimizing the formation of oligomeric byproducts that typically plague polyamine synthesis. The resulting intermediate can be easily purified and subsequently deprotected under mild conditions to yield spermidine or its salts. This novel route represents a paradigm shift, offering a balance of safety, efficiency, and cost-effectiveness that is essential for modern fine chemical manufacturing.
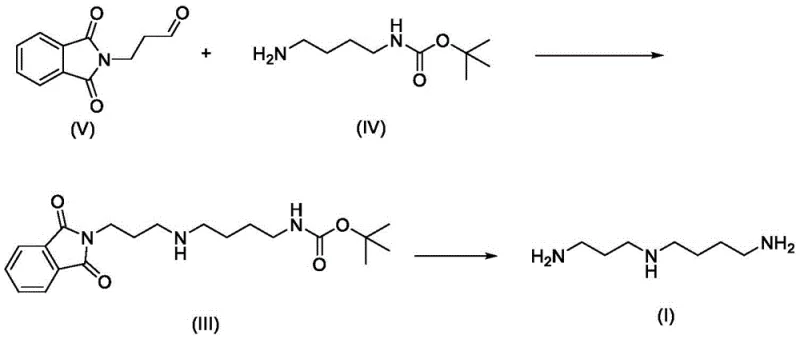
Mechanistic Insights into Reductive Amination and Deprotection
The core of this innovative synthesis lies in the precise execution of the reductive amination step, where the aldehyde functionality of 3-phthalimidopropionaldehyde reacts with the primary amine of the protected butanediamine. This reaction proceeds through the formation of an imine or iminium ion intermediate, which is subsequently reduced in situ to form the secondary amine linkage. The choice of reducing agent is critical; the patent highlights the efficacy of sodium triacetoxyborohydride or catalytic hydrogenation systems using Pd/C or Raney-Ni. These reagents operate under mild pH conditions, preventing the hydrolysis of the sensitive Boc protecting group during the bond-forming event. This mechanistic control is paramount for maintaining the integrity of the molecular scaffold and ensuring that the final product possesses the correct chain length and amine spacing required for biological activity.
Furthermore, the deprotection strategy employed in the final stages demonstrates a sophisticated understanding of orthogonal chemistry. The phthalimide group serves as a robust protector for the propylamine segment, stable against the basic conditions of the amination but readily cleavable by hydrazine hydrate or acid hydrolysis. Simultaneously, the Boc group on the butylamine segment provides stability during the initial coupling but can be removed under acidic conditions to reveal the terminal primary amine. This dual-protection scheme allows for the sequential unveiling of reactive sites, effectively suppressing polymerization and ensuring that the final spermidine molecule is obtained with high structural fidelity. The ability to isolate the intermediate as a stable trihydrochloride salt further enhances the process by facilitating purification and storage prior to final liberation.
How to Synthesize Spermidine Efficiently
Implementing this synthesis requires careful attention to reaction parameters, particularly temperature control and stoichiometry during the reductive amination phase. The process begins with the preparation of the key aldehyde and amine building blocks, followed by their convergence in a polar protic solvent. Detailed operational guidelines regarding solvent selection, catalyst loading, and workup procedures are essential for replicating the high yields reported in the patent literature. Manufacturers must adhere to strict protocols to manage the exothermic nature of the reduction and ensure complete conversion before proceeding to the deprotection stage. For a comprehensive breakdown of the standardized operating procedures, please refer to the technical guide below.
- Prepare key intermediates: Synthesize 3-phthalimidopropionaldehyde (V) from phthalimide and acrolein, and prepare N-tert-butoxycarbonyl-1,4-butanediamine (IV) via mono-protection of 1,4-butanediamine.
- Perform Reductive Amination: React intermediate (V) and (IV) in a solvent like methanol or ethanol using a reducing agent such as sodium triacetoxyborohydride or catalytic hydrogenation to form the protected spermidine intermediate (III).
- Deprotection and Isolation: Remove the phthalimide and Boc protecting groups from intermediate (III) using acid hydrolysis or hydrazine hydrate to yield spermidine trihydrochloride (II), which can be further liberated to free base spermidine (I).
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthesis route offers transformative benefits that directly impact the bottom line. By eliminating the need for exotic or hazardous reagents like lithium aluminum hydride, manufacturers can drastically simplify their safety protocols and reduce the capital expenditure associated with specialized containment infrastructure. The use of commodity chemicals such as methanol, ethanol, and common amines ensures a stable and resilient supply chain, mitigating the risks associated with raw material shortages. Moreover, the simplified purification steps reduce the consumption of solvents and energy, leading to substantial cost savings in utility usage and waste treatment. This process optimization translates into a more competitive pricing structure for the final spermidine product, enabling broader market penetration.
- Cost Reduction in Manufacturing: The elimination of expensive and difficult-to-handle reagents significantly lowers the direct material costs associated with production. By avoiding the Mitsunobu reaction, the process removes the burden of triphenylphosphine oxide removal, which traditionally requires extensive chromatography or recrystallization steps. This reduction in downstream processing complexity allows for higher throughput and lower labor costs, creating a leaner manufacturing model that maximizes resource efficiency without compromising product quality.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials ensures that production schedules are not disrupted by the volatility of niche chemical markets. The robustness of the reaction conditions means that the process is less susceptible to minor fluctuations in environmental parameters, leading to consistent batch-to-batch performance. This reliability is crucial for long-term supply agreements with pharmaceutical clients who demand unwavering quality and timely delivery, thereby strengthening the manufacturer's reputation as a dependable partner in the global value chain.
- Scalability and Environmental Compliance: The synthetic route is inherently designed for scale-up, utilizing reaction types that are well-understood in large-scale chemical engineering. The reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, reducing the liability and cost associated with waste disposal. The ability to produce spermidine trihydrochloride as a stable intermediate further aids in logistics, allowing for flexible production scheduling and inventory management to meet fluctuating market demands efficiently.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its potential for integration into existing production lines. The following questions address common concerns regarding purity, scalability, and regulatory compliance. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering a transparent view of the technology's capabilities and limitations.
Q: What are the primary advantages of this new spermidine synthesis route compared to traditional methods?
A: The novel route described in patent CN113548971A avoids hazardous reagents like lithium aluminum hydride and complex reactions like Mitsunobu coupling. It utilizes a straightforward reductive amination strategy that simplifies purification, reduces three-waste generation, and significantly lowers raw material costs, making it highly suitable for industrial scale-up.
Q: How does this process ensure high purity for pharmaceutical applications?
A: The process employs robust protecting group strategies (Boc and Phthalimide) that allow for selective reactions and easy removal of impurities. The final deprotection steps yield spermidine trihydrochloride with high molar yields and purity levels exceeding 97% after rectification, meeting stringent quality specifications for API intermediates.
Q: Is this synthesis method scalable for commercial production?
A: Yes, the method is explicitly designed for industrial mass production. It uses common solvents like methanol and ethanol, operates under mild temperature conditions, and avoids difficult-to-remove catalysts or byproducts, ensuring a reliable supply chain and consistent batch-to-batch quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spermidine Supplier
As the demand for high-quality polyamines continues to grow, partnering with an experienced CDMO is essential for navigating the complexities of chemical synthesis. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of spermidine meets the highest international standards for pharmaceutical and nutraceutical applications.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific requirements. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating how this efficient route can optimize your procurement budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable supply of this critical bioactive compound.
