Scalable Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Intermediates for Global Pharma Supply Chains
The pharmaceutical and agrochemical industries continuously seek robust synthetic pathways for nitrogen-containing heterocycles, particularly 1,2,4-triazoles, due to their pervasive presence in bioactive molecular frameworks. As highlighted in recent literature, these structures serve not only as critical pharmacophores in drug candidates but also as versatile ligands in material science applications such as OLEDs. A pivotal advancement in this domain is disclosed in Chinese Patent CN113307790B, which details a highly efficient preparation method for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds. This technology represents a significant leap forward for any reliable pharmaceutical intermediate supplier aiming to streamline the production of complex heterocyclic scaffolds. The innovation lies in its ability to construct these valuable cores through a direct oxidative cyclization strategy that bypasses the limitations of classical multistep syntheses.
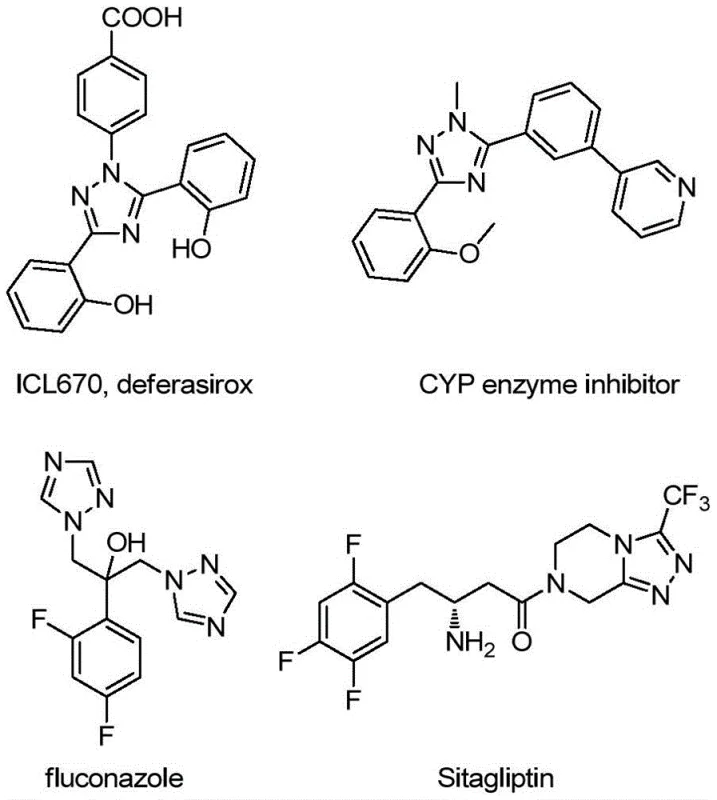
The strategic value of this patent extends beyond mere academic interest; it addresses critical bottlenecks in cost reduction in pharmaceutical intermediate manufacturing. By leveraging cheap and readily available starting materials such as 2-methylquinoline and trifluoroethylimine hydrazide, the process offers a commercially viable route that is amenable to large-scale operations. For R&D directors and procurement managers alike, understanding the nuances of this metal-free catalytic system is essential for evaluating potential supply chain partnerships and optimizing the synthesis of next-generation therapeutic agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolyl-substituted 1,2,4-triazoles has been plagued by inefficiencies that render them unsuitable for modern industrial demands. Traditional methodologies often rely on quinoline-2-carboxylic acid as a primary raw material, necessitating a cumbersome five-step reaction sequence to achieve the final target molecule. This legacy approach suffers from a dismal total yield of approximately 17%, which is economically prohibitive for large-scale production. Furthermore, these conventional routes typically demand severe reaction conditions, including stringent temperature controls and the use of hazardous reagents, which complicate safety protocols and waste management. The reliance on such inefficient pathways results in excessive resource consumption and generates significant environmental burdens, making it difficult for manufacturers to maintain competitive pricing while adhering to green chemistry principles.
The Novel Approach
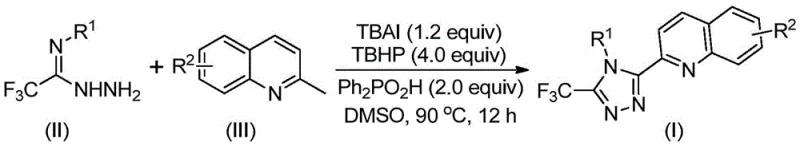
In stark contrast, the novel methodology described in the patent introduces a streamlined, one-pot oxidative cyclization process that dramatically enhances efficiency and sustainability. By utilizing 2-methylquinoline and trifluoroethylimine hydrazide as starting materials, the reaction proceeds via a direct C-H functionalization and cyclization mechanism promoted by tetrabutylammonium iodide (TBAI) and tert-butyl peroxide (TBHP). This approach eliminates the need for pre-functionalized precursors and reduces the synthetic steps significantly. The reaction operates under relatively mild conditions, heating to just 80-100°C in a polar aprotic solvent like DMSO, and crucially, it avoids the use of toxic heavy metal catalysts. This metal-free nature simplifies the downstream purification process, as there is no need for expensive metal scavenging resins or complex extraction procedures to meet strict residual metal specifications required by regulatory bodies.
Mechanistic Insights into TBAI/TBHP Promoted Oxidative Cyclization
The core of this technological breakthrough lies in the intricate interplay between the iodide catalyst and the peroxide oxidant, which facilitates a radical-mediated transformation. Mechanistically, the tetrabutylammonium iodide serves as a source of iodine species that, in the presence of tert-butyl peroxide, generates reactive radicals capable of activating the methyl group on the 2-methylquinoline substrate. This initial oxidation converts the methyl group into an aldehyde intermediate in situ, which then undergoes condensation with the trifluoroethylimine hydrazide to form a dehydrated hydrazone. Subsequent oxidative iodination and intramolecular electrophilic substitution drive the cyclization process, ultimately leading to aromatization and the formation of the stable 1,2,4-triazole ring. The inclusion of diphenylphosphinic acid as an additive further stabilizes the reaction environment, likely by modulating the acidity and enhancing the electrophilicity of the intermediates, thereby ensuring high conversion rates and selectivity.
From an impurity control perspective, this mechanism offers distinct advantages over transition-metal catalyzed alternatives. The absence of metals like copper or palladium removes a major source of potential contamination, such as metal-ligand complexes or colloidal metal particles, which are notoriously difficult to remove from the final API intermediate. Furthermore, the radical nature of the reaction, while powerful, is well-controlled by the stoichiometric balance of the oxidant and the iodide source, minimizing the formation of over-oxidized byproducts or polymerization side products. The use of DMSO as the solvent not only ensures excellent solubility for the diverse range of substrates but also participates in stabilizing the polar transition states, contributing to the observed high yields ranging from 51% to 97% across various substituted derivatives. This robustness allows for the tolerance of diverse functional groups, including halogens, nitro groups, and alkoxy substituents, expanding the chemical space accessible to medicinal chemists.
How to Synthesize 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Efficiently
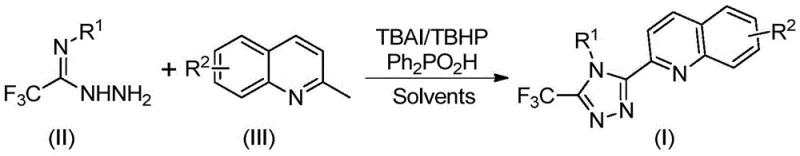
Implementing this synthesis in a laboratory or pilot plant setting requires precise adherence to the optimized parameters outlined in the patent to ensure reproducibility and safety. The process is designed to be operationally simple, avoiding the need for specialized equipment such as gloveboxes or high-pressure reactors, which lowers the barrier to entry for contract manufacturing organizations. The standard protocol involves charging the reactor with the specific molar ratios of reagents—typically a slight excess of the hydrazide derivative relative to the quinoline substrate—to drive the equilibrium towards product formation. The reaction mixture is then heated to the optimal temperature window of 80-100°C and maintained for a duration of 8 to 14 hours, depending on the specific electronic nature of the substituents on the aromatic rings.
- Mix tetrabutylammonium iodide (TBAI), tert-butyl peroxide (TBHP), diphenylphosphinic acid, trifluoroethylimine hydrazide, and 2-methylquinoline in DMSO solvent.
- Heat the reaction mixture to 80-100°C and stir for 8-14 hours to allow oxidative cyclization to proceed.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic benefits that extend far beyond the laboratory bench. The shift towards a metal-free catalytic system fundamentally alters the cost structure of manufacturing these complex intermediates. By eliminating the requirement for precious metal catalysts, companies can avoid the volatility associated with the pricing of metals like palladium or rhodium, leading to more predictable budgeting and long-term cost stability. Additionally, the simplified workup procedure, which involves basic filtration and chromatography rather than complex metal scavenging, reduces the consumption of auxiliary materials and shortens the overall production cycle time, thereby enhancing throughput capacity without requiring additional capital investment in infrastructure.
- Cost Reduction in Manufacturing: The economic impact of removing heavy metal catalysts from the process cannot be overstated. In traditional synthesis, the cost of the catalyst itself, combined with the expensive scavengers required to reduce metal residues to ppm levels for pharmaceutical compliance, constitutes a significant portion of the COGS (Cost of Goods Sold). This new route utilizes inexpensive, commodity-grade chemicals like TBAI and TBHP, which are widely available and cost-effective. Furthermore, the high atom economy and improved yields mean that less raw material is wasted, directly improving the margin profile for the final product. The ability to run the reaction in common solvents like DMSO, which are cheaper and easier to recover than specialized fluorinated solvents often used in trifluoromethylation reactions, adds another layer of cost efficiency to the overall process.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the availability and stability of raw materials. The starting materials for this synthesis, specifically 2-methylquinoline and various substituted anilines used to prepare the hydrazide, are bulk chemicals produced by numerous global suppliers. This diversity in the supply base mitigates the risk of shortages that can occur when relying on niche, custom-synthesized building blocks. Moreover, the robustness of the reaction conditions—specifically the lack of sensitivity to moisture and oxygen—means that the manufacturing process is less prone to batch failures caused by environmental fluctuations. This reliability ensures consistent delivery schedules and reduces the need for safety stock, allowing for a leaner and more responsive inventory management strategy.
- Scalability and Environmental Compliance: Scaling chemical processes from gram to ton scale often introduces unforeseen challenges, particularly regarding heat management and exothermic control. However, the moderate temperature range (80-100°C) and the use of aqueous tert-butyl peroxide solutions suggest a manageable thermal profile that is conducive to scale-up in standard stainless steel reactors. From an environmental standpoint, the avoidance of heavy metals aligns perfectly with increasingly stringent global regulations regarding wastewater discharge and solid waste disposal. The reduction in hazardous waste generation not only lowers disposal costs but also enhances the company's sustainability profile, a factor that is becoming increasingly important for multinational corporations when selecting vendors. The simplicity of the post-treatment, involving silica gel mixing and chromatography, is a standard unit operation that can be easily adapted to continuous processing or large-batch crystallization methods as the volume increases.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of what partners can expect when integrating this route into their supply chains. Understanding these details is crucial for making informed decisions about process validation and regulatory filing strategies.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the patented method utilizes a metal-free catalytic system based on tetrabutylammonium iodide (TBAI) and tert-butyl peroxide (TBHP), eliminating the need for costly heavy metal removal steps.
Q: What are the typical reaction yields for this triazole formation?
A: The process demonstrates high efficiency, with reported isolated yields ranging from 51% to 97% across various substituted substrates, significantly outperforming traditional multi-step routes.
Q: Is the reaction sensitive to moisture or oxygen?
A: The method is robust and does not require strict anhydrous or anaerobic conditions, allowing for simpler operational setups and reduced infrastructure costs compared to sensitive organometallic reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the metal-free oxidative cyclization technology disclosed in CN113307790B for the production of high-value heterocyclic intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical building blocks. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, ensuring that every batch of 3-quinolyl-5-trifluoromethyl-1,2,4-triazole meets the exacting standards required for pharmaceutical development and commercial manufacturing.
We invite global innovators to collaborate with us to leverage this efficient synthetic route for their drug discovery programs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in process optimization can accelerate your project timelines and reduce your overall development costs.
